DOI: 10.31038/JNNC.2025813
Abstract
Background: Neurological conditions are among the leading causes of long-term disability, with gait and balance impairments affecting up to 60% of patients and significantly limiting their mobility. Conventional gait therapies often lack the intensity required to promote neuroplasticity, are physically demanding for the therapist, and often require the presence of more than one qualified professional. Robot-assisted gait training (RAGT) presents a viable alternative, addressing these limitations by enabling high-volume, consistent rehabilitation.
Objective: This study aims to evaluate the efficacy of a novel end-effector RAGT system in adult patients with gait impairments resulting from neurological disorders of various etiologies. The primary objective is to quantify gait improvements using key spatiotemporal parameters and to explore potential differences in rehabilitation outcomes across diagnostic subgroups.
Material and methods: Twenty-eight adult neurological patients with gait impairments were recruited from two rehabilitation centers and classified into three groups based on the etiology of their condition. Each patient underwent 12 sessions of end-effector-based RAGT. Gait improvements were evaluated by comparing step count, walked distance, and cadence between the first and final therapy sessions.
Results: Statistically significant improvements were observed across all gait parameters and diagnostic groups. The greatest relative gains were seen in patients with cerebrovascular disorders, while the smallest improvements were noted in those with spinal cord-related neuropathies. On average, step count increased by approximately 45%, walked distance by 59%, and cadence by 48%.
Conclusion: The end-effector-based RAGT system demonstrated a significant positive effect on gait improvement in adults with various neurological conditions, reflecting the heterogeneity typically encountered in routine physiotherapy practice. These findings suggest that this technology may serve as a viable alternative to conventional gait training offering the advantages of high-intensity repetitive therapy that can be delivered in a safe environment even in the early stages of neurological recovery.
Keywords
RAGT, end-effector, Gait training, Neurological disorders, Stroke, Cerebral palsy, Spinal cord injury
Introduction
Neurological disorders are a major cause of long-term disability, with up to 60% of patients experiencing mobility impairments due to gait and balance dysfunction. Loss of independent walking is one of the most serious consequences, driven by muscle weakness, limited joint mobility, and poor coordination, leading to asymmetry and instability [1-3].
The most common neurological etiologies associated with gait impairment include stroke, cerebral palsy (CP), spinal cord injury (SCI), multiple sclerosis (MS), and Parkinson’s disease [2,4]. Stroke remains the most prevalent, affecting over 100 million people globally, with more than 80% of survivors experiencing gait limitations [5,4]. CP, although congenital, presents persistent motor symptoms into adulthood, with 25–58% of adults reporting a progressive decline in mobility [5]. SCI affects approximately 15 million people worldwide [4]. The severity of gait impairment and the potential for recovery depend on the type and extent of the lesion. More than half of SCI cases are incomplete, where some level of walking recovery is possible, unlike in complete injuries, where the chance of regaining ambulation is minimal [6]. In MS and Parkinson’s disease, gait impairments typically develop gradually due to the progressive nature of these disorders [2].
Beyond motor dysfunction, gait impairment significantly affects patients’ psychosocial and physical health. It is associated with increased rates of depression, anxiety, reduced social participation, lower health-related quality of life and higher unemployment rates [7]. In conditions such as stroke and SCI, limited mobility also reduces physical activity, negatively affecting modifiable cardiovascular risk factors. Up to 60% of stroke survivors present with comorbidities like hypertension, coronary artery disease, diabetes, or obesity. Individuals with SCI are nearly three times more likely to develop cardiovascular disease compared to age-matched healthy controls [8]. Finally, gait disturbances are a major risk factor for falls across all these conditions, often leading to additional complications, hospitalizations, and further decline in independence [9].
Conventional gait therapies, such as overground training and body-weight-supported treadmill training (BWSTT), are widely used in neurorehabilitation; however, their application is limited by several factors [10-12]. These include an insufficient number of step repetitions to promote neuroplasticity, high physical demands on therapists, frequent involvement of multiple physiotherapists simultaneously, and an increased risk of patient falls, which can reduce both safety and therapeutic intensity [13-15]. For example, in the study by Duncan et al., which examined the effects of BWSTT in stroke patients, falls were the most commonly reported adverse event, occurring in over 40% of participants [13].
