DOI: 10.31038/SRR.2018111
Abstract
Reconstruction and restoration of dura is an important issue in many cases of craniotomy. In cases that must be replaced large defects of dura, the usage of artificial dura is increasing.
During the period between 2013 till 2017, a total of 94 patients who underwent craniotomy for various reasons and collagen Dural plates were applied for Dural defects repairing were recruited to the study. During the follow-up of these patients, 10 patients were excluded from the study, which from the total of the remaining 84 patients, 11 cases of infection “13%” were observed. Eight cases of infection occurred as subdural empyema which were cured with reoperation treatment and evacuation of pus and dural repair. Cerebrospinal fluid leakage was seen 15.4% in 13 patients that all of them were improved with non-surgical supportive care.
Conclusion
With regards to the ease of using and least complications associated with the use of these artificial dural plates, it can be recommended to use it to dural restoration in the various craniotomy surgeries
Introduction
Proper Dural repair should be done to prevent cerebrospinal fluid leakage occurrence in craniotomy with any reason. In the cases that the primary defect of dura is too large that cannot be restored by the primary Dural tissue of the brain, we have to use alternatives to repair the underlying Dura. It is recommended to use a various type of dural alternatives that include the use of fasciae latae or peri-cranial tissue or artificial synthetic dura or ultimately tissues derived from allografts (cadaveric Dura) that each one has its own advantages and disadvantages [1–5].The usage of fasciae latae auto grafts don’t have much attractive due to the need for cutting and additional procedures with possible complications and pain caused by the removal of grafts ‘site or in cases where there are some reports from transmission of Creutzfeldtjakob caused by the use of allograft cases[6–8]. Synthetic collagen dural sheets are also safe alternatives to use as dural repair; because they are both easy to use and their reports of granulomatous reaction occurrence are rare[5, 9–12].In this study, we have discussed about results of the use of “ Aesculap onlay dura mater “ which exist as an artificial sticky sheets of Dura.
Materials and method
During the period between2013 till 2017, a total of 94 patients who underwent craniotomy for various reasons and collagen Dural plates were applied for Dural defects repairing were recruited prospectively to the study. The reasons that Patients were undergoing craniotomy include: tumors, brain hemorrhages, cerebral infarction and cerebro-vascular disease. In the process of dural restoration, we have used absorbable collagen plates of Dura “Aesculap onlay dura mater” in the size of 10cm × 10cm for each patient based on the underlying dural defects. These plates have been made of the underlying collagen scaffolds [10–12]. Sheets application is without the need for suture them to underlying dura. It will be embedded with the cover margin of at least 1cm from surrounding healthy Dura on the defect site (figure 1). Patients were followed up for three months after surgery. During the follow-up period, 10 patients died in a short period after surgery due to underlying medical conditions and were excluded from study. Of the remaining 84 patients, 25 patients “29.7%” had been subjected to surgery because of trauma, and 11 patients “% 13” had undergone craniotomy for brain infarction, 15 patients (17.8%) due to cerebral hemorrhage, and 12 patients “14.2%” for brain tumor and 21 patients “% 25” followed by cerebro-vascular lesions. (Table 1)
Table 1. Collagen dura usage results in patients
|
|
All cases |
Sub-galeal space Infections |
Sub-dural empyema |
Leak of cerebrospinal fluid |
|
trauma |
25 |
2 |
4 |
4 |
|
Cerebral infarctions |
11 |
0 |
0 |
0 |
|
Cerebral hemorrhage |
15 |
1 |
1 |
1 |
|
Tumor |
12 |
0 |
1 |
2 |
|
Vascular lesions |
21 |
0 |
2 |
6 |
|
total |
84 |
3 |
8 |
13 |
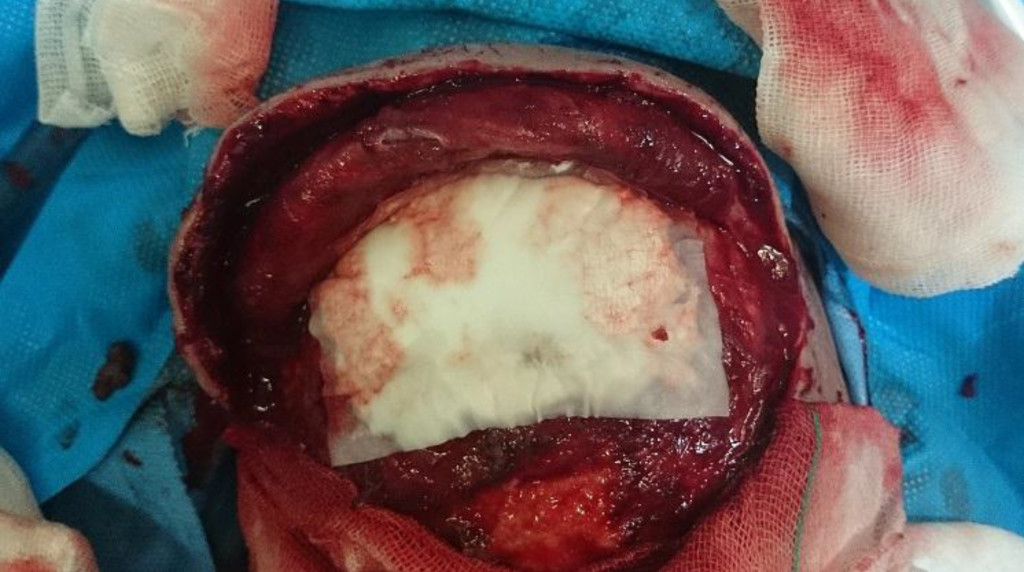
Figure 1. Collagen tissue dura sheets application
Underlying dural defects in traumatic bifrontal craniotomy , without the need for suture collagen tissue dura sheet is embedded with the cover margin of at least 1cm from surrounding healthy Dura on the defect site.
