Abstract
A 51-year-old man with hypertension and diabetes presented with proximal muscle weakness and severe hypokalemia. Hormonal evaluation confirmed ACTH-dependent Cushing’s syndrome, with extremely elevated cortisol levels (>1100 ng/ml), consistent with a severe form of the condition. Imaging and biopsy identified an ectopic ACTH-secreting thymic neuroendocrine tumor (atypical carcinoid) with chromogranin A positivity. Initial medical therapy with fluconazole failed to reduce cortisol levels. To mitigate perioperative risk from hypercortisolemia, a low-dose overnight etomidate infusion was initiated, successfully lowering cortisol levels below 300 ng/dl. Surgical excision of the tumor was then performed. Histology confirmed atypical carcinoid with a Ki-67 index of 7.5%, and evidence of capsular and vascular invasion. Postoperatively, cortisol and ACTH levels dropped significantly, and the patient was started on physiological hydrocortisone replacement. He experienced no complications from cortisol excess and is currently being monitored for disease re-staging. The case underscores etomidate’s value in preoperative cortisol control in severe ectopic Cushing’s syndrome.
Keywords
Ectopic Cushing’s syndrome, Atypical thymic carcinoid, Etomidate, Severe Cushing’s syndrome
Background
Ectopic Cushing’s syndrome is a rare condition that contributes to 10-20% of Adrenocorticotropic Hormone (ACTH)-dependent Cushing’s syndrome cases. Severe Cushing’s syndrome, which is defined when serum cortisol levels are above 1100 ng/ml, is rare in ectopic Cushing’s syndrome and carries higher perioperative and postoperative morbidity and mortality. Distinguishing ectopic Cushing’s syndrome from other causes of Cushing’s syndrome is imperative, as removing the tumor can cure the condition. Normalizing cortisol levels prior to surgical removal is crucial to reduce peri- and postoperative morbidity and mortality.
We present a case of severe ACTH-dependent Cushing’s syndrome due to an ectopic ACTH-secreting atypical thymic carcinoid tumor, which we successfully treated with etomidate pre-operatively, leading to good postoperative outcomes. In environments where intensive care facilities are scarce, high-dependency units (HDUs) may offer a safe and effective alternative for administering low-dose etomidate infusion to manage hypercortisolemia. In this particular case, serum cortisol levels were successfully reduced to approximately 300 nmol/L without inducing cardio-respiratory compromise.
Pre-operative etomidate prior to definitive surgical treatment of thymic carcinoid tumors helps to minimize complications.
Introduction
Hypercortisolism and the range of symptoms associated with it are collectively referred to as Cushing’s syndrome. Exogenous Cushing’s syndrome is the most common type, while endogenous Cushing’s syndrome occurs secondary to excess cortisol, which may be due to ACTH-dependent and ACTH-independent mechanisms. ACTH-dependent causes account for 70–80% of cases, while 20–30% of cases are ACTH-independent. ACTH-dependent Cushing’s syndrome can occur due to pituitary tumors or tumors at ectopic sites, and ACTH-independent Cushing’s syndrome is usually caused by adrenal lesions [1,2].
Ectopic Cushing’s syndrome is a rare condition contributing 5-20% of all Cushing’s syndrome cases and 10-20% of all ACTH-dependent Cushing’s syndrome cases. The commonest sites for ectopic ACTH secretion are the lung, mediastinum, gastro-entero-pancreatic neuroendocrine tumors, or pheochromocytomas, respectively [3].
Severe Cushing’s syndrome is defined by random serum cortisol levels more than 40 μg/dL (1100 nmol/L) at any time or a 24-hour urinary free cortisol more than fourfold the upper limit of normal and/or severe hypokalemia (<3.0 mmol/L). Severe hypercortisolism is associated with a higher risk of infection, septicemia, thromboembolism, and postoperative morbidity compared to mild and moderate Cushing’s syndrome. Thus, reducing hypercortisolism at the time of intervention may benefit these patients. Depending on the lesion site, we can categorize the available treatment options into medical, surgical, and radiotherapy-related treatments. It’s pivotal to differentiate ectopic Cushing’s syndrome from other causes because identification of the ectopic site and surgical removal may completely cure the disease [4].
