Abstract
A wild-caught adult female Chacma baboon (Papio ursinus) developed weight loss, general weakness, and anorexia. Clinical examination revealed numerous firm subcutaneous nodules and generalized lymphadenomegaly. Biopsies were taken for histopathology and sections were processed with immunoperoxidase staining for CD3 (T-cell) and CD20 (B-cell) markers. There was strong and widespread positive staining for CD3. Based on the clinical and histopathologic findings, and the positive immunocytochemical results, the diagnosis was T-cell lymphoma and the animal was euthanized. Although the type of T-cell was not established, this case resembled human T-cell lymphoma (HTLV) with similarities in the clinical onset and histopathology resembling simian T-cell (STLV) as previously reported in two Vervet monkeys from the same facility.
Keywords
leukaemia, human lymphoma, T-cell lymphoma, Primate T-lymphotropic virus, Simian T-lymphotropic virus, non-human primates, Chacma baboon
Introduction
Human T-cell lymphotropic virus (HTLV) has been implicated in several diseases. Four HTLV sub-types have been identified. HTLV-1, a C-type retrovirus, and associated with adult T-cell leukaemia/lymphoma (ATL) [1], is characterized by the malignant proliferation of CD4 + T lymphocytes. The symptoms include hypercalcemia, lymphadenopathy, subcutaneous nodules [2] and the involvement of the liver or the spleen [3]. The HTLV-1, human T-cell lymphotropic virus type II (HTLV-II) and simian T-cell lymphotropic virus type I (STLV-1) belong to a group of viruses that have been classified as primate T-cell lymphotropic virus type I (PTLV-1) [4].
Antibodies against HTLV antigens have been found in several Old World primate species such as Japanese macaques [5], rhesus macaques, crab-eating macaques [6] and baboons [7]. Miyoshi [8] reported 10-50% of macaque species are seropositive for HTLV. Additionally, antibodies against HTLV-1 were also described in an African green monkey (Vervet monkey) and baboons [9, 10]. Several reports indicate that many, but not all, troops of Old World primates in Asia and Africa, both in the wild and in captivity, have antibodies to HTLV-1. There is evidence of transmission of HTLV-1 between human and non-human primates [9] and cross-species transmission among primates such as between Vervet monkeys and Chacma baboons [10].
Simian T-cell lymphotropic virus (STLV) is a C-type member of the coronavirus subgroup of retroviruses and was identified shortly after the initial isolation of HTLV-1 [11]. As the case with HTLV-1, many Old World primates from Asia and Africa also harbour STLV-1 that is closely related to HTLV-1 [12]. STLV-1 can cause an ATL-like pathology in infected monkeys. The former was found in several wild-caught baboon species [12, 13, 14] and Vervet monkeys [4]. One study reported an infection rate of 40% in a captive baboon population from the Southwest Foundation for Biomedical Research (SFBR) [12]. Furthermore, cross-species transmission of STLV-1 from wild-caught Chacma baboons and Olive baboons have been reported [14] as has interspecies transmission of Rhesus macaques to Hamadrya baboons after an outbreak of malignant lymphoma in the baboons of Sukhumi Primate Centre [15]. The author suggested the possibility that the transmission episodes occurred after capture and it remains unclear whether natural STLV-1 isolates exist in the wild. Most studies in New World primates have failed to find STLV.
Whilst some infected animals remain in a stage of latency and asymptomatic, there are some reports of malignant lymphoma cases in non-human primates such as baboons [11, 16] and Vervet monkeys [17, 15, 9, 18]. The Sukhumi outbreak was the largest outbreak of a malignant disease described in non-human primates [16]. By 1992, more than 300 baboons had died of malignant lymphoma and in a later investigation by Voevodin [15] [20], it was suggested that the Rhesus type STLV-1 was the most likely etiologic agent of this malignancy. The intrinsic need for the virus to replicate through clonal expansion of lymphocytes, and the tax gene’s effects on cellular transcriptional regulation, presumably accounts for its lymphomagenic potential [13]. Malignant lymphomas in non-human primates are remarkably similar to non-Hodgkin’s lymphomas (NHLs) found in humans. The most common findings are weight loss, overall weakness, and dyspnea and generalized lymphadenopathy [13, 19].
