DOI: 10.31038/IDT.2025613
Abstract
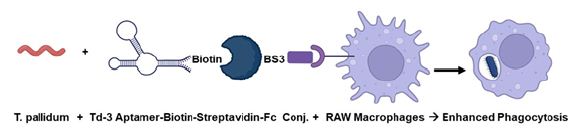
Extant anti-Treponema denticola DNA aptamers developed by Park et al. (2015) were evaluated for their ability to cross-react and bind live Treponema pallidum by fluorescence microscopy and spectrofluorometry. The highest affinity binder among the eight T. denticola aptamers designated Td-3 was biotinylated on its 3’ end and added in an equal molar ratio to a streptavidin-BS3 linker-murine IgG Fc fragment conjugate to emulate an antibody and used in in vitro T. pallidum plus murine RAW 264.7 macrophage co-culture experiments to determine if the indirect aptamer-Fc conjugate could enhance (opsonize) phagocytosis of T. pallidum compared to a bacteria alone co-culture. Preliminary results suggest that after only one hour of co-culture the Td-3 aptamer-Fc conjugate enhanced phagocytosis of T. pallidum about 7-fold compared to ~14-fold when the same experiment is conducted with a commercial murine anti-T. pallidum monoclonal antibody. Thus, although not yet optimized, the aptamer-Fc conjugates show promise as potential therapeutic opsonins in the battle against emerging antibiotic-resistant syphilis.
Keywords
Aptamer, Fc fragment, Macrophage, Opsonin, Phagocytosis, Syphilis
Introduction
The ancient scourge of sexually transmitted and congenital syphilis remains and is actually increasing in modern society with an estimated 8 million new cases per year in adults between 15 and 49 years of age [1-4]. Despite the continued relative efficacy of single dose injectable benzathine penicillin for adults [2,5], the combination of benzathine penicillin supply shortages [5], the lack of a vaccine [6], and increasing antibiotic resistance [1], has enabled Treponema pallidum to continue killing about 220,000 people worldwide per year including infants and adults [4].
The principle of rapid microbial evolution predicts that bacteria can eventually overcome all antibiotics. Thus, researchers have turned to alternative approaches involving passive immunity through mechanisms such as complement-mediated lysis (CML) and enhanced phagocytosis (opsonization) both of which are known to be at least somewhat effective against T. pallidum [7-14] to eventually treat emerging antibiotic-resistant syphilis. Unfortunately, development and production of humanized monoclonal antibodies for passive CML or opsonization therapy of syphilis or other bacterial diseases would be an expensive proposition. Thus, Bruno et al. previously explored the potentially much less expensive use of DNA aptamer-C1q and -Fc conjugates for CML [15,16] and opsonization [17] of pathogenic bacteria. In addition, the late Nobel prize laureate for PCR, Dr. Kary Mullis, developed a similar, but less direct, concept with alpha-gal sugar (galactose-α-1,3-galactose) conjugation to antibacterial aptamers (known as “alphamers”) to attract circulating anti-alpha-gal antibodies [18]. That work later led to the founding and continued alphamer work of Centauri Therapeutics Ltd. in the UK.
This history and therapeutic needs have brought us to the present report of our efforts to utilize eight extant aptamer DNA sequences developed by Park et al. [19] against a related species (T. denticola) in an effort to produce aptamer-Fc conjugates capable of both CML and opsonization of T. pallidum both in vitro and eventually in vivo. The following is a summary of our group’s initial progress to date relating in particular to the ability of one T. denticola aptamer (designated Td-3 in Table 1) to bind T. pallidum and when in conjunction with the Fc fragment of murine IgG to couple with Fc receptors on the surface of the murine macrophage cell line RAW 264.7 cells to significantly enhance phagocytosis by at least seven fold.
Table 1: T. denticola DNA aptamer sequences published by Park et al. [19].

Note: Yellow highlighted regions are identical or nearly identical between the Td-3, -6 and -8 sequences.