We present a case report on a 51-year-old Sri Lankan male who was diagnosed with severe ACTH-dependent Cushing’s syndrome secondary to ectopic ACTH-secreting atypical thymic carcinoid tumor. He was successfully treated using low-dose etomidate with minimal postoperative complications. This case report highlights the benefits of preoperative low-dose etomidate treatment, leading to a successful postoperative outcome in managing severe hypercortisolism. Also, in environments where intensive care facilities are scarce, high-dependency units (HDUs) may offer a safe and effective alternative for administering low-dose etomidate infusion to manage hypercortisolemia. In this particular case, serum cortisol levels were successfully reduced to approximately 300 nmol/L without inducing cardio-respiratory compromise.
Case Presentation
A 51-year-old male who is a known patient with well-controlled diabetes mellitus and hypertension presented with the complaint of episodic diarrhea over a 2-week duration and difficulty in standing from a squatting position, suggesting proximal muscle weakness. He also had sinister lower back pain that was persistent throughout the day with the same intensity, with no recent history of trauma. He noted facial flushing and mild facial puffiness along with exertional shortness of breath and excessive tiredness with routine tasks.
He had severe constitutional symptoms, including weight loss of nearly 10 kg over the last 2 months with loss of appetite. The patient denied any recent onset of pigmentation, palpitations, episodic flushing, or wheezing episodes in the recent past. The patient complained of visual and auditory hallucinations with depressive symptoms, episodically over the last two months. There were no visual disturbances, history of dyspeptic symptoms, renal stone disease, or any family history of similar disease to suggest multiple endocrine neoplasia (MEN) syndrome.
On examination, the patient had a BMI of 19.53 kg/m² with an initial blood pressure of 180/100 mmHg. He had flushing involving the face and upper chest with thin skin and subtle nail hyperpigmentation. Overt clinical signs of Cushing’s syndrome, including buffalo hump, purple abdominal striae, or hirsutism, were not seen. The patient exhibited proximal muscle weakness with a power of 3/5 in both the upper and lower limbs, in contrast to distal muscle power, which was 4/5. His mental status examination revealed visual and auditory hallucinations with a moderate degree of depression. Throughout the hospital stay, it was noted that he had poor blood pressure control with evidence of resistant hypertension and poor glycemic control (Table 1).
Table 1: Investigations
| White cell count | 14.98 × 109 |
| Hemoglobin | 11.7 g/dL |
| Platelet count | 238 × 103 |
| Serum sodium | 141 mmol/L (136-145) |
| Serum potassium | 2.1 mmol/L (3.5-5.1) |
| Erythrocyte sedimentation rate | 45 mm/1st hour |
| Aspartate transferase | 58 U/L (11-34) |
| Alanine transaminase | 145 U/L (<45) |
| Total Bilirubin | 1.9 mg/dl |
| Direct Bilirubin | 1.0 mg/dl |
| Alkaline phosphatase | 350 mu/L |
| Gamma-glutamyl transferase | 284 mu/L |
| Albumin | 2.1 g/dL |
| Globulin | 2.7 g/dL |
| 9am cortisol (initial) | 1220 nmol/l (118.6-618) |
| 9am cortisol (Post fluconazole for 1 week) | 1100 nmol/L |
| Overnight dexamethasone suppression test (ODST) | 880.33 nmol/L (≤50) |
| Low dose dexamethasone suppression test (LDDST) | 260.3 nmol/L (≤50) |
| Cortisol day curve | 9am 674.95 nmol/L |
| 11am 1038.61 nmol/L | |
| 3pm 760.38 nmol/L | |
| 5pm 797.55 nmol/L | |
| Adrenocorticotrophic Hormone (ACTH) level | 172 pg/ml (4.7-48.8)—— 81 pg/ml (Post operatively) |
| Albumin corrected Calcium | 8.9 mg/dL (8.6 to 10.3 mg/dL) |
| Serum Prolactin | 203.24 mIU/L (73-412) |
| Abdominal ultrasound (USS) | Normal USS abdomen with no evidence of organomegaly or adrenal lesions. |
| Contrast-Enhanced Computed Tomography (CECT) chest- abdomen- pelvis | Lobulated soft tissue density lesion in the anterior mediastinum measuring 3.3 cm × 4.8 cm × 5 cm.