We previously reported on T-cell lymphoma from two Vervet monkeys [20] and since then, four additional cases have emerged. This report describes T-cell lymphoma in a Chacma baboon with comparable symptoms and pathology previously described and reported in the two Vervet monkeys from the same holding facility.
Humane Care Guidelines
This 16-year-old wild-caught female Chacma baboon was a member of a colony of ninety individuals maintained outdoors at the Delft Animal Centre of the Medical Research Council, Cape Town, South Africa. Individuals were housed in single cages with full visual, olfactory and auditory contact with conspecifics. Grooming with adjacent individuals was possible through wire mesh grooming panels.
The animals were maintained on a standard diet of special monkey cubes (Equifeeds, Cape Town, South Africa) and supplemented with seasonal fruit and vegetables. Additional enrichment devices were provided daily and water was available ad libitum. All animals were housed and maintained in accordance with the South African National Standard for the Care and Use of Animals for Scientific Purposes (The SANS 10386: 2008). The use of wild-caught baboons for scientific purposes has since been discontinued.
Case history
The clinical signs presented were a general weakness, loss of appetite and weight loss. Physical examination revealed conspicuous enlargements of several superficial lymph nodes in the right pre-scapular, sub-mandibular and bilateral inguinal areas. Additionally, there appeared to be a focal area of superficial skin ulceration on the thorax. A biopsy was taken under Ketamine (Biopharm, Cape Town, South Africa) at 10mg/kg bodyweight from the right inguinal lymph node for histopathology. The sections were prepared for immunoperoxidase staining with CD3 (T-cell) and CD20 (B-cell) markers. There was strong and widespread positive staining for CD3 and limited positive staining for CD20. T-cell lymphoma was diagnosed and the animal was euthanized.
Macroscopic observations during necropsy
The carcass was moderately emaciated. There was marked enlargement of the following lymph nodes, with some presenting focal areas of necrosis: left and right axillary, left and right inguinal, left and right renal, left and right retropharyngeal, and several intra-abdominal lymph nodes as well as nodes embedded along the thoracic aorta (Figure 1). Enlarged intra-thoracic lymph nodes caused compression of the adjacent vertebral bodies (T2 and T11), with possible infiltration of lymphocytes (Figure 2). The spleen contained prominent lymphoid follicles and the liver was friable with an uneven surface. The left kidney was moderately enlarged and pale as a result of nephrosis, with multifocal retention cysts in the cortex, and the right kidney showed focal subacute to chronic infarction.
Histopathology
For histopathological examinations, tissue samples containing lesions were fixed in 10% neutral buffered formalin for 24 hours. After tissue dehydration, paraffin embedding (Paraplast Plus, Monoject Scientific Inc.), 4-micron tissue sections were stained with hematoxylin and eosin. All sections were evaluated under a light microscope using 10x and 40x objective.