Results
Totally, infection was observed in 11 patients (13%) during follow-up period (Table1). According to the clinical and MRI findings, eight cases “% 9.5” were diagnosed as subdural empyema infection. sub-galeal space infection in three cases (3.5%). Diagnosis of sub-galeal space infection was obtained by pus discharge at the incision site and MRI findings. The cerebrospinal fluid leakage was observed in a total of 13 patients (15.4%) as a discharge of cerebrospinal clear fluid secretions from the incision site during the follow-up period. Of the total cases suffered from leakage, six cases of them were underwent surgery for cerebro-vascular lesions (46%) and four cases underwent surgery for traumatic brain injury (% 31); tumor and brain hemorrhage were constituted two patients (15%) and one patient (8%) respectively (Chart 1). All of CSF leakages were treated by conservative measures; however, there were three cases from four patients with cerebrospinal fluid leaks among patients undergoing surgery due to traumatic events experiencing subdural empyema. Two cases of cerebrospinal fluid leakage were among patients that undergoing surgery due to cerebro-vascular lesions who were experiencing subdural empyema. Cerebrospinal fluid leak in patients with cerebral hemorrhage and brain tumors has been improved by the conservative measures generally and none of the cases led to subdural empyema, or infection.
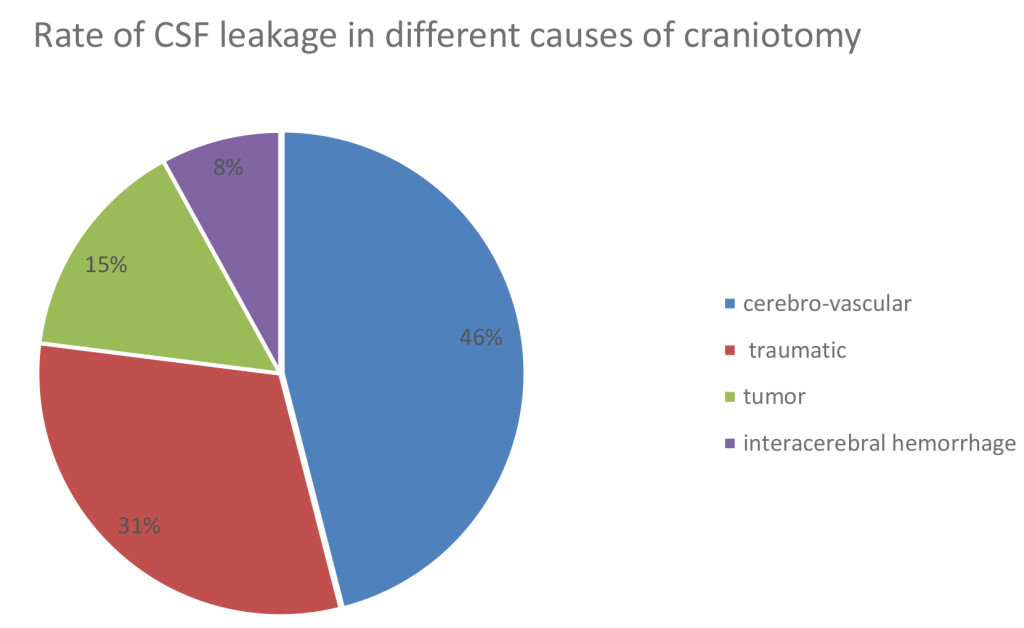
Chart 1. Rate of CSF leakage in different causes of craniotomy
Epidural accumulation of pus was occurred in all the three patients that two of them were observed in traumatic patients and one case occurred in a patient who was undergoing surgery due to brain hemorrhage. Subdural empyema patients were undergoing reoperation and the products of previous synthetic dura and pus collection were irrigated and evacuated completely, so that it was used from fasciaelatae autograft tissue to repair dural defect. After drainage, washing and continuing antibiotic treatment, six patients of a total of eight patients with subdural empyema were improved completely and pus accumulation in the subdural space was resolved in subsequent imaging and did not recur. These two patients were again subjected to surgery and re-drainage of the pus and subdural space washing due to the formation of pus in the subdural space that full recovery was observed with surgical repetition in these patients.
