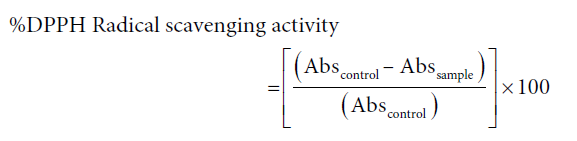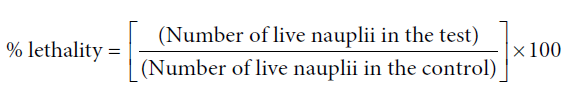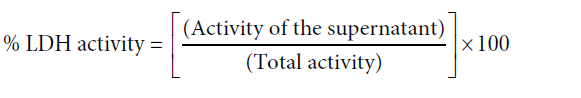DOI: 10.31038/CST.2017231
Abstract
Background: Evidence of cancer cure or improvement of quality of life in cancer patients has been reported using prescriptions based on traditional knowledge, but not scientifically investigated. The objectives of the current study is to evaluate the anti-proliferative action of a poly herbal drug which is currently used in traditional medicine name “Le Pana Guliya (LPG)”
Methods: The aqueous extract of LPG was freeze dried. Total phenolic content was assayed using Folin Ciocalteu reagent. Antioxidant activity of the extract was evaluated using DPPH radical scavenging assay in vitro. Brine shrimp assay was conducted to determine the cytotoxicity (LD50) of the LPG. The cell viability was determined by MTT assay and cytotoxicity by LDH leakage assay after 24 and 48 hours incubation with LPG. Morphological Changes after the treatment of the drug was observed using an inverted light microscope. RD, MCF-7 cells and CC1 cells were used in all experiments.
Results: The TPC of the LPG was 5.31 ± 0.14 W/W% of GAE and antioxidant capacity is comparable to ascorbic acid. LPG exhibited strong cytotoxic activity against RD and MCF-7 cell lines with MTT assay. A 50% leakage of LDH was observed at concentrations less than 30μg/mL and 10 μg/mL for both RD and MCF-7 cells respectively after 24 hour exposure. While, LPG exhibited strong cytotoxic activity against RD and MCF-7 cells the brine shrimp and CC1 cells results (EC50>100μg/mL) suggest that the LPG have minimum cytotoxicity towards the normal healthy cells.
Conclusion: The present study provides evidence for potent anti cancer activity of LPG on RD and MCF-7 cells. The brine shrimp bio-assay and CC1 cells results suggests that the extract have minimum cytotoxicity towards the normal healthy cells.
Keywords
Anti-proliferative activity, Cytotoxicity, MTT assays, LDH assay, Brine shrimp assay, Light microscopy, RD cells, MCF-7, CC1, DPPH
Introduction
Natural products have played an imperative role in the lead-finding of candidates for the development of present-day cancer chemotherapy due to their low toxicity and side effects. They offer a valuable source of a wide variety of chemical structures with biological activities (lead molecules) for the development of novel drugs [1]. Approximately 60% of all drugs currently undergoing clinical trials for cancer treatment are natural products or compounds derived from natural products [2, 3]. These substances embrace some of the most exciting new chemotherapeutic agents currently available for use in a clinical setting [3].
Sri Lanka is gifted with many plants which have vast medicinal value. Use of plants for medicinal preparations is a primary part of the Sri Lankan cultural life and this is implausible to change in the years to come. The Sri Lankan traditional medicine system shows extensive use of plant products in cancer treatment. A significant surge of interest in chemoprevention research has thus led to the discovery of many phytochemicals as successful chemo preventive agents.
Many traditional healers and folklore medical practitioners in the country have been treating cancer patients for many years using various medicinal plant species. There is evidence of case reports on cancer cure using traditional knowledge, but they are not scientifically investigated. In addition to use of a single plant, poly herbal formulations of drugs are intensively used in Sri Lanka. The poly herbal drug ‘Le Pana Guliya’ (LPG) found in Ola leaf inscriptions is prescribed to treat different types of cancers by traditional doctors.This study was aimed at investigating of its cytotoxic effect against two different cancer cell lines compared with healthy CC1 cells.
Materials And Methods
Chemicals and equipment
Chemicals needed for cell culture and cytotoxicity studies were purchased from Sigma –Aldrich (St Louis, MO63178, USA). 1-Diphenyl-2-picrylhydrazyl (DPPH), TritonX-100, was purchased from Fluka. Tris base was purchased from Promega (Madison, WI 53711–5399, USA). All chemicals used were of analytical grade.
Shimadzu UV 1601 UV visible spectrophotometer (Kyoto, Japan) was used to measure the absorbance. LFT 600 EC freeze dryer was used to obtain the freeze dried powder of the poly herbal drug. Cells were incubated at 37°C in a humidified CO2 incubator (SHEL LAB/Sheldon manufacturing Inc. Cornelius, OR 97113, USA). Inverted fluorescence microscope (Olympus Optical Co. Ltd. 1X70-S1F2, Japan) for observation of cells, and photographs were taken using microscope digital camera (MDC200 2M PIXELS, 2.0 USB). Deionized water from UV ultra-filtered water system (Waterproplus LABCONCO Corporation, Kansas city, Missouri 64132–2696) and distilled water was used in all experiments.