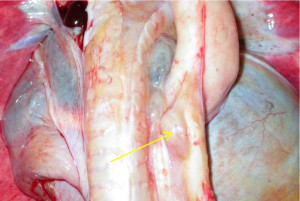
Figure 1. Intra-thoracic lymph nodes (arrow) embedded along the thoracic aorta
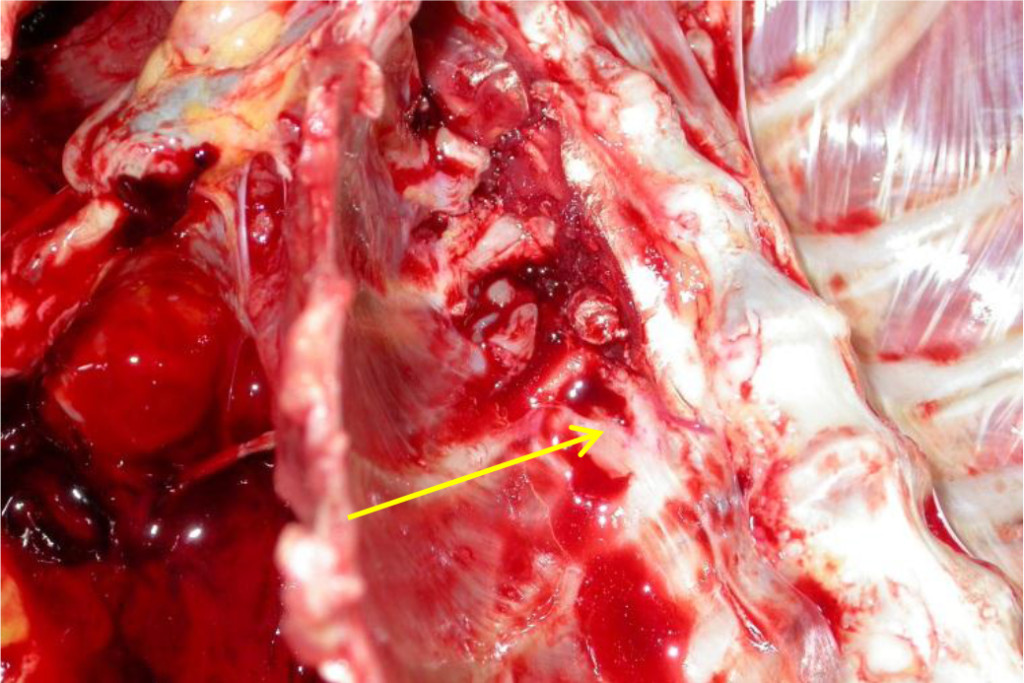
Figure 2. Vertebral infiltrations of intra-thoracic lymph node
The normal architecture of the lymph nodes (Figure 3a), tonsils and lymphoid tissues of the spleen was effaced as a result of the infiltration of sheets of medium to large lymphocytes and lymphoblasts (Figure 3b), sometimes arranged in distinct follicles. Numerous mitotic figures were present and multifocal areas of necrosis were visible with infiltration of mild to moderate numbers of neutrophils. The necrotic foci were surrounded by a thin layer of macrophages. Lesions in the other organs reflected the macroscopic findings.
Immunohistochemical analysis of neoplastic lymph nodes was performed using antibodies to CD3 and CD20 antigens. The positive staining for CD3 confirmed the biopsy findings reported earlier (Figure 3c).
Discussion
In this case study of an adult female Chacma baboon, common symptoms such as weight loss, generalized lymphadenopathy, fatigue, and dyspnea were reported. The onset of these clinical symptoms is in agreement with our previous findings on the two Vervet monkeys [20] and other reports of lymphoma in non-human primates [13, 17, 2].
Visceral lymph nodes were noted with an accumulation of neoplastic lymphocytes in multiple organs. As previously reported for the Vervet monkeys, this individual also presented an enlarged liver and spleen. Histological examination revealed infiltration of lymph nodes with sheets of neoplastic lymphocytes. These lymph nodes had a homogeneous, white coloured, fleshy appearance with some hemorrhagic or necrotic foci. Neoplastic cells were pleomorphic, with convoluted or lobulated nuclei, and there was bone marrow involvement. These findings are similar to those described in humans. HTLV-1 is characterized by dermal neoplastic involvement, hepatomegaly, lytic bone lesions with associated hypercalcemia, and enlarged neoplastic lymph nodes with infiltration of neoplastic cells [2].