Robotic-assisted gait training (RAGT) has emerged as a promising intervention that overcomes key limitations of conventional gait therapies. By enabling high-intensity, repetitive, and task-specific training, RAGT promotes motor recovery through principles of neuroplasticity [16]. Two main types of RAGT systems exist: end-effector and exoskeleton-based devices. While exoskeletons provide joint-specific guidance and support, end-effector systems guide the movement of the distal segments (typically the feet), promoting a more physiological gait pattern and often allowing for greater step repetition, which may enhance training intensity and motor recovery [7,15]. Its efficacy has been demonstrated across various neurological conditions, including spinal cord injury, stroke, multiple sclerosis, and Parkinson’s disease [9,11,17-19], with some studies showing superior effectiveness compared to conventional therapies [9,17,18]. The majority of studies investigating RAGT systems primarily focus on specific neurological diagnoses. Research involving heterogeneous patient groups is notably limited. Within the context of end-effector RAGT, only a singular study addresses feasibility, with effectiveness being assessed as a secondary outcome [7].
This study aims to evaluate the efficacy of a novel end-effector RAGT system in adult patients with gait impairments resulting from neurological disorders of various etiologies. The primary objective is to quantify improvements in key spatiotemporal parameters including gait cadence, walking distance and step count, and to explore potential differences in rehabilitation outcomes across diagnostic subgroups.
Material and Methods
This multicenter prospective interventional study was conducted between February 2024 and April 2025 at two rehabilitation centers, one located in Austria and the other in the Czech Republic. A total of 30 neurological patients were enrolled and received end-effector RAGT therapy as part of their rehabilitation program.
Eligible participants were adults (aged 18 or older) with gait impairments resulting from neurological conditions, including both diseases and injuries, who were medically stable and capable of participating in active rehabilitation. All participants had sufficient cognitive and physical capacity to engage in robotic gait training and were selected for therapy by a healthcare professional.
Exclusion criteria included conditions contraindicating RAGT, such as major joint contractures, unhealed fractures, osteoporosis or osteopenia, severe cardiovascular disease, epilepsy, open wounds in the device contact area, acute infections, cognitive or psychological impairments preventing safe or effective participation in therapy, or any acute medical condition that could compromise therapy safety.
Before participation, all patients received detailed information about the therapy and its possible outcomes, and voluntarily signed an informed consent form. Anonymity was maintained by assigning each patient a unique identification number. The study adhered to the ethical principles of the 1975 Declaration of Helsinki and relevant guidelines (Council of Europe, 1997; WMA, 1997–2000).
Each participant completed 12 gait training sessions using the end-effector-based RAGT system (R-Gait, BTL Industries Ltd.). Sessions were administered at a minimum frequency of three times per week, with each session lasting 30 minutes. At the start of each session, a trained therapist carefully positioned the patient and securely fitted them into the harness and footplates, allowing unrestricted movement of the lower limbs and pelvis. Therapy parameters such as a gait cadence, step length, and weight support were personalized to the patient’s abilities and adjusted as needed during the session. A dynamic support system continuously adjusted body weight unloading in synchronization with the gait cycle to promote a natural walking pattern. The footplates simulated key walking phases, while integrated sensors monitored foot activity and weight support levels. The device automatically recorded therapy progress, including key walking parameters.
Gait parameters, specifically the number of steps, distance walked (in meters), and walking cadence (steps per minute), were evaluated. These parameters were recorded by a device during therapy sessions and automatically saved. Data from the first and last therapy sessions were compared. Subsequent data analysis was performed using a custom script in MATLAB (MatLab R2010b, MathWorks, Inc., Natick, MA, USA). The normality of the data was assessed using the Shapiro-Wilk test. As the data did not meet the assumption of normality, a Wilcoxon signed-rank test was performed to determine the statistical significance of differences between the initial and final therapy sessions.
Results
A total of 30 patients participated in the study, with a mean age of 46 ± 18.64 years. Two patients did not complete the full course of treatment due to health complications unrelated to the therapy being investigated. Overall, the therapy was well tolerated, and no serious adverse effects were observed, aside from common muscle fatigue. Patients were categorized into three groups based on the etiology of their neurological condition: cerebrovascular disorders, neurodevelopmental disorders, and spinal cord-related neuropathies. Detailed information about the patients is presented in Table 1.