Cell cultures
Human Rhabdomyosarcoma cell line (RD), and human breast adenocarcinoma cell line (MCF-7) were cultured in Dulbecco’s Modified Eagle Medium (DMEM), supplemented with 10% heat inactivated fetal bovine serum (FBS), penicillin (100 U/mL) and streptomycin (100 U/mL). The cells were maintained in 25 cm2 plastic tissue culture flasks at 37oC in a humidified atmosphere containing 5% CO2 in air. Exponentially growing cells were used in all experiments. The normal rat fibroblast (CC1) cell line was employed as the control.
Preparation of poly-herbal extract
The poly herbal drug (5 g) soaked in distilled water (100 mL) was kept in the rotary shaker for 48 hours in an air tight dark bottle. The extract was then filtered through a layer of muslin cloth and filtrate was centrifuged at 3,000 rpm for 15 minutes at 4oC to remove any debris.
The supernatant was freeze dried, and stored at -20°C in an air tight vial until used. The freeze dried extract was reconstituted with distilled water for experimental purposes.
Antioxidant activity
The DPPH assay was used to determine the radical scavenging activity of the extract, as reported previously by Perera (2008) [4]. Briefly different concentrations of plant extract (100 µL) was mixed with DPPH reagent prepared in absolute ethanol (100 µM, 900 µL) and incubated for 30 minutes in darkness at 37oC, in a water bath. The percentage of de-colorization was obtained spectro-photometrically at 517 nm. The control was prepared as above without adding extract while L-ascorbic acid was used as the positive control.
At least three independent tests were performed for each sample. Percentage inhibition was calculated from the following formula:
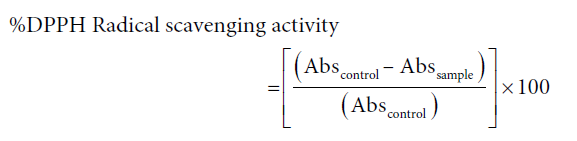
The effective concentration of the sample required to scavenge the respective radical by 50% (EC50) was calculated using the linear segment of the curve obtained with percentage inhibition against concentration.
Total phenolic content (TPC)
The total phenolic content of the lyophilized sample (n = 6) of the poly herbal drug was determined using the Folin-Ciocalteu method [5].
Brine shrimp bioassay
Brine shrimp assay was conducted to determine the cytotoxicity (LD50) of the poly herbal drug as explained previously by Soysa (2014) and Middleton (2005) [6, 7].
Briefly, dried cysts of brine shrimps (10 mg) were allowed to hatch at room temperature in sterile sea water. After 24 hours the free nauplii were selected and 10 were added to each petridish containing different concentrations of the extract in sea water. After 24 and 48 hours, the petridishes were observed using a magnifying glass and the number of survived nauplii in each petridish was counted.
The mortality end-point of this bioassay was defined as the absence of controlled forward motion during 30 seconds of observation [7].
The percentage lethality was determined by comparing the surviving larvae of the test and the control. The percentage survival was calculated as:
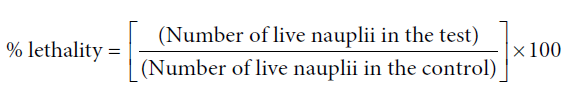
Cell viability assay
The effect of aqueous extract of the drug on the cell viability was determined by MTT assay. The live cells reduce yellow MTT to purple formazan crystals by mitochondrial dehydrogenase enzyme [8]. Cells were suspended in the growth medium and seeded in 24-well plates at 2 x 105 cells/well for overnight. The cells were then treated with different concentrations of the drug at 37oC for 24 and 48 hours. A positive control with cyclohexamide (50 g/mL) and a negative control without the drug were simultaneously conducted.
Percentage viability of the cells treated with the drug was calculated comparing the viability of the un-treated cells.
After 24/48 hours, the utilized growth media was subsequently replaced with 1.0 mL of minimum essential media (MEM), and 100 µL of MTT (5 mg/mL in PBS). The cells were incubated at 37o C for 4 hours and the medium was carefully removed. The formazan product was dissolved in acidified isopropanol (0.05 M HCl in isopropyl alcohol (IPA); 750 µL) and absorbance was read at 570 nm. Cell survival was expressed as a percentage of viable cells of treated samples to that of untreated cells (negative control). The drug extracts were prepared in triplicate and each experiment was performed in triplicates to each preparation.
Lactate dehydrogenase (LDH) activity
Cytotoxicity induced by the LPG evaluated by lactate dehydrogenase (LDH) leakage into the culture medium, as explained in Fotakis and Timbrell (2006) [9]. Cells seeded in 24 well plates (2 x 105 cells/well) were exposed to different concentrations of the drug. After 24/48 hours incubation the culture medium was aspirated and centrifuged at 4000 rpm for 5 min in order to obtain a cell free supernatant. The cell lysate was prepared by treating the cells with Triton X 100 (0.1%; 1 mL) and sonicating the contents for 20 seconds. The final suspension was centrifuged at 4000 rpm for 5 minutes. Medium and the lysate were subjected to LDH assay. The activity of LDH in the medium was determined using a commercially available, LDH assay kit (HUMAN).