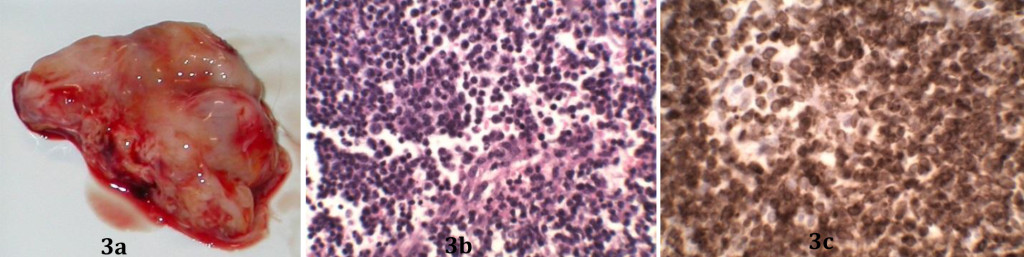
Figure 3. (a) Inguinal lymph node mass (b) The lesions are composed of small lymphoid cells. (c) Most neoplastic cells stain positively for CD3.
Infection rates of <25% with HTLV-1 have been previously reported in Chacma baboons and African green monkeys [10]. Likewise, for STLV-1, 29% of wild Vervet and 33% of wild baboons in South Africa are also seropositive [9]. Higher rates are seen in females than males and there is a gradual increase in seroprevalence with age. A relatively low prevalence of infection is found in younger individuals [13, 10] . Furthermore, non-human primates are usually diagnosed late in their illness, so lymphocytosis of frank leukemia may already be present that would grade the majority of these animals as either suffering from chronic or acute forms of lymphosarcoma.
In both HTLV and STLV age and gender plays a critical role. Higher rates of HTLV infection were seen in adult females than males and in Japanese macaques, more than 70% were reported to be seropositive for HTLV [6]. In the case of STLV, it was found that the infection rate approached 80% in an aged baboon population at the Southwest Foundation [12].
As opposed to endogenous retroviruses, which are spread vertically in the germline, HTLV-1 is an exogenous retrovirus which spreads horizontally. Therefore infection and consequently the incidence of antibodies can be expected to increase with age [12, 9] and sexual behaviour have been reported to be the important factor in establishing the prevalence of infection [10]. With reference to our previous report on the Vervet monkeys, we speculated sexual transmission to be the cause of infection as these animals formed part of a breeding program [20]. An alternative source reported, is interspecies transmission in the wild [9, 10] [3, 4] and in captivity [14, 15, 12] and data suggest that STLV-1 transmission only occurs after capture [14]. However, the scenario of transmission in captivity might not be applicable to this case as this female baboon was never utilized as a breeding animal in captivity.
Conclusion
The adult female Chacma baboon described in this report was diagnosed with T-Cell lymphoma resembling human T-cell lymphoma (HTLV). Even though the type of T-cell was not determined, the prevalence of seropositive STLV reported in wild populations of African non-human primates, such as the Chama baboon, renders the possibility that this individual was naturally infected with STLV in the wild.
Primary screening and surveillance tests are essential to identify individuals infected with simian retroviruses. Management practices in non-human primate breeding colonies need to take into account the possible presence of STLV-1 and aim to reduce transmission by preventing sexual contact between positive and negative animals. Lymphoma has to be considered as one of the more common causes of wasting and death in non-human primates used in biomedical research.
Acknowledgements: We thank Dr. Tertius Gous for conducting the necropsy, histopathological examinations and immunohistochemistry.
Declaration of Conflicting Interests: The author declares no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author received no financial support for the research, authorship, and/or publication of this article.
References
- Poiesz BJ, Ruscetti FW, Gazdar AF, Bunn PA, Minna JD, et al. (1980) Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc Natl Acad Sci U S A 77: 7415–7419. [crossref]
- Jayo MJ, Laber-Laird K, Bullock BC, et al. (1990) T-cell Lymphosarcoma in a female African Green monkey (Cercopithecus aethiops). Lab Anim Sci 40: 37–41.
- Stewart SA, Poon B and Chen ISY (1994) Mechanisms of HTLV leukaemogenisis. In “Viruses and Cancer”. A. Minson, J. Neil and M. McCrae (eds), Cambridge Univ. Press, Cambridge, UK. 189–212.