Materials and Methods
Treponema pallidum and Bacterial Culture
T. pallidum was purchased from American Type Culture Collection (ATCC; Manassas, VA) as Treponema phagedenis (ex Brumpt) Smibert, Kazan 8 strain (ATCC No. 27087) in lyophilized form. The freeze- dried culture was rehydrated in a 10 ml Oral Treponema Enrichment Broth (OTEB) tube from Anaerobe Systems Inc. (Morgan Hill, CA; Cat. No. AS-603) at 37°C in a slowly rotating closed incubator with a microaerophilic environment induced by Becton Dickinson Gas Pak™ pouches (Cat. No. 260680). T. pallidum cultures were capped and passaged weekly by 1: 10 dilution of the previous passage in OTEB.
Macrophage Cell Line and Culture
The RAW 264.7 murine macrophage cell line was purchased from ATCC and cultured in filter-sterilized RPMI-1640 medium complete with 10% heat-inactivated fetal bovine serum (FBS), L-glutamine, penicillin and streptomycin. RAW 264.7 macrophages were routinely grown to a near confluent monolayer in T-175 flasks and then scraped from the plastic substrate and one ml was resuspended in 20 ml of complete RPMI-1640. Macrophages were passaged at a 1: 20 ratio once or twice per week as needed in a 37°C humid incubator with 5% CO2 atmosphere.
T. denticola Aptamers, Monoclonal Antibody and Bacterial Binding Validation
Although we have an ongoing effort to develop our own T. pallidum aptamer DNA sequences to capture intellectual property in this domain, existing T. denticola aptamers published by Park et al. [19] where explored initially as potentially useful sequences that could accelerate our progress toward the ultimate goal of robustly opsonizing T. pallidum. Table 1 lists the eight published Park et al. T. denticola aptamer DNA sequences which were purchased from Integrated DNA Technologies (IDT; Coralville, IA) as 5’ Alexa Fluor 647 and 3’ biotinylated conjugates. In the case of actual phagocytosis experiments, we utilized a 3’-biotin-poly A 20mer spacer-Td-3 aptamer DNA oligonucleotide to provide extra distance between the Fc tail of IgG and the T. pallidum bacterial surface. All aptamer sequences were rehydrated in sterile phosphate buffered saline (PBS) without calcium or magnesium and used in fluorescence spectroscopy, microscopy or other experiments as discussed later. For comparison, an Invitrogen anti-T. pallidum murine monoclonal antibody (Cat. No. MA5-33297) was purchased from Thermo Fisher.
Aptamer binding was validated by spectrofluorometric analysis of aptamer-Alexa Fluor (AF) -647 staining and fluorescence microscopy as follows. One hundred µl of each AF-647-labeled Td aptamer at 100 nanoMolar concentration was added to 1 ml of ~ 106 T. pallidum (5 day culture in OTEB) in sterile PBS for 15 minutes at room temperature. The samples were then spun down for 5 minutes at 13,000 x g and the supernates was carefully siphoned out so as not to disturb the stained bacterial pellets which were the resuspended in 1 ml of fresh PBS. The 1 ml samples were then diluted to a total volume of 3 ml in PBS in polystyrene 1 cm path length cuvettes, capped, inverted several times and immediately analyzed using a Cary-Varian Eclipse™ spectrofluorometer with excitation at 650 nm and fluorescence emission scanning from 655 to 700 nm using a photomultiplier tube (PMT) setting of 1,000V. Samples were then placed in separate wells of a sterile 6 well plate and imaged using the red channel of an Invitrogen EVOS M5000 fluorescence microscope with 400X zoom setting.
Streptavidin-Fc Conjugation
To facilitate rapid exchange of the eight different T. denticola aptamers from Table 1, we chose to covalently conjugate streptavidin (SAv) from Thermo Fisher (Invitrogen Cat. No. 434301) with a murine IgG Fc fragment (Rockland Immunochemicals, Cat. No. 010-0103) using the bifunctional linker BS3 (bis(sulfosuccinimidyl)suberate) from Thermo Scientific (Cat. No. A39266. The coupling reaction was conducted with a 2: 1 molar ratio of Fc to streptavidin and 500X molar excess of BS3 at 4°C for 2 hours in sterile PBS buffer. For the electrophoretic band shift characterization, 10 to 20 µl samples of the components and conjugate in PAGE loading buffer were run on an Invitrogen Novex™ Value™ Tris-Glycine Mini 4-12% polyacrylamide gel at 105 V until the loading dye reach the bottom of the gel. The gel was then fixed and stained with an Invitrogen SilverQuest™ kit according the manufacturer’s instructions.