Table 1: Participant demographics and the distribution of study indications.
|
Indication group
|
Total number |
Gender male/female |
Age, mean (SD)
|
| Cerebrovascular disorders |
8
|
2/6 |
60.25 (17.9)
|
| Neurodevelopmental disorder |
10
|
4/6 |
32.2 (12.35)
|
| Spinal cord-related neuropathies |
10
|
6/4 |
49.1 (15.45)
|
| Total |
28
|
12/16 |
46.25 (18.64)
|
Across all evaluated parameters (steps walked, walking distance, and walking cadence) statistically significant improvements were observed in each diagnostic group (see Table 2). On average, patients demonstrated an increase of approximately 45% in the number of steps, 59% in walking distance, and 48% in walking cadence (all p< 0.001). The most pronounced relative improvements were recorded in the cerebrovascular disorder group, whereas the spinal cord–related neuropathy group exhibited the smallest, though still statistically significant, gains. These within-group distributions and changes are detailed in the box plots shown in Figures 1-3. Notably, walked distance showed the greatest average improvement across all groups. The percentage-improvement bar chart (Figure 4) further illustrates these relative changes, facilitating direct comparisons between diagnostic categories.
Table 2: Mean (±SD) values for walked steps, distance, and cadence measured during the first and last therapy sessions across diagnostic groups. Percentage differences represent relative change.
|
Parameter
|
Indication |
Before |
After |
Difference (%) |
P-value
|
| Walked steps |
Cerebrovascular disorders |
1040.00 ± 301.85
|
1606.50 ± 454.63 |
54.47 |
0.014
|
| Neurodevelopmental disorder |
935.00 ± 328.39
|
1368.00 ± 379.68 |
46.31 |
0.004
|
| Spinal cord-related neuropathies |
1071.00 ± 425.36
|
1450.00 ± 468.10 |
35.39 |
0.009
|
| Total |
1015.33 ± 351.86
|
1474.83 ± 434.14 |
45.26 |
< 0.001
|
| Walked distance (m) |
Cerebrovascular disorders |
390.65 ± 158.66
|
664.50 ± 173.03 |
70.10 |
0.014
|
| Neurodevelopmental disorder |
349.50 ± 171.34
|
548.20 ± 260.46 |
56.85 |
0.004
|
| Spinal cord-related neuropathies |
561.00 ± 142.86
|
850.00 ± 170.90 |
51.52 |
0.009
|
| Total |
433.72 ± 157.62
|
687.66 ± 201.46 |
58.53 |
< 0.001
|
| Cadence (steps/min) |
Cerebrovascular disorders |
32.89 ± 7.51
|
52.27 ± 4.65 |
58.95 |
0.014
|
| Neurodevelopmental disorder |
32.13 ± 10.10
|
48.53 ± 11.98 |
51.04 |
0.004
|
| Spinal cord-related neuropathies |
38.13 ± 11.78
|
51.43 ± 10.37 |
34.88 |
0.009
|
| Total |
34.38 ± 9.79
|
50.74 ± 9.00 |
47.59 |
< 0.001
|
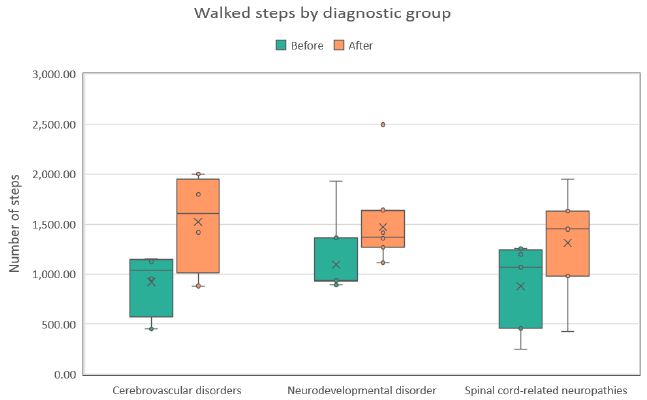
Figure 1: Distribution of steps walked during the first (Before) and last (After) therapy sessions across three diagnostic groups. Each boxplot shows the median, interquartile range, and any outliers.