Negative control and positive control with cyclohexamide (50g/mL) were also carried out along with the experiment to measure the percentage LDH leakage.
The absorbance was measured at 340 nm at intervals of 15 seconds for 1.5 minutes using an air blank. The rate of decline (gradients of the graphs) of NADH concentration was used to calculate the LDH activity in the supernatant and the lysate.
The percentage LDH leakage to the medium was calculated using the following equation.
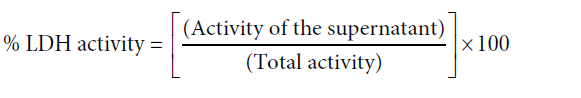
Where total LDH activity = LDH activity of supernatant + LDH activity of the lysate.
Light microscopy
RD, MCF-7 and CC1 cells grown at 70% confluence were treated with different concentrations of aqueous drug extracts for 24, 48 and 72 hours and observed under phase-contrast inverted fluorescence microscope (40X). The changes in morphology were compared with positive and negative controls.
Statistical analysis
All the results of the experiments were expressed as the mean ± standard deviation (Mean ± SD). The measurements were performed in triplicate and values shown are representatives of at least three independent experiments. Least square linear regression analysis was applied using Microsoft excel to determine the EC50/LD50 values and for the calibration curves.
R2 > 0.99 was considered as linear for the calibration curves.
Significant differences of each test result were statistically analyzed using “Mann-Whitney U” test significances with 95% significance using SPSS version 16.
Results
Anti-oxidant capacity and total Phenolic content
The aqueous extracts of the poly herbal drug showed moderate free radical scavenging activity with a value of EC50 = 68.0 ± 2.8 g/mL with compared to the positive control, L- ascorbic acid (3.4±0.9g/mL; n=6).
The percentage total phenolic content of the aqueous extract of the poly herbal drug was 5.3 ± 0.1 W/W % of Gallic Acid Equivalence (GAE).
Brine shrimp bioassay
LPG shows no lethality towards the brine shrimp even after 48 hours exposure up to a concentration of 10 to 2000 μg/mL. This results show that the aqueous extract either does not possess any significant (p>0.05) alterations in the cellular functions or no significant cytotoxity.
Cell viability assay
The effect of LPG on cell viability of RD, MCF-7 and CC1 cells was evaluated by tetrazolium assay (MTT).
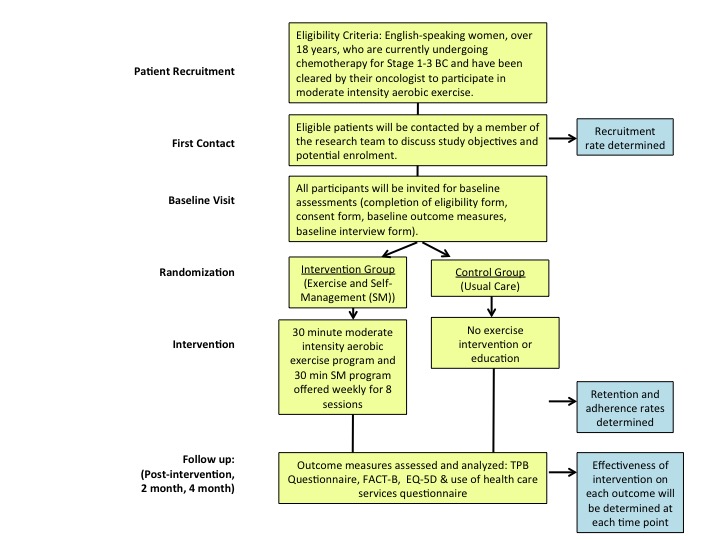
Multiple concentrations of the aqueous extract of the poly herbal drug were used and effective concentrations were calculated for each cell line (EC50) from dose response curve. The percentage cell viability of RD, MCF-7 and CC1 are shown in Figure 1.