In Vitro Bacteria-Macrophage Co-Culture Phagocytosis Experiments
A total of 2 x 105 RAW 264.7 macrophages were inoculated into 2 ml of complete RPMI-1640 medium in each well of sterile 6-well polystyrene plates and incubated overnight at 37°C in a humid 5% CO2 atmosphere to allow macrophages to adhere and acclimate. The next day, ~106 T. pallidum bacteria were added in test wells. No bacteria, aptamers or conjugates were added to untreated control wells. Other control wells consisted of 3.6 nanomoles of poly-A20- Td-3 aptamer-3’-biotin or 3.6 nanomoles of streptavidin-Fc conjugate alone or in combination with 106 T. pallidum bacteria and the RAW 264.7 macrophages. The full experimental test wells contained poly- A20-Td-3 aptamer-3’-biotin and streptavidin-Fc conjugate (premixed in PBS at 3.6 nanomoles each) plus 106 bacteria and RAW 264.7 macrophages. In positive controls groups, 3.6 nanomoles of the commercial anti-T. pallidum monoclonal antibody were added. Plates were gently swirled and placed back in the incubator for 1 hour. Some wells were aspirated and gently washed with 2 ml of sterile PBS, aspirated, and covered with 2 ml of PBS and 2 µl of 10 mg/ml acridine orange (AO; Biotium Inc.) for 15 minutes at room temperature with gentle swirling. Some wells were fixed in 2 ml of pure methanol followed by aspiration and Wright’s staining using a kit from Carolina Biological Inc. AO-stained samples were aspirated again and covered with 2 ml of PBS prior to imaging at 400X zoom setting (approximately 200X total magnification) using the green channel of the EVOS M5000 fluorescence microscope. Wright’s stained wells were rinsed with 2 ml of deionized water and air dried followed by examination under a brightfield microscope using immersion oil and a 100X oil immersion objective.
Results
The first hurdle to overcome was consistent culture of the very oxygen-sensitive T. pallidum spirochetes which is not trivial [20]. Figure 1 (left panel) illustrates that commercially available OTEB tubes provide a reliable and convenient low oxygen tension environment in which to grow T. pallidum. Figure 1 (right panel) also illustrates that the 5-day cultures consisted predominately of spirochetes when viewed under phase-contrast microscopy at 400X magnification. However, we frequently saw the much larger, albeit lesser known, cyst-like forms of T. pallidum [21] as well, especially in cultures older than 5 days.
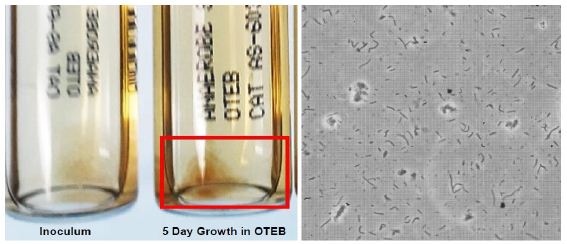
Figure 1: Left panel – OTEB anaerobic culture tubes showing an initial inoculum and clump of 5 day growth of T. pallidum. Right panel – Phase contrast image at 400X magnification of a mixture of spirochete and cyst forms of T. pallidum from the 5 day OTEB culture.
When tube stained with each of the eight individual Park et al. T. denticola aptamer-AF 647 conjugates from Table 1 and washed, it became clear that Td-3 was the strongest binder as shown in Figure 2 (red trace). Interestingly, Td-6 (purple trace) was the second strongest binder. As shown by the yellow highlights in Table 1, Td-3 and Td-6 only differ by 3 bases on the 5’ end of the aptamers. In addition, the Td-8 aptamer is highly homologous to both Td-3 and Td-6 (differs by only 4 bases on the 5’ end as seen in Table 1) and it ranked approximately fourth (gray trace) according to the spectrofluorometric data in Figure 2. So, there appeared to be some sequence-dependent nature to the affinity ranking as theoretically expected. With Td-3 appearing to be the clear winner of the binding competition, we focused on it for fluorescence microscopy validation studies.