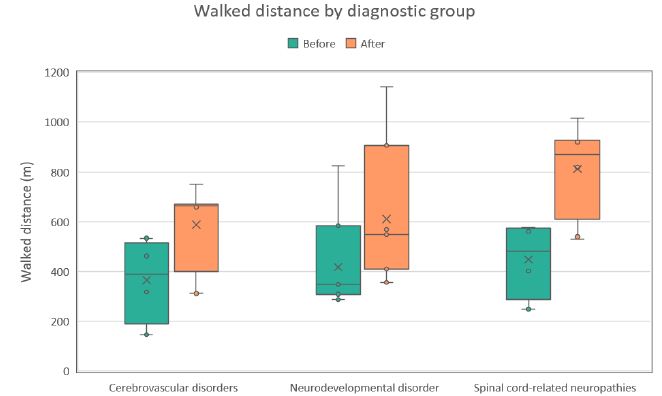
Figure 2: Distribution of walked distance values (m) during the first (Before) and last (After) therapy sessions across three diagnostic groups. Each boxplot shows the median, interquartile range, and potential outliers.
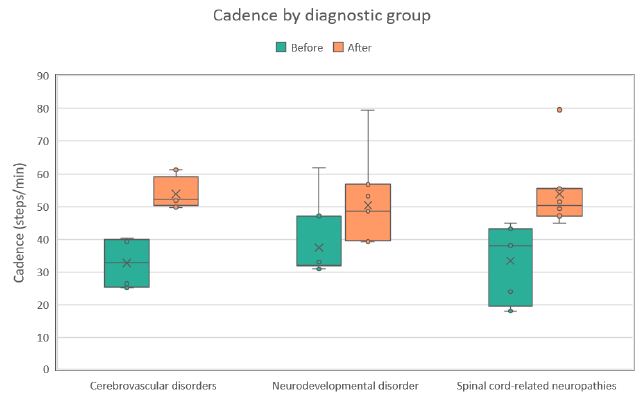
Figure 3: Distribution of walking cadence values (steps/min) during the first (Before) and last (After) therapy sessions across three diagnostic groups. Each boxplot shows the median, interquartile range, and any outliers.
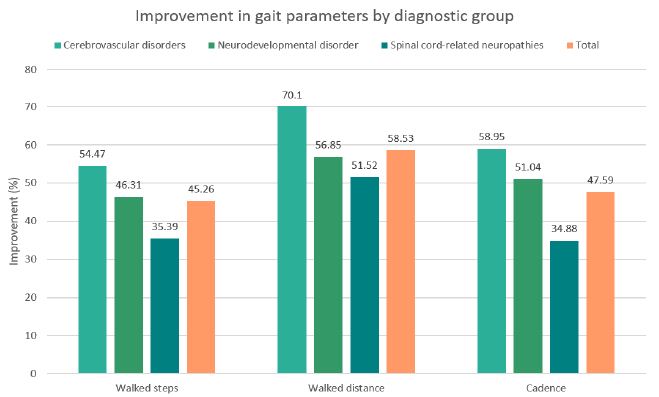
Figure 4: Percentage improvement in walked steps, walked distance, and walking cadence across diagnostic groups following the intervention, including the overall mean across all groups.
Discussion
This study aimed to assess the benefits of end-effector-based RAGT in adults with diverse neurological diagnoses by monitoring gait parameters, including step count, walking cadence, and distance covered. Furthermore, the study explores potential patterns and differences in rehabilitation potential across distinct neurological etiologies. To the best of the author’s knowledge, this is the first study primarily focusing on the impact of an end-effector-based RAGT system on gait parameters in such a heterogeneous patient population, as most previous research has been limited to single-diagnosis cohorts.
Following 12 RAGT sessions, a statistically significant improvement in gait parameters was observed across all diagnostic groups. The greatest relative gains were noted in patients with cerebrovascular disorders, followed by those with neurodevelopmental conditions. Although patients with spinal cord-related neuropathies demonstrated the lowest magnitude of improvement, their outcomes remained statistically significant. For comparison, a previous study by Hotz et al. also examined the effects of end-effector-based RAGT in patients with various neurological disorders, although gait outcomes were only reported as secondary endpoints. Clinically meaningful improvements (≥20%) in walking speed and distance were observed in a subset of participants. The limited overall effect may be due to the inclusion of conditions with low rehabilitation potential, such as motor neuron disease [7].