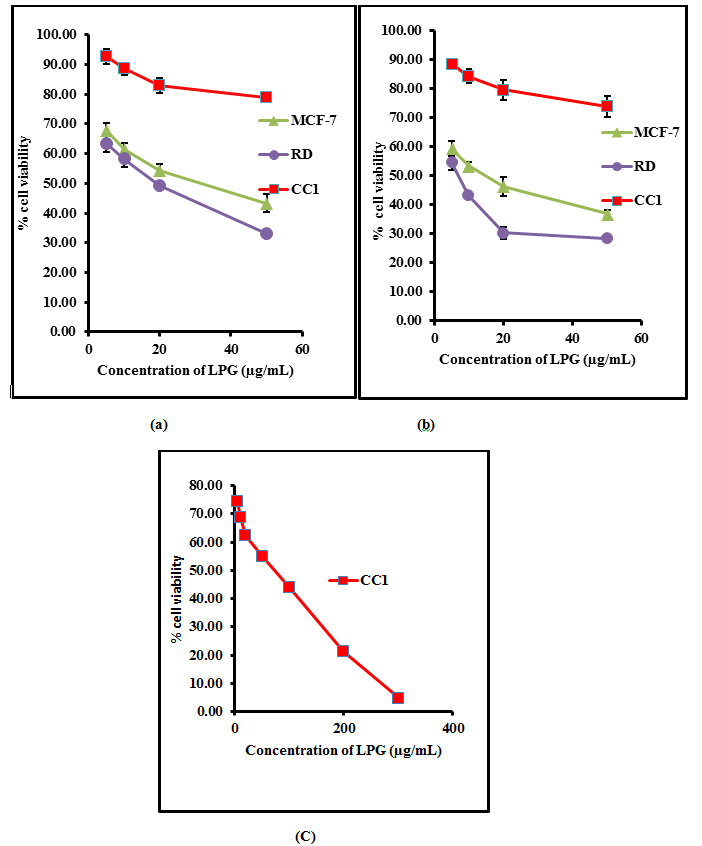
Figure 1. The percentage cell viability of on RD, CC1 and MCF-7 cell lines as determined by MTT assay, (a) after 24 hours treatment (b) 48 hours and (c) 72 hours with aqueous extract of the poly herbal drug. The data are presented as mean ± SD of six independent experiments.
The aqueous extract of the LPG exhibited no significant cytotoxic activity (p>0.05) against CC1 cell line, achieving an EC50 of 85.3 ± 9.8 µg/mL after 72 hour incubation.
On the contrary, the aqueous extract of the poly herbal drug exhibited significant cytotoxic activity (p<0.05) against RD and MCF-7 cell lines compared to the CC1 cells as shown in Table I.
Table I. EC50 values obtained for MTT assay for the three different cell lines.
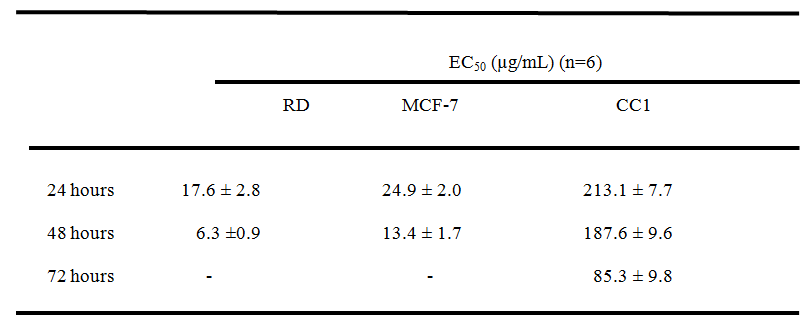
*All results were mean of 6 independent measurements ± standard deviation. “Mann-Whitney U” test at 95% confidence level showed a significant difference (p < 0.05) in both RD and MCF-7 cells compared to CC1 cells.
After 24 and 48 hour incubation with Cyclohexamide (Positive control) at a concentration of 50µg/mL, the percentage viability showed by tested cells depicted in Table 2. The percentage cell viability obtained for MTT assay at a concentration of 50µg/mL of LPG was calculated using respective regression equations to compare the capacity to inhibit cell proliferation, with that of the positive control. The values are shown in Table 2.These results exhibited the significant cytotoxicity (p< 0.05) exert by the aqueous extract of LPG against cancer cells compared to CC1 cells as determined by MTT assay.
Table 2. Percentage cell viability obtained by MTT assay at 50 µg/mL Cyclohexamide (+ve Control) and 50 µg/mL LPG after 24 hours and 48 hours exposure
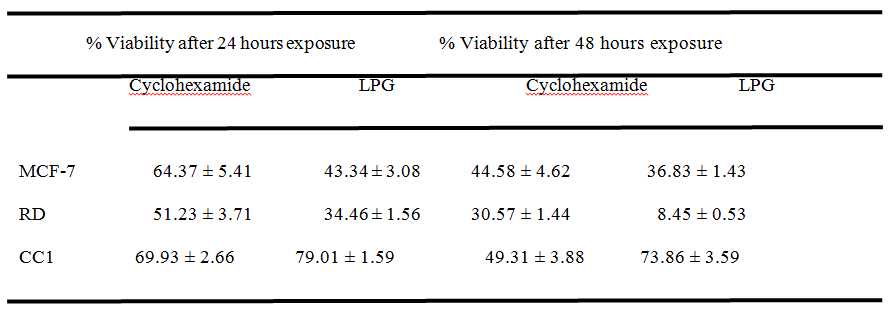
*All results were mean n=6 measurements ± standard deviation in three independent experiments.