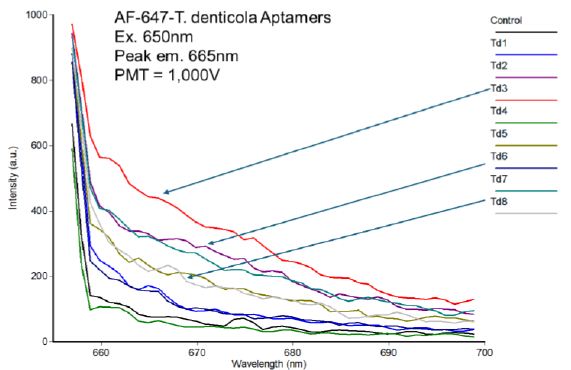
Figure 2: Spectrofluorometric results comparing the binding of the eight different Park et al. T. denticola aptamers from Table 1 labeled with Alexa Fluor (AF)-647 on their 5’ ends to T. pallidum. Excitation was at 650 nm with the PMT setting at 1,000 Volts.
As seen in Figure 3, Td-3 showed relatively strong fluorescence from the AF-647 dye tag especially when bound to the large cyst-like clumps vs. much fainter individual spirochetes in the background of Figure 3 (right panel). Figure 3 validated the spectrofluorometric binding analysis and we chose to proceed with the Td-3 aptamer in subsequent in vitro phagocytosis experiments.
Figure 3: Left panel – 400X magnified phase contrast image of T. pallidum again showing prominent cyst forms as well as spirochetes. In the right panel, the intensely stained red-fluorescing cyst forms dominate the fluorescence microscopy image, although some fainter spirochete aptamer-AF 647 staining can be seen in the background.
Still, to leave our options flexible for facile exchange of any of the eight Park et al. T. denticola aptamers, we chose to make streptavidin- BS3 linker-murine Fc conjugates that could be easily bind to 3’ or 5’ biotinylated aptamers versus a covalent aptamer-Fc conjugate for initial experiments. Figure 4 depicts successful BS3 conjugation of streptavidin (SAv) to the Fc tail of murine IgG in a silver-stained polyacrylamide electrophoresis gel. The red arrow in Figure 4 shows a band shift to a higher molecular weight band in the conjugate that does not appear in either the Fc or SAv lanes indicating some level of conjugation. The conjugation can likely be optimized to give a greater percent yield, but it appeared sufficient for us to consider conducting initial phagocytosis enhancement studies.

Figure 4: Silver stained 4-12% polyacrylamide electrophoresis gel showing a band shift (red arrow) for the streptavidin (SAv)-BS3-Fc conjugate to a higher molecular weight band than is seen in either of the component (SAv or Fc) lanes indicating some covalent conjugate formation.
Figure 5 suggests that when studied by AO staining and fluorescence microscopy, there appeared to be noticeably more internalized “blobs” or bacterial fragments in the RAW 264.7 plus bacteria and aptamer-Fc conjugate group (some of which are pointed to by yellow arrows) than in the untreated and macrophage plus bacteria only groups. Figure 6 represents a further magnified version of Figure 5C to better enable visualization of the internalized bacterial fragments. One must consider that these co-cultures were washed twice with PBS after 1 hour of bacterial exposure which probably removed most intact spirochetes bound to the macrophage surfaces, thus no spirochetes are seen in Figures 5 and 6. However, internalized bacterial fragments would remain in the cytoplasm and be stained by AO after washing.
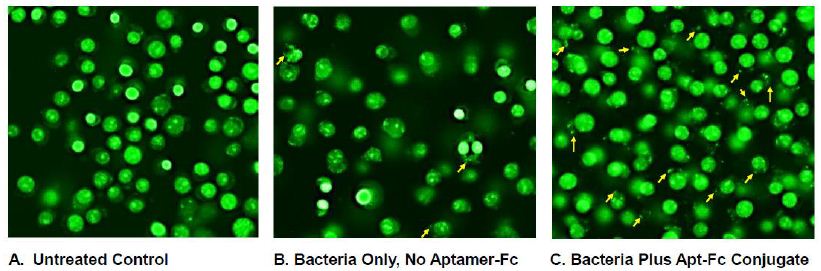
Figure 5: Acridine Orange (AO)-stained comparison of (A) untreated RAW 264.7 macrophages with (B) T. pallidum only co-culture with RAW 264.7 cells and (C) T. pallidum bacteria plus the aptamer-biotin-SAv-BS3-Fc conjugate in the presence of RAW 264.7 macrophages showing enhanced phagocytosis and internalization of T. pallidum bacterial fragments (yellow arrows).