Lesion shows avid contrast enhancement with internal non-enhancing areas. No calcifications. It compresses the left subclavian vein posteriorly, and in-between fat planes are obliterated. Fat planes between the lesion and SVC are also obliterated. Enlarged lymph nodes are seen in the right lower paratracheal and upper paratracheal regions.
|
| USS guided FNAC of the mediastinal lesion | Cellular smears reveal epithelial clusters and single cells with acinar arrangement and sheets.
Nuclei are prominent with fine speckled chromatin with a few enlarged nuclei and eosinophilic cytoplasm. No admixed lymphocytes or keratin pearls. Ki67 30% Suggestive of neuroendocrine carcinoma of the thymus. |
| Serum Chromogranin A | <39 ng/L (Normal- < than 39 ng/l) |
| Serum 5- HIAA levels | 2.1 mg/24 hrs (2-9) |
| Histology of the thymic gland biopsy specimen (Figure 4) | Atypical Carcinoid tumor with lympho-vascular invasion and infiltration of inferior margins and capsule invasion.
All mediastinal and pre-tracheal nodes were positive for tumor invasion. (PT1a pN2 IV B) |
| Post operative 9am cortisol | 358 nmol/L |
| Post operative ACTH | 81 pg/ml |
| DOTATE-PET imaging | Not done due to unavailability |
| Post-operative short synacthen test | Basal 121.50 nmol/L
30 min 233.50 nmol/L 60 min 315.60 |
After confirming ectopic ACTH-dependent severe Cushing’s syndrome with elevated serum cortisol levels above 1100 ng/dl and non-suppressed ODST and LDDST, we started the patient on oral fluconazole 150 mg twice daily as a steroid synthesis inhibitor. Given the presence of severe hypokalemia, ectopic ACTH syndrome was suspected. As a result, we proceeded directly with a CECT of the chest, abdomen, and pelvis to search for an ectopic tumor, which revealed a thymic lesion in the anterior mediastinum. Severe hypokalemia with serum potassium levels of 2.1 mg/dl was treated with oral and intravenous supplementation of potassium. Even though there was no liver metastasis, the presence of facial flushing and the history of diarrhea prompted us to exclude the possibility of carcinoid syndrome in this patient. USS guided FNAC of the mediastinal lesion confirmed the tumor as a neuroendocrine carcinoid tumor of the thymus, without evidence of carcinoid syndrome, as indicated by non-measurable levels of 5-HIAA and serum Chromogranin A.
Treatment
Thus, the cardiothoracic surgical team’s opinion was taken to embark on surgery. Given the risk of mobilization of the carcinoid tumor to prevent carcinoid crisis, the patient was started on subcutaneous octreotide 50 mg every 6 hours two weeks prior to surgery.
Protocol for Low Dose Etomidate
The patient was started on a low-dose etomidate infusion of 0.05 mg/kg/hr on the day before surgery in an intensive care setup to closely monitor respiratory compromise and hemodynamic instability (Figures 1 and 2).
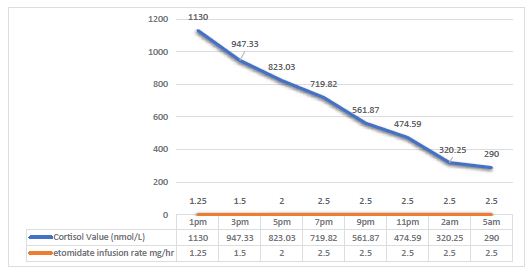
Figure 1: Serum cortisol levels following low dose etomidate infusion.
Patient’s serum cortisol levels were measured 2 hourly, aiming at a target cortisol level of <300 ng/dl. The etomidate infusion rate was slowly titrated to obtain a cortisol drop of around 100 nmol/l/hr.
Once cortisol levels reached <300 nmol/L, low-dose etomidate infusion was stopped. Within 16 hours of low-dose etomidate infusion, the expected range of cortisol was achieved (Figures 1 and 2).