Of the three patients with sub-galeal accumulation, all of them were initially subjected to reoperation and pus discharge from the space of sub-galeal. One patient had fully recovered by this action and re-accumulation of pus was not found, but two other patients were undergoing surgical removal of previous synthetic dural tissues and replacing it with autograft Fasciae latae tissue due to re-accumulation of pus in follow-up MRIs which finally resulted in complete remission in these patients.
Discussion
Overall, infections in our patients was ultimately 13% during follow-up period. Infection rate of traumatic patients was totally more than the other groups that were subjected to surgery [54% of overall infection cases]. The least infections were among patients with cerebral infarction (no patient).
According to the Setsuko et al. (2003) which finally completed on 56 patients[13].The overall infection rates(14.3%) were reported in the use of artificial dural plates which was slightly higher than our study of collagen plates in the repair process. The percentage of sub-dural empyema incidence was reported 12.5% after the use of Dura plates here. While it has been observed 9.5% in our research.
Generally, the incidence of CSF leakage has been reported between4 -17% in other studies [14, 15], that is defined as sub-galeal accumulations of cerebrospinal fluid and its outflow through the sutures or its surrounding areas as the rhinorrhea or othorrhea . Totally, cerebrospinal fluid leakage was 15.4% in all of our studied patients. Highest amount of observed leakage was happened among the patients undergoing surgery for cerebro-vascular lesions “46%”, and the lowest amount was in patients undergoing surgery for cerebral infarctions that no case of leakage was found among them. The CSF leakage in patients who subjected to cerebro-vascular lesions surgery may be due to the need for a wider dissection in the sub-arachnoid space during surgery to create a wider space and visibility.
In the research of Than ko (2008), the amount of CSF leakage in the patients who had been used artificial dura to repair dural defects, was reported10%[14]. Huttera et al (2014) did not report any advantage in the reduction of CSF leakage by this method on patients using artificial dura as a normal suture compared to the use of artificial dura as a strengthening method; but in general, announced their two groups of patients CSF leakage incidence as 13.5% [15].
In general, it has been used artificial collagen plates in the studies and have shown good results, also the granulomatous reactions are relatively little to it. According to the present studies, fibroblastic cells begin to multiply after about a month in underlying tissue of artificial dura and coverage of fibroblastic tissue will be created along with surrounding dura [16, 17].
In our study, all the patients with subdural or sub-galeal infections were eventually recovered completely by reoperation, washing and removing the remnants of artificial dura and using fasciae latae tissue to repair underlying dura.
By comparing the results of similar studies in the use of artificial dural plates and the results of our study, the acceptable complications rate and control power to prevent the cerebrospinal fluid leakage incidence was perfect in the use of artificial dural plates, although using this material could be imagine a proper alternative to dural repair and prevention of a cerebro-spinal fluid leakage considering to the easiness of these artificial dural plates and no need to remove autograft tissue to repairing dura, which has its own complications.
Conclusion
With regards to the ease of using and least complications associated with the use of these artificial dural plates, it can be recommended to use it to dural restoration in the various craniotomy surgeries.