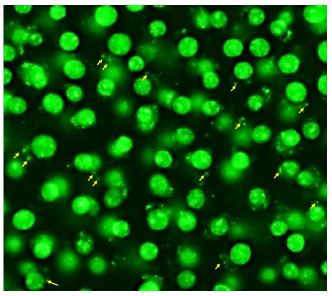
Figure 6: Further magnified image of Figure 5C showing details of the internalized bacterial fragments pointed to by yellow arrows.
A similar visual trend in aptamer-Fc conjugate-enhanced phagocytosis was noted for the Wright’s stained populations as illustrated in Figures 7 and 8. In Figure 7A, no internal blobs are seen in the macrophage cytoplasm of untreated controls presumably because no bacteria were present and the macrophages do not appear perturbed. However, a few internalized bacterial fragments appear in the cytoplasm of macrophages exposed to bacteria alone for one hour as seen in Figure 7B. The number of internalized fragments or blobs appears to increase in Figure 7C in the presence of the aptamer-Fc conjugate (pointed to by red arrows). Figure 8 illustrates some of the more extreme cases of phagocytosis of T. pallidum in the presence of the aptamer-Fc conjugate especially in multinucleated fused giant cells. In addition, macrophages in Figures 7B, 7C and 8 appear “angry” and foamy in the presence of T. pallidum bacteria versus the untreated control cells in Figure 7A.
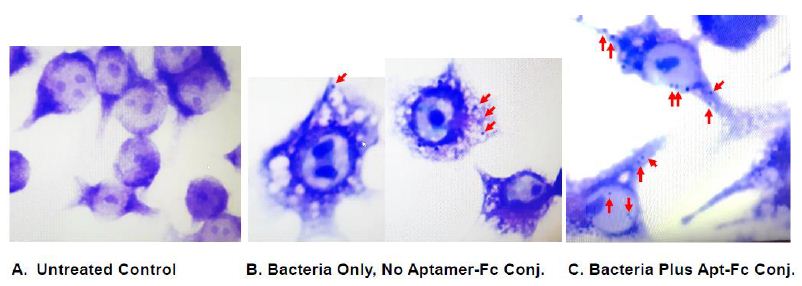
Figure 7: Wright’s stained comparison of (A) untreated RAW 264.7 macrophages with (B) T. pallidum only co-culture with RAW 264.7 cells and (C) T. pallidum bacteria plus the aptamer-biotin- SAv-BS3-Fc conjugate in the presence of RAW 264.7 macrophages showing enhanced phagocytosis and internalization of T. pallidum bacterial fragments (red arrows).
Figure 8: Two examples of extreme phagocytosis by multinucleated fused giant RAW 264.7 cells that engulfed numerous T. pallidum bacteria (red arrows) in the presence of the Td-3 aptamer-Fc conjugate after one hour in culture.
The commercial anti-T. pallidum monoclonal antibody appeared to produce the most avid phagocytosis as shown by a few examples in Figure 9. Figure 10 is a graph that quantitatively compares the average numbers of internalized bacteria or their fragments counted by the authors per macrophage with a minimum of 200 macrophages counted per treatment group. If the monoclonal antibody is used as the gold standard for opsonization of T. pallidum, then we have only achieved about 50% efficiency with the aptamer-Fc conjugate to this point in our development process (i.e., on average 7.24 bacteria or fragments per phagocyte due to the aptamer-Fc conjugate vs. 14.21 with the monoclonal antibody). However again, these are initial results from a system that has not yet been truly optimized.