- Engelbrecht S, van Rensburg EJ and Robson BAN(1996) Sequence variation and subtyping of Human and Simian T-cell Lymphotropic Virus Type I Strains from South Africa. J Acq Im Def Syn Hum Retro 12: 298–302.
- Miyoshi I, Yoshimoto S, Fujishita M, Taguchi H, Kubonishi I, et al. (1982) Natural adult T-cell leukemia virus infection in Japanese monkeys. Lancet 2: 658. [crossref]
- Miyoshi I, Fujishita M, Taguchi H, Matsubayashi K, Miwa N, et al. (1983) Natural infection in non-human primates with adult T-cell leukemia virus or a closely related agent. Int J Cancer 32: 333–336. [crossref]
- Guo GG, Wong-Staal F and Gallo RC (1984) Novel viral sequences related to human T-cell leukaemia virus in T-cells of a seropositive baboon. Science 223: 1195–1196.
- Miyoshi I, Fujishita M, Taguchi H, Matsubayashi K, Miwa N, et al. (1983) Natural infection in non-human primates with adult T-cell leukemia virus or a closely related agent. Int J Cancer 32: 333–336. [crossref]
- Becker WB, Becker MLB, Homma T, et al (1985) Serum antibodies to human T-cell leukaemia virus type I in different ethnic groups and in non-human primates in South Africa. S Afr Med J 67: 445–449.
- Botha MC, Jones M, de Klerk WA and Yamamoto N (1985) Spread and distribution of human T-cell leukaemia virus type I-reactive antibody among baboon and monkeys in the northern and eastern Transvaal. SAMJ 67: 665–668.
- Lerche NW (2005) Common Viral Infections of Laboratory Primates. In “The Laboratory Primate” Sonia Wolfe-Coote (ed), Elsevier Academic Press, Amsterdam 78–79.
- Moné J, Whitehead E, Leland M, Hubbard G, Allan JS (1992) Simian T-cell leukemia virus type I infection in captive baboons. AIDS Res Hum Retroviruses 8: 1653–1661. [crossref]
- Allan JS, Leland M, Broussard S, et al. (2001) Simian T-cell lymphotropic viruses (STLVs) and lymphomas in African non-human Primates. Cancer Inves 19: 383–395.
- Mahieux R, Pecon-Slattery J, Chen GM and Gessain A (1998) Evolutionary inference of novel Simian T Lymphotropic virus type I from wild-caught Chacma baboons (Papio ursinus) and Olive baboons (Papio anubis). Virology 251: 71–84.
- Voevodin A, Samilchuk E, Schätzl H, et al. (1996) Interspecies transmission of Macaque Simian T-cell leukaemia/lymphoma virus type I in baboons resulted in an outbreak of malignant lymphoma. J Virol 70: 1633–1639.
- Lapin B (1988) Baboon lymphoma viruses. In “Virus Diseases in Laboratory and Captive Animals. Darai g. (ed). Nijhoff Publishers, Boston. 135–151.
- Sato Y, Matsuura S, Kadota K and Miyazawa I (1999) T-cell lymphoma in a Savanna monkey (Cercopethicus aethiops) probably related to Simian T-cell leukaemia virus infection. J Vet Med. Sci 61: 49–52.
- Tsujimoto H, Noda Y, Ishikawa K, et al. (1987) Development of adult T-cell leukaemia-like disease in African green monkey associated with clonal integration of simian T-cell leukaemia virus type I. Cancer Res 47: 269–274.
- Hubbard GB, Mone JP, Allan JS, et al. (1993) Spontaneously generated non-Hodgkin’s lymphoma in twenty-seven simian T-cell leukaemia virus type I antibody-positive baboons (Papio species). Lab Anim Sci 43: 301–309.
- de Villiers C, Seier JV (2011) Weight loss, weakness and lymphadenopathy in two vervet monkeys (Chlorocebus spp.). Lab Anim (NY) 40: 177–178, 180–2. [crossref]