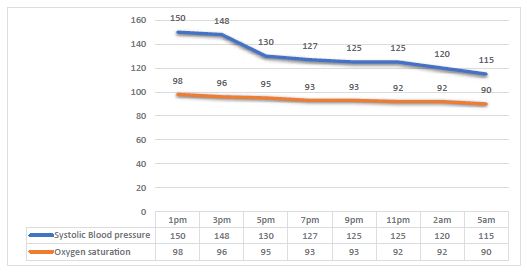
Figure 2: Blood pressure and oxygen saturation alteration with time.
The patient was operated on through median sternotomy, and an anterior mediastinal mass was removed. The tumor was measured 5 cm x 6 cm x 3 cm and weighed 140 g (Figure 3). Histology of the anterior mediastinal mass was suggestive of atypical carcinoid tumor of the thymic gland with lymphovascular invasion and infiltration of inferior margins and capsule invasion (Figure 4).
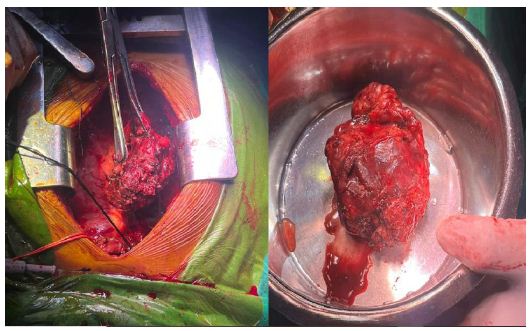
Figure 3: Macroscopic appearance of the tumor during surgery.
Outcome and Follow Up
The patient had postoperative symptomatic and biochemical improvement (Table 1). The post-operative cortisol level was 78 nmol/L and ACTH level was 81pg/ml showing partial cure of disease. Despite the partial cure patient did not develop any complications of cortisol excess during the post-operative period including infections or venous thromboembolism. Patient was started on physiological doses of hydrocortisone until the recovery of hypothalamo-pituitary -adrenal axis. (HPA axis). He is awaiting re-imaging for re-staging of the thymic tumor in 6 months with assessment for HPA axis recovery (Figure 3 and 4).
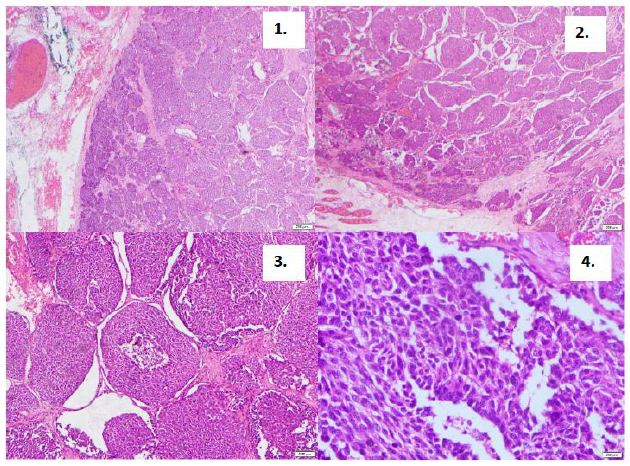
Figure 4: Thymic gland histology : hematoxylin and eosin (x400 magnification).
Discussion
Ectopic ACTH-secreting tumors account for 5 to 10% of cases of ACTH-dependent Cushing’s syndrome, which is commonly caused by thymic, bronchial, gastrointestinal, and pancreatic neuroendocrine tumors. Ectopic ACTH-secreting tumors can be challenging to identify, with up to 19% of cases lacking clear tumor localization. Hypokalemia is an important clue pointing toward ectopic ACTH-secreting tumors. It occurs in 80% of ectopic ACTH-secreting tumors, which is due to the mineralocorticoid action of excess steroids and the decreased 11-hydroxysteroid dehydrogenase type 2 [7].
Carcinoid tumors, which were renamed by the World Health Organization as neuroendocrine tumors in 2000, are derived from enterochromaffin cells. These tumors release biologically active amines and peptides, including serotonin, histamine, and prostaglandins. Carcinoid syndrome is a group of symptoms brought on by the release of biologically active substances. It happens in about 10% of neuroendocrine tumors. Most of these clinical features are due to serotonin, which is an end product of tryptophan metabolism. But the thymic neuroendocrine tumor, which was observed in our patient, is a foregut tumor that lacks the enzyme aromatic L-amino acid decarboxylase, which metabolizes 5-hydroxytryptophan to serotonin [27]. Therefore, they theoretically do not produce serotonin or cause carcinoid syndrome, which was true in view of our patient.