Lactate dehydrogenase (LDH) assay
Measurement of LDH activity is also an indicator of cell viability through evaluation of the cell membrane permeability. The enzyme activity is measured externally, as it leaks from dead cells which lose their membrane integrity. The LDH release curves for RD, MCF-7 and CC1 cell lines treated with different concentrations of the LPG suggested that the cytotoxic effect of the extract was concentration dependent (Figure 2).
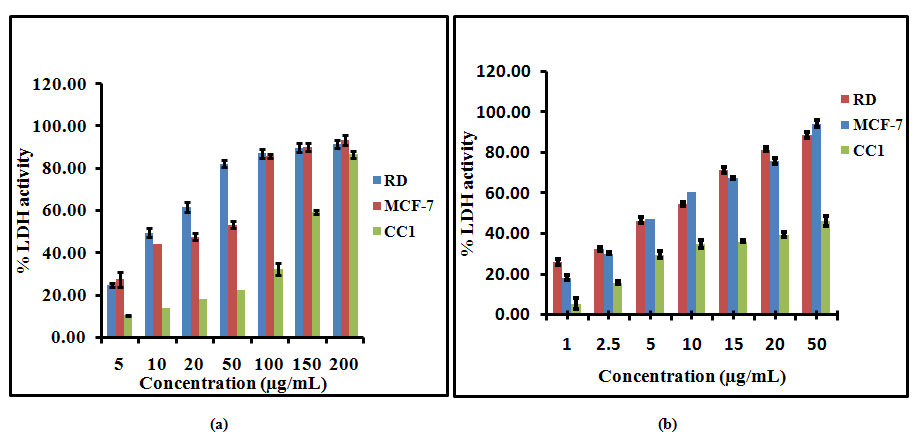
Figure 2. Lactate Dehydrogenase (LDH) release from RD, MCF-7 and CC1 cells with aqueous extract of the poly herbal drug, (a) after 24 hour, (b) after 48 hour incubation. The data are presented as mean ± SD of six independent experiments.
Fifty percent leakage of LDH to the media was observed at 26.9± 1.6 and 30.5 ±2.8μg/mL for RD and MCF-7 cells after 24 hour incubation respectively. After 48 hours of incubation with LPG the EC50 values further decreased and 50% LDH leakage arise at 8.2± 0.2 and 6.0± 0.2 μg/mL for RD and MCF-7 cells respectively. In contrast to RD and MCF-7 cells the CC1 cells showed 50% leakage of LDH at 159.3± 3.1 and 103.1 ±5.2μg/mL LPG concentrations after 24 and 48 hours respectively.
Light microscopy
Morphological alterations of RD, MCF-7 and CC1 cell lines upon exposure to the aqueous extract of the poly herbal drug was observed under the phase contrast inverted microscope. The microscopic observations revealed that the aqueous extract has a significant cytotoxic effect on RD and MCF-7 cells compared to the negative control. However CC1 cells treated with the drug did not show significant cell death (Figure 3).
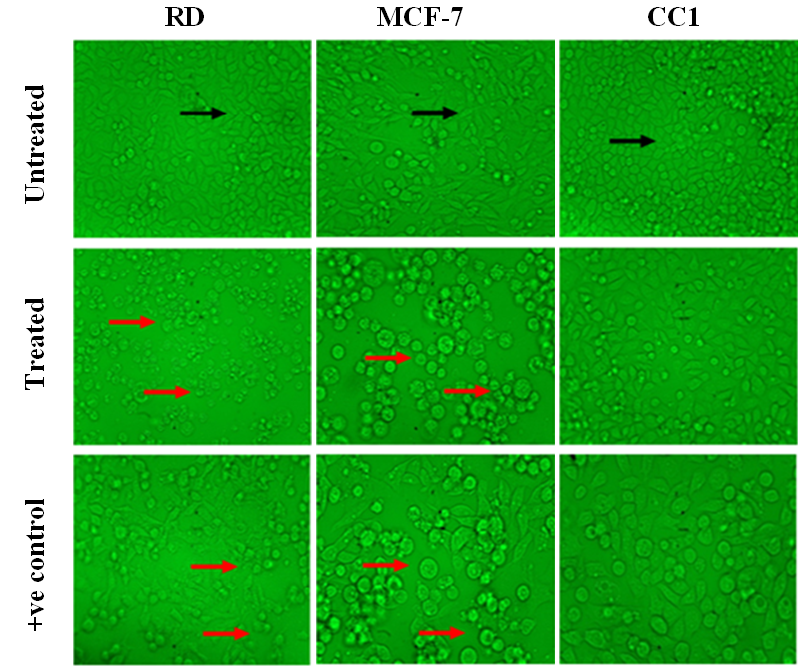
Figure 3. Light microscopy images of RD, MCF-7 and CC1 cells treated with their respective EC50 values of LPG, with magnification of 40X. (Black arrow – indicates healthy spindle shape cells; Red arrow – dead and shrinkage cells due to the LPG treatment).