References
- Costa BS, Cavalcanti-Mendes Gde A, de Abreu MS, de Sousa AA. (2010) Clinical experience with a novel bovine collagen dura mater substitute. Asian J Neurosurg. 5(2): 31–4. [Crossref]
- Danish SF, Samdani A, Hanna A, Storm P, Sutton L. (2006) Experience with acellular humandura and bovine collagen matrix for duraplasty after posterior fossa decompression for Chiari malformations. Journal of neurosurgery. 104(1 Suppl): 16–20. [Crossref]
- Knopp U, Christmann F, Reusche E, Sepehrnia A. (2005) A new collagen biomatrix of equine origin versus a cadaveric dura graft for the repair of dural defects–a comparative animal experimental study. Acta neurochirurgica. 147(8): 877–87. [Crossref]
- Vakis A, Koutentakis D, Karabetsos D, Kalostos G. (2006) Use of polytetrafluoroethylene dural substitute as adhesionpreventive material during craniectomies. Clinical neurology and neurosurgery. 108(8): 798–802. [Crossref]
- Zahrai A, Shah J, Narotam P, M. G. (2005) A PROSPECTIVE CLINICAL STUDY OF THE USE OF COLLAGEN MATRIX AS A DURAL GRAFT IN SPINAL SURGERY. J Bone Joint Surg. 87: 295. [Crossref]
- Fushimi M, Sato K, Shimizu T, Hadeishi H. (2002) PLEDs in Creutzfeldt-Jakob disease following a cadaveric dural graft. Clinical neurophysiology: official journal of the International Federation of Clinical Neurophysiology. 113(7): 1030–5. [Crossref]
- Heath CA, Barker RA, Esmonde TF, Harvey P, Roberts R, Trend P, et al. (2006) Dura mater-associated Creutzfeldt-Jakob disease: experience from surveillance in the UK. Journal of neurology, neurosurgery, and psychiatry. 77(7): 880–2. [Crossref]
- Martinez-LageJF, Rabano A, Bermejo J, Martinez Perez M, Guerrero MC, Contreras MA, et al. (2005) Creutzfeldt-Jakob disease acquired via a dural graft: failure of therapy with quinacrine and chlorpromazine. Surgical neurology. 64(6): 542–5, discussion 5. [Crossref]
- Dongmei He, David G. Genecov, Morley Herbert, Raul Barcelo, Mohammed E. Elsalanty, Bradley E. Weprin, et al. (2010) Effect of recombinant human bone morphogenetic protein–2 on bone regeneration in large defects of the growing canine skull after dura mater replacement with a dura mater substitute. Journal of neurosurgery. 112(2): 319–28. [Crossref]
- Parlato C, di Nuzzo G, Luongo M, Parlato RS, Accardo M, Cuccurullo L, et al. (2011) Use of a collagen biomatrix (TissuDura) for dura repair: a long-term neuroradiological and neuropathological evaluation. Acta neurochirurgica. 153(1): 142–7. [Crossref]
- Ruediger Stendel, Marco Danne, Ingo Fiss, Ilse Klein, Andreas Schilling, Stefanie Hammersen, et al. (2008) Efficacy and safety of a collagen matrix for cranial and spinal dural reconstruction using different fixation techniques. Journal of neurosurgery. 109(2): 215–21. [Crossref]
- Narotam PK, Qiao F, Nathoo N. (2009) Collagen matrix duraplasty for posterior fossa surgery: evaluation of surgical technique in 52 adult patients. Clinical article. Journal of neurosurgery. 111(2): 380–6. [Crossref]
- Nakagawa S1, T. H. (2003) Postoperative infection after duraplasty with expanded polytetrafluoroethylene sheet. Neurol Med Chir (Tokyo). 43(3): 120–4. [Crossref]
- Than KD, Baird CJ, Olivi A. (2008) Polyethylene glycol hydrogel dural sealant may reduce incisional cerebrospinal fluid leak after posterior fossa surgery. Neurosurgery. 63(1 Suppl 1): ONS182–6; discussion ONS6-7. [Crossref]
- Hutter G, von Felten S, Sailer MH, Schulz M, Mariani L. (2014) Risk factors for postoperative CSF leakage after elective craniotomy and the efficacy of fleece-bound tissue sealing against dural suturing alone: a randomized controlled trial. Journal of neurosurgery. 121(3): 735–44. [Crossref]
- Neulen A, Gutenberg A, Takacs I, Weber G, Wegmann J, Schulz-Schaeffer W, et al. (2011) Evaluation of efficacy and biocompatibility of a novel semisynthetic collagen matrix as a dural onlay graft in a large animal model. Acta neurochirurgica. 153(11): 2241–50. [Crossref]
- Matsumoto Y, Aikawa H, Tsutsumi M, Narita S, Yoshida H, Etou H, et al. (2013) Histological examination of expanded polytetrafluoroethylene artificial dura mater at 14 years after craniotomy: case report. Neurol Med Chir (Tokyo). 53(1): 43–6. [Crossref]