Neuroendocrine tumors of the thymus are rare, accounting for approximately 0.4% of all neuroendocrine tumors [20]. 80% of these tumors are malignant [21]. These tumors are larger in size (median 7.9 cm) compared to thymic carcinoma and are usually present before the advanced stage [22]. According to the 2015 tumor classification by the World Health Organization, NETTs were included in the thymic cancer group. These were classified into well-differentiated neuroendocrine carcinomas (typical and atypical carcinoids) and poorly differentiated ones (small-cell and large-cell neuroendocrine carcinomas) [23]. Atypical carcinoid, which was the diagnosis in our patient, accounts for about 40–50% of NETTs, and middle-aged adults (48–55 years) were commonly affected [24].
Approximately 50% of NETTs were associated with endocrinopathies, including Cushing’s syndrome (ectopic ACTH production) and acromegaly (growth hormone releasing hormone hypersecretion). Multiple endocrine neoplasia-1 was seen in approximately 20% of NETTs [25].
Surgical resection of the tumor removes the source of ectopic ACTH secretion, which is the treatment of choice for ectopic ACTH-secreting tumors. In NETTs, the resectability rate ranged from 28% to 100% in a published single-center case series, but this may depend on the surgical experience in that center [26]. Hypercortisolemia can increase peri- and postoperative complications and mortality in these patients. So, it is important to normalize cortisol levels as early as possible and preoperatively to minimize the adverse effects associated with hypercortisolism, such as increased risk of infection, thromboembolism, etc., which are the main factors that contribute to Cushing’s syndrome mortality [12].
There are several medical options that can be used to control Cushing’s syndrome, including ketoconazole, mitotane, metyrapone, etomidate, and the newer medication osilodrostat. Etomidate, an induction anesthetic, in its lower doses reduced serum cortisol levels by inhibiting 11β-hydroxylase action. 11β-hydroxylase catalyzes cortisol conversion from deoxycortisol, whereas at its higher concentrations, it inhibits the conversion of cholesterol to pregnenolone [13].
The mean rate of drop of cortisol in literature is 104.3 nmol/L/hr; that is when etomidate is used as a first-line medication to reduce hypercortisolism. It has been observed to have a lesser mean percentage of drop when it becomes the second or third line treatment modality, 62% and 41%, respectively [14]. Our patient had a mean rate of drop of cortisol of ~134.3 nmol/L/hr, where we used etomidate as second-line therapy. Also, according to literature, when etomidate was used as the first-line medication, the time to reach the expected baseline was around 15 hours, and if cortisol is used as a second-line or third-line, it has been observed to take more time to achieve its effect. (80 hours and 48 hours, respectively) [14]. In our patient, even though we used etomidate as a second-line medication, we were able to achieve the expected baseline value within 16 hours of initiating the infusion.
It has been shown that during the treatment for hypercortisolemia with etomidate, ACTH-independent Cushing’s syndrome had a higher incidence of adrenal insufficiency than ACTH-dependent Cushing’s syndrome. The mechanism responsible would be the increased production of cortisol under the influence of ACTH. Therefore, ACTH-dependent Cushing’s syndrome might need higher doses of etomidate in the management [15]. Our patient needed the dose to achieve the expected cortisol level, which was 0.05 mg/kg/hr. This might infer higher benefit and safety of etomidate in cases of ACTH-dependent Cushing’s syndrome, rather than in ACTH-independent cases.
The use of etomidate for acute management of severe hypercortisolemia was first described in 2012, and the current protocol for continuous infusion of low-dose etomidate for Cushing’s syndrome was validated in 2019 by Carroll et al. [16]. They describe giving a 5 mg bolus dose followed by an infusion of etomidate with a dose of 0.02 mg/kg/hr and up titration aiming for a maximum dose of 0.3 mg/kg/hr with 6-hourly up-titration of the etomidate dose if necessary. This method was proven to be minimally associated with cardio-respiratory compromise. In our patient we did not give the bolus dose, rather continued the low dose continuous infusion of etomidate with up-titration until the expected rate of decline was achieved.