Discussion
Cancer has become the most leading cause of death worldwide. It has become an emerging health problem affecting both developing and developed countries. According to the World Health Organization reports published in 2014, 8.2 million patients have died from cancer in 2012 and the number of annual cancer cases reported in 2012 was 14 million. It has been also estimated that the number of annual cancer cases would have increased from 14 million in 2012 to 22 million within the next two decades [10]. The most effective treatment approaches in cancer are chemotherapy and radiotherapy. Nevertheless, the higher incidence in side effects, have made investigators engage in finding novel anticancer compounds with less adverse effects. As a result of this, plants and other natural sources have provided nearly 60% of anti cancer agents which are currently in use [11]. Especially the most novel chemotherapeutic agents more than ever before are botanical in kind [2].
Traditional folklore medicine which involves the use of natural elements uses either single or multiple herbs as a mixture (polyherbs) for treatment of diseases. The use of polyherbal formulation is found to be more effective than the use of a single herb as the active phytochemical constituents of the individual plants are insufficient to achieve the desirable therapeutic effect. But when combining the herbs in a particular ratio the synergistic effect produced by the active phytochemicals of several plants give a better therapeutic effect [12].
Polyherbal preparations are used worldwide for treatment of various disease conditions including cancer. As the polyherbal formulations have gained more attention, many studies have been conducted to identify the mechanism of action and the efficacy of these polyherbal preparations. A latest study [13] which gives a global perspective on polyherbal formulations reports that for the past six years, only 2 publications have been made regarding polyherbal formulations that show/have anticancer activity.
Zyflamend® used in China [14], Sho-saiko-to® used in Japan [15] and Arbudhcure® used in India [16] are few of the polyherbal preparations used for cancer treatment where attempts have been made to validate scientifically for their mechanism of action and therapeutic efficacy.
With compared to the aforementioned poly herbal drugs the polyherbal LPG is not commercialized and no scientific research has been carried out up to this study to identify its mechanism of action or evaluate its effectiveness on treatment of cancer even though it is being used to treat 32 different types of cancer in traditional medicinal system in Sri Lanka.
Secondary metabolites of plants including phenolic compounds are very important for their essential functions in reproduction, growth and in defense mechanisms [17]. Phenolic compounds also provide natural antioxidants for protection against many diseases including cancers. It has been reported that antioxidant effects of plants are mainly attributed to the radical scavenging activity of phenolic compounds such as flavonoids, polyphenols, tannins, and phenolic terpenes. The polyphenols have the capability to scavenge ROS as well as oxidatively generated free radicals which derive from bio-molecules [18].
As depicted in results the quantitative measurement of poly phenolic content obtained in the present study showed that the water extract of LPG has moderate levels of polyphenols. Similarly the EC50 values obtained for the different samples of LPG for DPPH assay showed higher values compared to EC50 of ascorbic acid which evidently indicate that the LPG has a lower anti-oxidant capacity.
It was reported by several authors, that the proliferation of cancer cells are inhibited by the high antioxidant and poly phenolic content present in plant extracts [14, 19]. This probable fact that presence of low polyphenolic content and low antioxidant potential was explained in Hamberger and Hastettman 1991[20], as possible changes occurring in the chemical composition of the plant materials during the processes of the drug manufacturing that could result in a reduced pharmacological activity in mixtures. Thus it can be suggested that even though the LPG exerts an effective antiproliferative activity and cytotoxicity, the mechanism of action must be in a different pathway compared to most of the other traditional medicinal drugs which exert antiproliferative activity via the phenolics and antioxidants.
Cytotoxic action in brine shrimp assay is brought about by the active ingredients present in the drug by disturbing the fundamental mechanisms associated with cell growth, mitotic activity, differentiation and function [21]. According to previous reports a crude plant extract is toxic (active) if it has an LD50< 1000 µg/mL while non-toxic (inactive) if LD50> 1000 µg/mL [22]. The present study evidently indicates that the aqueous extract of LPG shows no lethality towards the brine shrimp even after 48 hours of exposure within the concentrations tested ranging 10 to 2000 µg/mL. This results show that the aqueous extract either does not possess any significant (p>0.05) alterations in the cellular functions or no significant cytotoxicity. Similar results were observed in a previous study which shows anticancer activity of the plant extract of Flueggea leucopyrus against HEp 2 cells but no toxicity for brine shrimps [6].
In this study the cell viability of the tested cell lines were determined by the MTT reduction assay. Each cell type was treated with different concentration of LPG extract and incubated for 24 and 48 hours. The EC50 value was obtained from the graph between the percentages of cell viability versus concentrations of LPG extract (Figure 1). The values were used to describe the degree of cytotoxicity of the extract towards cell lines. The cell viability of CC1 cells were further determined after 72 hours due to the high viability of the cells at 48 hours of incubation (187.58 ± 9.59 µg/mL).
It was established by the American National Cancer Institute (NCI), that if a crude extract shows EC50< 30 µg/mL after an incubation of 72 hours it is considered as cytotoxic [23]. However crude extract with EC50 less than 20µg/mL after 48 hours incubation is supposed to be highly cytotoxic [24]. Present study shows a potent cytotoxic effect on RD and MCF-7 cells with the aqueous extract of the LPG even at 24 hours which is much lower than the value specified by the NCI. However, the extract was less cytotoxic towards normal mouse fibroblast cell line CC1.