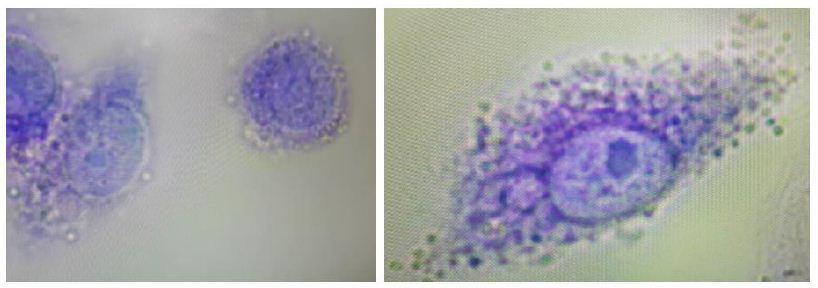
Figure 9: Two examples of extreme phagocytosis by RAW 264.7 macrophages that engulfed numerous T. pallidum bacteria in the presence of the commercial anti-T. pallidum murine monoclonal antibody after one hour in culture.
Figure 10: Graphical comparison of mean numbers of phagocytized T. pallidum or their fragments per macrophage between the various control and treatment groups. A minimum of 200 RAW 264.7 macrophages were counted per group and that number of cells was used as the denominator to divide the total number of bacteria or bacterial fragments counted inside the phagocytes for each Wright’s stained group.
Discussion
The present report extends the first author’s previous publication on aptamer-Fc conjugates to opsonize the poly-D-glutamic acid capsule of vegetative Bacillus anthracis using capsule-coated magnetic microbeads as the phagocytic targets [17], except that this time, real bacteria were ingested as evidenced by stained internalized bacterial fragments after buffer washing of the macrophage surface. Clearly, the data suggest some degree of successful opsonization when one compares the bacteria alone treatment groups with the effects of the addition of biotinylated aptamers or SAv-Fc conjugate alone with the full test group (i.e., macrophages plus bacteria and aptamer-biotin-SAv-Fc conjugate) with an approximate 7-fold increase in phagocytosis. It is also clear by comparison to the monoclonal antibody-opsonized group results that the enhanced phagocytosis could be optimized and perhaps at least doubled to an average 14 bacteria or bacterial fragments per macrophage (Figure 10). Aspects that can be optimized are: 1) the percent yield of the SAv-Fc conjugate (apparent from incomplete conjugation in Figure 4) or the use of direct aptamer-3’-Fc covalent conjugate, 2) the amount of aptamer-Fc conjugate used per test, 3) the incubation time and ratio of bacteria to phagocytes and 4) generation of even higher affinity T. pallidum aptamer DNA sequences which we are currently developing. Still, this is a promising preliminary report suggesting a new future class of aptamer-3’-Fc conjugates to emulate monoclonal antibodies and perhaps lower the cost of future passive immunity therapy for syphilis. For use in vivo especially, attachment of the Fc to the 3’ end will be key to extending aptamer conjugate pharmacokinetics by slowing kidney clearance and inhibiting serum exonuclease degradation of the aptamer moiety of the conjugate [22-24].
Conclusions
Preliminary data suggesting seven-fold opsonization of live T. pallidum bacteria by an indirect aptamer-Fc conjugate using an aptamer developed against a close relative (T. denticola) were presented. While promising as future passive immunity therapeutics, such aptamer-Fc simulants of monoclonal antibodies can obviously be further optimized for greater enhancement of phagocytosis.
Acknowledgments
Funding was provided by US NIH (NIAID) SBIR Contract No. 75N93025C00010. The authors thank Eroica Luckowski of Austin Community College’s Biotechnology Department in Round Rock, TX for assistance with cell culture and related phagocytosis experiments and microscopy.
References
- Orbe-Orihuela YC, Sánchez-Alemán M (2022) Syphilis as Re-Emerging Disease, Antibiotic Resistance, and Vulnerable Population: Global Systematic Review and Meta-Analysis 11.
- Peeling RW, Mabey D, Kamb ML, Chen, -S., Radolf JD, et al. (2017) Syphilis. Nature Reviews Disease Primers 3: 17073.
- Stamm LV (2016) Syphilis: Re-emergence of an old Microbial cell (Graz, Austria) 3: 363-370.