Interpreted within the context of individual diagnostic groups, indicate that despite fewer sessions and a relatively shorter therapy duration, our results fall within the range of improvements observed in previously published studies. RAGT outcomes in both stroke and spinal cord injury (SCI) cohorts exhibit significant variability, influenced by the recovery phase, specific therapy protocol, and the type of robotic system employed.
For stroke patients in the acute and subacute phases, typical 10MWT improvements range from 20% to 60%, with 6MWT distance gains often greater [40–85%), sometimes involving up to 36 sessions [18,21,22]. In the chronic phase, outcomes tend to be more limited. For instance, Aprile et al. reported a 21% increase in walking speed and a 25% improvement in distance [23]. Even smaller effects were observed by Kelly C.P. et al., with minimal changes in 10MWT and only a 7% gain in 6MWT using an exoskeleton-based RAGT device [24].
Similarly, in SCI, studies report a range of outcomes: Varoqui et al. noted a modest 14% increase in walking speed with no significant 6MWT improvement after 12 sessions [25], while Chang et al. observed a 23% gain in speed and a 34% increase in distance after 15 sessions [26]. Conversely, Wirz et al. demonstrated substantial improvements exceeding 50% in both parameters after 36 sessions [27]. Beyond the significant differences in the number of sessions, this variability in SCI outcomes may also be attributed to the extent and location of the injury [28].
Research on RAGT in adult cerebral palsy (CP) patients is limited, with most studies focusing on pediatric populations. The findings of this study, however, are consistent with studies utilizing the same RAGT device in pediatric patients [29]. A potential explanation for this alignment is the relatively low mean age [(32.2 ± 12.35 years) of the CP cohort, suggesting a population with greater neuroplastic potential and functional adaptability compared to older adults, which may have positively influenced their training response despite typical concerns about limited adaptability in adult CP populations.
In addition, studies have demonstrated that RAGT is more effective than conventional gait therapy, particularly in individuals with moderate to severe motor impairments [9,17,18,21,23]. Beyond the core principles of RAGT, such as high-intensity, repetitive, and precisely guided gait training that drives neuroplasticity, a key advantage is the possibility to start rehabilitation early, even in the acute phase after injury. This is especially important in cases like stroke, where the greatest potential for recovery occurs within the first three months [14,18]. End-effector devices in particular offer several advantages, including a more physiological gait pattern, increased freedom of movement, and a more demanding postural environment, as users are required to actively maintain balance. These characteristics may contribute to findings from some studies showing that end-effector systems lead to better performance in functional tests and higher rates of independent walking compared to, especially, stationary exoskeleton-based systems [7,15,18,22].
The experimental outcomes of the present study align with the theoretical recovery potential inherent to each condition targeted by RAGT. Post-stroke patients, particularly in the acute and subacute phases, demonstrate the greatest potential for gait restoration. This is primarily due to preserved spinal cord function and the brain’s robust capacity for neuroplastic reorganization, where new neural connections are formed and existing ones strengthened following injury. Furthermore, RAGT actively stimulates central pattern generators (CPGs) in the spinal cord, which control the basic rhythmic patterns of walking, and may also activate dormant but viable motor units, thereby facilitating improved motor output and functional recovery [30,31]. In contrast, patients with spinal cord–related neuropathies may exhibit a lower recovery potential. Damage to spinal pathways directly disrupts crucial brain–body communication and impairs CPG function. This, combined with the spinal cord’s inherently limited plasticity and the frequent presence of severe structural lesions, further constrains functional recovery. Consequently, improvements in this group tend to be smaller and slower [28,32,33].
This study has several limitations that should be considered. The lack of a control group prevents direct comparison with conventional therapy and limits conclusions about the specific effects of RAGT. The small sample size in each subgroup reduces statistical power, while the high heterogeneity of the participants complicates interpretation. Important clinical variables such as lesion extent, time since injury, and baseline functional status (e.g. FAC score) were not clearly defined or stratified, which may have introduced confounding factors. Moreover, the study would benefit from the inclusion of objective functional assessments or patient-reported outcome measures to better capture the impact of RAGT on daily functioning and quality of life.