After 24 and 48 hours of incubation with Cyclohexamide (Positive control) at a concentration of 50μg/mL, the percentage viabilities of the tested cells are showed in Table 2. In contrast, the percentage viability obtained for MTT assay at the same concentration of LPG (50 μg/mL) treatment was markedly lower for RD and MCF-7 cells (Table 2).The EC50values obtained for MTT assay indicate that the cytotoxicity of the LPG extract against the cancer cell lines is within the limit of cytotoxicity (EC50 < 30 μg/mL), as reported by the American National Cancer Institute (NCI) over 72 hour post exposure and it is beyond the limits for CC1 cells (Table 1) Thus LPG shows significant anti-proliferative activity on cancer cells studied compared to the standard anti cancer drug. Cyclohexamide showed a % cell viability of 69.93 ± 9.26% and 49.31 ± 3.88% for 24 and 48 hours respectively for CC1 cells, suggesting its high toxicity towards the normal cells compared to the LPG (Table 2).
It is reported that the poly herbal anticancer drug Zyflamend® shows an EC50 value of 200 µg/mL against prostate cancer cell line (PCC) after an incubation period of 96 hours [14]. Japanese poly herbal drug Sho-saiko-to® shows an EC50 value of 353.5 µg/mL against human hepatocellular carcinoma (HCC-KIM) at 72 hour incubation [15], while Arbudhcure® shows an EC50 of 33.0 µg/mL against HeLa cells for 48 hours post exposure to the respective drug [16]. The current study demonstrates that LPG shows higher anti-proliferative activity on four different cancer cell lines investigated in the present study in comparison with aforementioned polyherbal drugs.
Measurement of LDH activity is another indicator of cell viability through evaluation of the cell membrane permeability. Previous studies suggest that LDH is a more reliable and accurate marker of cytotoxicity, since damaged cells is fragmented completely during the course of prolonged incubation with substances [25]. The intracellular LDH release to the medium is a measure of irreversible cell death. Xia (2007) [26] reported that there is a correlation between the direct involvement of LDH up regulation and subsequent induction of apoptosis, which is ideal for an effective anti cancer drug.
Fifty percent leakage of LDH to the media was observed at 26.9± 1.6 and 30.5 ±2.8μg/mL for RD and MCF-7 cells after 24 hour incubation respectively. After 48 hours of incubation with LPG the EC50 values further decreased and 50% LDH leakage arise at 8.2± 0.2 and 6.0± 0.2 μg/mL for RD and MCF-7 cells respectively. In contrast to RD and MCF-7 cells the CC1 cells showed 50% leakage of LDH at 159.3± 3.1 and 103.1 ±5.2μg/mL LPG concentrations after 24 and 48 hours respectively. These results indicate that the LPG is having minimal cytotoxic effect on control cells while it possesses a remarkable cytotoxicity on two cancer cell types studied.
Morphological alterations of RD, MCF-7 and CC1 cell lines upon exposure to the aqueous extract of the LPG revealed that even at low concentrations the morphology of the RD and MCF-7 cells changed into an irregular shape with extremely condensed nuclear contents, signs of membrane blebbing, suggesting an autophagic mechanism of cell death. The untreated cells (negative control)of RD and MCF-7 cell lines show cells with normal morphology (Spindle shape/ elongated cells) that adhered to the culture plate with no signs of cells death and growth disorders.
The positive control also shows the typical morphological changes which indicate the cellular damage in RD and MCF-7 cells. In contrast to the RD and MCF-7 cells the CC1 cells do not show any significant morphological changes even after 72 hour incubation with the extract.
Conclusions
The present study provides strong evidence for potent anti-cancer activity of the poly herbal drug “Le Pana Guliya” on RD and MCF-7 cells compared to controls. The brine shrimp and CC1 cells results suggest that the extract has minimum cytotoxicity towards the normal healthy cells. This calls for further studies on the active components and the mechanism of action involved in cytotoxicity of the chemotherapeutic action of LPG.
Acknowledement: The financial support by World Class University Research Grant No. AP/3/2012/CG/10is highly acknowledged.
Conflict of Interest: No conflict of interest between authors.
References
- Vuorelaa P, Leinonenb M, Saikkuc P, Tammelaa P, Rauhad JP, Wennberge T and Vuorela H (2004) Natural products in the process of finding new drug candidates. Current Medicinal Chemistry11:1375-1389.
- Biswas J, Roy M and Mukherjee A (2015) Anticancer Drug Development Based on Phytochemicals. Journal of Drug Discovery, Development and Delivery 2: 1012.
- Hosseini A and Ghorbani A (2015) Cancer therapy with phytochemicals: evidence from clinical studies. Avicenna Journal of Phytomedicine 5:84-97.
- Perera MGAN, Soysa SSSB, Abeytunga DTU and Ramesh R (2008) Antioxidant cytotoxic properties of three traditional decoctions used for the treatment of cancer in Sri Lanka. Pharmacognosy Magazine 4: 172-181.