Studies have experimented on the use of etomidate in ICU setups vs. ward setups [17,18]. Agnieszka et al. had carried out very low dose infusions of 0.1-0.2 mg/kg/hr with a bolus dose of 2.5 mg at the outset for prolonged periods of time without any hemodynamic compromise, thus suggesting the safety of etomidate infusion, whereas in Constantinescu et al., the study did not reveal any difference in the outcomes when etomidate was used in the ICU setup vs. the inward setup. For our patient, we were able to arrange an ICU bed for close monitoring of parameters. In our patient, we used a 0.05 mg/kg/hr infusion safely without any fluctuations in hemodynamic status. However, in a resource-poor setup, for the acute management of severe hypercortisolemia, unavailability of an intensive care unit should not be a limiting factor, given the extensive availability of the drug due to its use for anesthetic purposes and its cost-effectiveness.
It is also possible to use the “block and replace” strategy with intravenous hydrocortisone to reach a serum cortisol goal of 500–800 nmol/L, but only for a short time [19]. But in our patient, we did not adhere to the block-and-replace strategy and only used etomidate at 0.05 mg/kg per hour. We maintained serum cortisol around 300 nmol/L. We did not observe any cardiorespiratory complications or drastic electrolyte alterations in our patient. Due to the difficulty in allocating an ICU bed upon our observation, we propose to start etomidate in the ward in the high dependency unit under careful monitoring. Etomidate, its complications, and cost analysis are areas that need future research.
Learning Points
- Ectopic ACTH-secreting atypical thymic carcinoid tumors are a rare cause of severe Cushing’s Syndrome.
- Administering an initial dose of 0.05 mg/kg/hour of the anesthetic agent etomidate in an HDU setting, even without Intensive care facilities to lower serum cortisol levels is safe while maintaining the serum cortisol around 300nmol/L without leading to any cardio-respiratory compromise.
- Pre-operative etomidate prior to definitive surgical treatment of thymic carcinoid tumors helps to minimize complications.
Funding
None
Competing Interests
Authors declare that they have no competing interests.
Contribution
Dr. W.M.D.A.S. Wanninayake, Dr. H.S.Senanayake, Dr. U.C.Hettiarachchi, Dr. Manilka Sumanathilake were involved in managing the patient. Dr.W.M.D.A.S. Wanninayake and Dr. Tilan Aponso did the literature review and writing of the initial manuscript was done by Dr.W.M.D.A.S. Wanninayake. Dr. Manilka Sumanathilake finalized the manuscript and gave expert opinion. All the authors read and approved the final manuscript.
Acknowledgements
None
Ethical Declaration
Not applicable
Consent for Publication
Informed written consent for publication of details was taken from the patient. Consent form can be made available to the editor on request.
Availability of Data and Materials
The data is available from the corresponding author on reasonable request.
References
- Lacroix A, Feelders RA, Stratakis CA, Nieman LK (2015). Cushing’s syndrome. Lancet. [crossref]
- Hirsch D, Tsvetov G, Manisterski Y, et al. (2017). Incidence of Cushing’s syndrome in patients with significant hypercortisoluria. Eur J Endocrinol. [crossref]
- Hayes AR, Grossman AB (2022). Distinguishing Cushing’s disease from the ectopic ACTH syndrome: Needles in a haystack or hiding in plain sight? J Neuroendocrinol. [crossref]
- Alexandraki KI, Grossman AB (2016). Therapeutic Strategies for the Treatment of Severe Cushing’s Syndrome. Drugs. [crossref]
- Maragliano R, Vanoli A, Albarello L, et al. (2015). ACTH-secreting pancreatic neoplasms associated with Cushing syndrome: clinicopathologic study of 11 cases and review of the literature. Am J Surg Pathol. [crossref]