Despite the mentioned limitations, particularly the small sample size and high heterogeneity within groups, the study demonstrated a positive effect of the novel end-effector based RAGT system on walking ability in patients with various neurological conditions, reflecting the complexity of real-world rehabilitation populations. Moreover, the results suggested a potential correlation between the type of neurological impairment and the capacity for gait recovery with RAGT.
Conclusion
The study demonstrated statistically significant improvements in gait parameters, specifically step count (an average increase of 45%), walking distance [(59%), and walking cadence (48%) in adults with various neurological conditions. The best results were observed in patients with cerebrovascular disorders, while the lowest improvements occurred in those with spinal cord-related neuropathies. These findings confirm the positive impact of the end-effector RAGT system across a wide spectrum of neurological diagnoses, reflecting the heterogeneity encountered in everyday clinical practice and indicating the potential of this technology as an effective alternative to conventional therapies, which often require significant personnel resources and may have limited efficacy.
References
- Gjesdal B, Jahnsen R, Morgan P, Opheim A, Mæland S (2020) Walking through life with cerebral palsy: Reflections on daily walking by adults with cerebral palsy. International Journal of Qualitative Studies on Health and Well-being. [crossref]
- Jankovic J. GAIT disorders. Neurologic Clinics. 2014.
- Wuehr M, Huppert A, Schenkel F, Decker J, Jahn K, Schniepp R. Independent domains of daily mobility in patients with neurological gait disorders. Journal of Neurology. 2020 [crossref]
- Institute for Health Metrics and Evaluation (IHME) Global Burden of Disease 2021: Findings from the GBD 2021 Study. Seattle (2024)
- Khalid NS, Malik NAN, Siddiqi NFA, Rathore NFA. Overview of gait rehabilitation in stroke. Journal of the Pakistan Medical Association. 2023 [crossref]
- Awai L, Bolliger M, Ferguson AR, Courtine G, Curt A. Influence of spinal cord integrity on GAIT control in human spinal cord injury. Neurorehabilitation and Neural Repair. 2015 [crossref]
- Hotz I, Mildner S, Stampfer-Kountchev M, et al. Robot-assisted gait training in patients with various neurological diseases: A mixed methods feasibility study. PLoS ONE. 2024. [crossref]
- Bonnevie T, Moily K, Barnes S, McConaghy M, Ilhan E. People with spinal cord injury or stroke are able to reach moderate-to-vigorous intensity while exercising on an end-effector robot assisted gait trainer: A pilot study. Journal of Rehabilitation and Assistive Technologies Engineering. 2025 [crossref]
- Bonanno M, De Pasquale P, Facciale AL, et al. May Patients with Chronic Stroke Benefit from Robotic Gait Training with an End-Effector? A Case-Control Study. Journal of Functional Morphology and Kinesiology. 2025 [crossref]
- Ada L, Dean CM, Morris ME, Simpson JM, Katrak P. Randomized trial of treadmill walking with body weight support to establish walking in subacute stroke. Stroke. 2010. [crossref]
- Capecci M, Pournajaf S, Galafate D, et al. Clinical effects of robot-assisted gait training and treadmill training for Parkinson’s disease. A randomized controlled trial. Annals of Physical and Rehabilitation Medicine. 2019 [crossref]
- Butt Y, Saghir M, Waseem I, Akmal S, Khan UA, Wdood A. Effects of Treadmill Training on Gross Motor Function, Spasticity and Gait Speed in Ambulatory Children with Spastic Cerebral Palsy. Journal of Riphah College of Rehabilitation Sciences. 2024
- Duncan PW, Sullivan KJ, Behrman AL, et al. Body-Weight–Supported Treadmill Rehabilitation after Stroke. New England Journal of Medicine. 2011 [crossref]
- Pichardo A, Malovec M. Enhancing post-stroke gait rehabilitation with robot-assisted therapy: a focus on step repetitions and neuroplasticity. Arch Clin Biomed Res. 2025.