- Velioglu, YS, Mazza G, Gao L and Oomah BD (1998) Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. Journal of Agricultural Food & Chemistry 46: 4113–4117.
- Soysa P, De Silva IS and Wijayabandara J (2014) Evaluation of antioxidant and antiproliferative activity of FlueggealeucopyrusWilld (katupila). BMC Complementary and Alternative Medicine 14:274.
- Middleton P, Stewart F, Al-Qahtani S, Egan P, O’Rourke C and Sarker SD (2005) Antioxidant, antibacterial activities and general toxicity of Alnusglutinosa, Fraxinus excelsior and Papaverrhoeas. Iranian Journal of Pharmaceutical Research 2:81-86.
- Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. Journal of Immunological Methods 65:55-63.
- Fotakis G and Timbrel J (2006) A In vitro cytotoxicity assays: Comparison of LDH, neutral red, MTT and protein assay in hepatoma cell lines following exposure to cadmium chloride. Toxicology Letters 160: 171–177.
- World Cancer report 2014. International Agency for Research on Cancer (IARC) 2014, World health organization.
- Cragg GM and Newman DJ (2000) Antineoplastic agents from natural sources: achievements and future directions. Expert Opinion on Investigational Drugs 9:2783-2797.
- Parasuraman S, Thing GS and Dhanaraj SA (2014) Polyherbal formulation: Concept of ayurveda. Pharmacognosy Reviews 8:73-80.
- Aslam MS, Ahmad MA, Mamat AS, Ahmad MZ and Salam F (2016) An Update Review on Polyherbal Formulation: A Global Perspective. Systematic Reviews in Pharmacy 7:35-41.
- Zhao Y, Donohoe D, Huang EC and Whelan J (2015) Zyflamend, a polyherbal mixture, inhibits lipogenesis and mTORC1 signalling via activation of AMPK. Journal of Functional Foods 18:147-158.
- Yano H, Mizoguchi A, Fukuda K, Haramaki M, Ogasawara S, Momosaki S and Kojiro M (1994) The herbal medicine sho-saiko-to inhibits proliferation of cancer cell lines by inducing apoptosis and arrest at the G0/G1 phase. Cancer Research 54: 448-454.
- Pankajaakshan PK and Hashim KM (2014) Anti-cancer activity of Arbudhcure prepared from Aswathy medical hall. European Journal of Experimental Biology 4:22-25.
- Prior RL, Wu, X and Schaich, K (2005) Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. Journal of Agriculture and Food chemistry 53: 4290-4302.
- Valko M, Leibfritz D, Moncola J, Cronin MTD, Mazura M and Telser J (2007) Free radicals and antioxidants in normal physiological functions and human disease. Review. International Journal of Biochemistry and Cell Biology 39: 44-84.
- Suryavanshi S, Choudhari A, Deshpande R, Kulkarni O and Kaul-Ghanekar R (2011) Analyzing the antioxidant potential of aqueous and ethanolic preparations of an herbal composition (HC9) and evaluating their cytotoxic activity in breast cancer cell lines. Biotechnology, Bioinformatics and Bioengineering 1:513-522.
- Hamberger M, Hastettman K (1991) Bioactivity in plants: the link between phytochemistry and medicine. Phytochemistry 30:3864-3874.
- Meyer BN, Ferrigni NR, Putnam JE, Jacobsen LB, Nichols DE and McLaughlin JL (1982) Brine shrimp: A convenient general bioassay for active plant constituents. Journal of Planta Medica 45: 31-34.
- Olowa LF and Nuñeza OM (2013) Brine Shrimp Lethality Assay of the Ethanolic Extracts of Three Selected Species of Medicinal Plants from Iligan City, Philippines. International Research Journal of Biological Sciences 2: 74-77.
- Abdel-Hameed ES, Salih A, Bazaid SA, Shohayeb MM, El- Sayed MM and El-Wakil EA (2012) Phytochemical studies and evaluation of antioxidant, anticancer and antimicrobial properties of Conocarpus erectus L. growing in Taif, Saudi Arabia. European Journal of Medicinal Plants 2: 93-112.
- Mahavorasirikul W, Viyanant V, Chaijaroenkul W, Itharat A, Na-Bangchang K (2010) Cytotoxic activity of Thai medicinal plants against human cholangiocarcinoma, laryngeal and hepatocarcinoma cells in vitro. BMC Complement Altern Med 10: 55. [crossref]
- Sarveswaran S, Marati RV and Marathaiveeran PB (2006) Effects of Terminalia arjuna bark extract on apoptosis of human hepatoma cell line (HEPG2). Journal of Gastroenterology 12: 1015-1024.
- Xia GH, Chen BA, Shao ZY, Lu HX, Konstanze D and Hartmut D (2007) Mechanism of 2-methoxyestradiol-induced apoptosis in myelodysplastic syndrome MUTZ-1 cell line. Zhongguo Shi Yan Xue Ye Xue Za Zhi 15:296-301.