- Davi’ MV, Cosaro E, Piacentini S, et al. (2017). Prognostic factors in ectopic Cushing’s syndrome due to neuroendocrine tumors: a multicenter study. Eur J Endocrinol. [crossref]
- Ilias I, Torpy DJ, Pacak K, Mullen N, Wesley RA, Nieman LK (2005). Cushing’s Syndrome Due to Ectopic Corticotropin Secretion: Twenty Years’ Experience at the National Institutes of Health. J Clin Endocrinol Metab. [crossref]
- Engels EA (2010). Epidemiology of thymoma and associated malignancies. J Thorac Oncol. [crossref]
- Chaer R, Massad MG, Evans A, et al. (2002). Primary neuroendocrine tumors of the thymus. Ann Thorac Surg. [crossref]
- Gaur P, Leary C, Yao JC (2010). Thymic Neuroendocrine Tumors: A SEER Database Analysis of 160 Patients. Ann Surg. [crossref]
- Meinardi JR, Wolffenbuttel BH, Dullaart RP (2007). Cyclic Cushing’s syndrome: a clinical challenge. Eur J Endocrinol. [crossref]
- Gaur P, Leary C, Yao JC (2010). Thymic neuroendocrine tumors: a SEER database analysis of 160 patients. Ann Surg. [crossref]
- Pence A, McGrath M, Lee SL, Raines DE (2022). Pharmacological management of severe Cushing’s syndrome: the role of etomidate. Ther Adv Endocrinol Metab. [crossref]
- Muthukuda D, Jayawickreme KP, Liyanarachchi K, Pathmanadan S, Sumanatilleke M, Kurupiah D, Cooray S, Antonypillai C, Weerakkody M, Somasundaram N, Subasinghe C (2024). Etomidate in the Treatment of Severe Cushing Syndrome: The Sri Lankan Experience. Sri Lanka J Diabetes Endocrinol Metab.
- Preda VA, Sen J, Karavitaki N, Grossman AB (2012). Etomidate in the management of hypercortisolaemia in Cushing’s syndrome: a review. Eur J Endocrinol.
- Carroll TB, Peppard WJ, Herrmann DJ, et al. (2018). Continuous Etomidate Infusion for the Management of Severe Cushing Syndrome: Validation of a Standard Protocol. J Endocr Soc. [crossref]
- Łebek-Szatańska A, Nowak KM, Papierska L (2020). Etomidate infusion for severe Cushing’s syndrome outside intensive care unit. Eur J Endocrinol. [crossref]
- Constantinescu SM, Driessens N, Lefebvre A, Furnica RM, Corvilain B, Maiter D (2020). Etomidate infusion at low doses is an effective and safe treatment for severe Cushing’s syndrome outside intensive care. Eur J Endocrinol. [crossref]
- Preda VA, Sen J, Karavitaki N, Grossman AB (2012). Etomidate in the management of hypercortisolaemia in Cushing’s syndrome: A review. Eur J Endocrinol. [crossref]
- Yao JC, Hassan M, Phan A, et al. (2008). One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. [crossref]
- Klemm KM, Moran CA (1999). Primary neuroendocrine carcinomas of the thymus. Semin Diagn Pathol. [crossref]
- Filosso PL, Yao X, Ruffini E, et al. (2016). Comparison of outcomes between neuroendocrine thymic tumours and other subtypes of thymic carcinomas: a joint analysis of the European Society of Thoracic Surgeons and the International Thymic Malignancy Interest Group. Eur J Cardiothorac Surg. [crossref]
- Travis WD, Brambilla E, Burke AP, et al. (2015). WHO classification of tumours of the lung, pleura, thymus and heart, Fourth edition. International Agency for Research on Cancer.
- Ströbel P, Zettl A, Shilo K, et al. (2014). Tumor genetics and survival of thymic neuroendocrine neoplasms: a multi-institutional clinicopathologic study. Genes Chromosomes Cancer. [crossref]
- Phan AT, Oberg K, Choi J, et al. (2010). NANETS consensus guideline for the diagnosis and management of neuroendocrine tumors: well-differentiated neuroendocrine tumors of the thorax (includes lung and thymus). Pancreas. [crossref]
- Cardillo G, Treggiari S, Paul MA (2010). Primary neuroendocrine tumors of the thymus: a clinico-pathologic and prognostic study in 19 patients. Eur J Cardiothorac Surg.
- Pinchot SN, Holen K, Sippel RS, Chen H (2008). Carcinoid tumors. Oncologist. [crossref]