- Choi S, Kim SW, Jeon HR, Lee JS, Kim DY, Lee JW. Feasibility of Robot-Assisted Gait Training with an End-Effector Type Device for Various Neurologic Disorders. Brain & Neurorehabilitation. 2019 [crossref]
- Park YH, Lee DH, Lee JH. A Comprehensive review: Robot-Assisted Treatments for GAIT Rehabilitation in Stroke patients. Medicina. 2024 [crossref]
- Yang FA, Chen SC, Chiu JF, et al. Body weight-supported gait training for patients with spinal cord injury: a network meta-analysis of randomised controlled trials. Scientific Reports. 2022 [crossref]
- Lee J, Kim DY, Lee SH, et al. End-effector lower limb robot-assisted gait training effects in subacute stroke patients: A randomized controlled pilot trial. Medicine. 2023 [crossref]
- Bonanno M, Maggio MG, Ciatto L, et al. Can Robotic Gait Training with End Effectors Improve Lower-Limb Functions in Patients Affected by Multiple Sclerosis? Results from a Retrospective Case–Control Study. Journal of Clinical Medicine. 2024 [crossref]
- Mehrholz J, Pohl M. Electromechanical-assisted gait training after stroke: A systematic review comparing end-effector and exoskeleton devices. Journal of Rehabilitation Medicine. 2012 [crossref]
- Aprile I, Iacovelli C, Goffredo M, et al. Efficacy of end-effector Robot-Assisted Gait Training in subacute stroke patients: Clinical and gait outcomes from a pilot bi-centre study. Neurorehabilitation. 2019 [crossref]
- Neves MVM, Furlan L, Fregni F, Battistella LR, Simis M. Robotic-Assisted GAIT Training (RAGT) in stroke Rehabilitation: a pilot study. Archives of Rehabilitation Research and Clinical Translation. 2023 [crossref]
- Aprile I, Iacovelli C, Padua L, et al. Efficacy of Robotic-Assisted Gait Training in chronic stroke patients: Preliminary results of an Italian bi-centre study. Neurorehabilitation. 2017. [crossref]
- Kelley CP, Childress J, Boake C, Noser EA. Over-ground and robotic-assisted locomotor training in adults with chronic stroke: a blinded randomized clinical trial. Disability and Rehabilitation Assistive Technology. 2012 [crossref]
- Varoqui D, Niu X, Mirbagheri MM. Ankle voluntary movement enhancement following robotic-assisted locomotor training in spinal cord injury. Journal of NeuroEngineering and Rehabilitation. 2014 [crossref]
- Chang SH, Afzal T, Berliner J, Francisco GE. Exoskeleton-assisted gait training to improve gait in individuals with spinal cord injury: a pilot randomized study. Pilot and Feasibility Studies. 2018 [crossref]
- Wirz M, Zemon DH, Rupp R, et al. Effectiveness of automated locomotor training in patients with chronic incomplete spinal cord injury: A multicenter trial. Archives of Physical Medicine and Rehabilitation. 2005 [crossref]
- Park JM, Kim YW, Lee SJ, Shin JC. Robot-Assisted GAIT training in Individuals with Spinal Cord Injury: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Annals of Rehabilitation Medicine. 2024 [crossref]
- Malovec M (2024) Impact of End-Effector Device on Gait Restoration in Children Suffering from Neurological Disorders. Archives of Clinical and Biomedical Research.
- Huang H, Su X, Zheng B, Cao M, Zhang Y, Chen J. Effect and optimal exercise prescription of robot-assisted gait training on lower extremity motor function in stroke patients: a network meta-analysis. Neurological Sciences. 2024. [crossref]
- Huang DF, Xu Y, Hou QH, et al. Neuroplasticity in post-stroke gait recovery and noninvasive brain stimulation. Neural Regeneration Research. 2015 [crossref]
- Bareyre FlorenceM, Loy K. Rehabilitation following spinal cord injury: how animal models can help our understanding of exercise-induced neuroplasticity. Neural Regeneration Research. 2018 [crossref]
- Harkema S, Dobkin B, Edgerton VR (2000) Pattern Generators in Locomotion: Implications for recovery of walking after spinal cord injury. Topics in Spinal Cord Injury Rehabilitation.