- Ramchandani MS, Cannon CA, Marra CM (2023) Syphilis: A Modern Infectious disease clinics of North America 37: 195-222. [crossref]
- Seghers F, Taylor MM (2024) Securing the supply of benzathine penicillin: a global perspective on risks and mitigation strategies to prevent future shortages. Int Health 16: 279-282. [crossref]
- Avila-Nieto C, Pedreno-Lopez N, Mitja O, Clotet B, Blanco J, et (2023) Syphilis vaccine: challenges, controversies and opportunities. Frontiers in immunology 14: 1126170. [crossref]
- Fitzgerald TJ (1987) Activation of the classical and alternative pathways of complement by Treponema pallidum subsp. pallidum and Treponema vincentii. Infect Immun 55: 2066-2073. [crossref]
- Yi DY, Xu QY, He , Zheng XQ, Yang TC (2024) Treponema pallidum protein Tp47 induced prostaglandin E2 to inhibit the phagocytosis in human macrophages. J Eur Acad Dermatol Venereol 38: 1166-1178. [crossref]
- Chen H, Tong M-L, Liu L-L, Lin L-R, Yang T-C (2022) The whole process of macrophage–Treponema pallidum interactions: Opsonic phagocytosis, nonopsonic phagocytosis and active invasion. International Immunopharmacology 107: 108657. [crossref]
- Hawley K, Cruz A, Benjamin S, Vake C, Cervantes J, et al. (2017) IFNγ Enhances CD64-Potentiated Phagocytosis of Treponema pallidum Opsonized with Human Syphilitic Serum by Human Frontiers in immunology 8: 1227. [crossref]
- Marangoni A, Aldini R, Guardigli M, Sambri V, Giacani L, et (2003) Phagocytosis of Treponema pallidum and reactive oxygen species production by isolated rat Kupffer cells. Medical Microbiology and Immunology 192: 183-188. [crossref]
- Lukehart SA, Shaffer JM, Baker-Zander SA (1992) A subpopulation of Treponema pallidum is resistant to phagocytosis: possible mechanism of The Journal of infectious diseases 166: 1449-1453. [crossref]
- Alder JD, Friess L, Tengowski M, Schell RF (1990) Phagocytosis of opsonized Treponema pallidum subsp. pallidum proceeds slowly. Infect Immun 58: 1167-1173. [crossref]
- COCKAYNE A, PENN CW, BAILEY MJ (1986) Surface Properties of Treponema pallidum in Relation to Phagocytosis by Human Polymorphonuclear Leucocytes in vitro. Microbiology 132: 133-141. [crossref]
- Bruno JG, Carrillo MP, Phillips T (2008) In vitro antibacterial effects of antilipopolysaccharide DNA aptamer-C1qrs complexes. Folia microbiologica 53: 295-302. [crossref]
- Bruno JG (2013) A review of therapeutic aptamer conjugates with emphasis on new Pharmaceuticals 6: 340-357. [crossref]
- Bruno JG, Carrillo MP, Crowell R (2009) Preliminary development of DNA aptamer- Fc conjugate Journal of biomedical materials research. Part A 90: 1152-1161. [crossref]
- Kristian SA, Hwang JH, Hall B, Leire E, Iacomini J, et (2015) Retargeting pre- existing human antibodies to a bacterial pathogen with an alpha-Gal conjugated aptamer. Journal of molecular medicine 93: 619-631. [crossref]
- Park JP, Shin HJ, Park SG, Oh HK, Choi CH, et (2015) Screening and development of DNA aptamers specific to several oral pathogens. Journal of microbiology and biotechnology 25: 393-398. [crossref]
- Cox DL (1994) Culture of Treponema pallidum. In Methods in Enzymology 63: 390-405.
- Arrazuria R, Caddey B, Cobo ER, Barkema HW, De Buck J (2021) Effects of different culture media on growth of Treponema isolated from digital dermatitis. Anaerobe 69: 102345. [crossref]
- Dougan H, Lyster DM, Vo CV, Stafford A, Weitz JI, et (2000) Extending the lifetime of anticoagulant oligodeoxynucleotide aptamers in blood. Nuclear medicine and biology 27: 289-297. [crossref]
- Bruno, G Crowell R (2008) Selective glutaraldehyde-mediated coupling of proteins to the 3’-adenine terminus of polymerase chain reaction products. Journal of biomolecular techniques: JBT 19: 177-183. [crossref]
- Keefe AD, PS Ellington A (2010) Aptamers as Nat Rev Drug Discov 9: 537-550.