Abstract
Background: Intra-atrial thrombi, though rare, can develop in either atrium, with a higher prevalence in the left atrium. The causes are often difficult to determine, leading many cases to be classified as having uncertain origins.
Case Presentation: We present the case of a 35-year-old non-smoking male with a history of deep vein thrombosis who experienced worsening shortness of breath and pleuritic chest pain. Echocardiography revealed a right atrial thrombus extending into the left atrium through a patent foramen ovale. The thrombus was surgically excised; however, the patient developed postoperative complications, including pericardial effusion, pulmonary embolism, and acute liver injury. Following treatment and close monitoring, his condition stabilized, and he was discharged on anticoagulation therapy. Diagnosing intra-atrial thrombi presents significant challenges due to a broad differential diagnosis. Accurate assessment often requires multimodal imaging techniques, including echocardiography, CT angiography, and PET scans. Treatment options include anticoagulation, thrombolysis, and surgical thrombectomy, but there is no universal consensus on the optimal management approach.
Conclusion: Therefore, early detection of intra-atrial thrombi, timely surgical intervention when indicated, and vigilant postoperative monitoring with continued anticoagulation are essential for improving patient outcomes. Consequently, further research is necessary to elucidate the underlying etiologies and enhance diagnostic and therapeutic strategies for this rare condition.
Introduction
Right atrial thrombi (RAT) are blood clots located within the heart’s right atrium. Unlike left heart thrombi, right heart thrombi may originate from two different sources: they may develop within the right heart chambers (autochthonous clots) or represent peripheral venous clots that, en route to the lungs, accidentally lodge in a patent foramen ovale, tricuspid chordae, or elsewhere (transferred clots) [1,2].
This condition can be caused by several underlying factors, such as atrial fibrillation, deep vein thrombosis, and the presence of foreign objects like pacemaker leads. Consequently, RAT poses a significant risk of severe morbidity, including pulmonary embolism and stroke, as the thrombus may pass through a patent foramen ovale (PFO) and enter other parts of the circulatory system. Furthermore, To classify right heart thrombi, the European Working Group on Echocardiography identifies three types. Type A – RAT in Transit are large, free-floating clots from the deep venous system passing through the right atrium, posing a high risk of pulmonary embolism. Type B – RAT in Situ are smaller clots that attach to the right atrial wall or intracardiac devices and form within the atrium. Type C – Mobile In Situ Thrombi have a stalk-like structure with a thin attachment to the atrial wall, resembling an atrial myxoma. In clinical settings, RAT can present with chest pain, dyspnea, and palpitations. Additionally, some patients may exhibit features of right heart failure, including peripheral edema and jugular venous distension [1-4].
The literature indicates that right heart thrombi, though rare, are clinically significant. Numerous case reports have documented these cases, and a high proportion of patients have succumbed to embolic complications. In this context, we present the case of a 35-year-old non-smoking male who presented with concerning respiratory and cardiac symptoms. Imaging revealed a right atrial thrombus extending through a patent foramen ovale. Following surgical removal of the thrombus, the patient developed significant postoperative complications, including pericardial effusion and pulmonary embolism, necessitating further interventions. The patient’s condition stabilized with treatment, and he was discharged with instructions for continued anticoagulation therapy and regular follow-up.
Case Presentation
A 35-year-old non-smoking male presented to the emergency department with a three-day history of worsening shortness of breath and pleuritic chest pain. His symptoms were aggravated by respiration but did not change with body position. Additionally, he reported post-exertion palpitations, which resolved spontaneously. Two weeks prior, the patient experienced constitutional symptoms, including headache, chills, joint pain, low-grade fever, and a non-productive cough suggestive of an upper respiratory tract infection. During this period, he also noted a significant decrease in physical activity, easy fatigability, and generalized weakness. Notably, the patient had a past medical history of deep vein thrombosis (DVT) in the right leg but was otherwise healthy, with a history of tonsillectomy as the only surgical intervention.
Following evaluation, the patient presented with critically low oxygen saturation of 75% on pulse oximetry, necessitating immediate hospital referral. Upon admission, oxygen support was initiated, resulting in an improvement in oxygen saturation to 90%. A comprehensive cardiovascular examination revealed no signs of DVT, peripheral edema, or clubbing. Chest X-ray findings indicated clear lung fields and the electrocardiogram (ECG) showed sinus rhythm without ischemic changes. A transthoracic echocardiogram revealed a distinct cylindrical mass originating from the RA and extending through a PFO into the Left atrium (LA), with attachment to the right atrial septum. The mobile edges of this structure raised suspicion for either myxoma or thrombus (Video 1).
Given the echocardiographic findings, the patient was prepared for the surgical excision of the right atrial mass. Intraoperatively, it was discovered that the cylindrical mass in the RA extended into the LA through a PFO (see Figure 1). The interatrial septum was subsequently opened, allowing for the complete excision of the mass. The excised mass was then sent for histopathological examination, which confirmed the diagnosis of a thrombus.
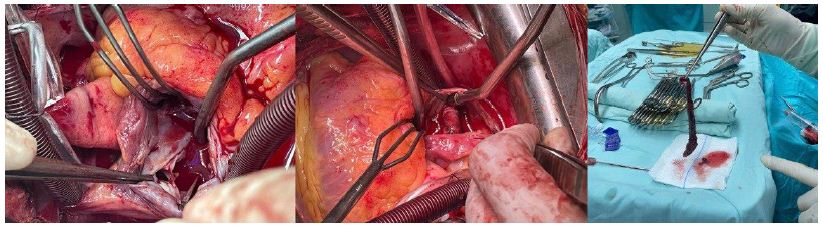
Figure 1: Demonstrates Intraoperative Views, The left image presents a close-up of the heart, highlighting the attachment point of the mass. The center image illustrates the cylindrical mass within the heart chambers. The right image depicts the excised long, cylindrical mass.
Following the successful excision of the right atrial mass, the patient was transferred to the intensive care unit (ICU) for close monitoring and comprehensive postoperative care. The immediate postoperative care involved continuous hemodynamic monitoring to ensure stability. Vital signs were closely watched, including heart rate, blood pressure, respiratory rate, and oxygen saturation. The patient remained hemodynamically stable, supported by inotropic agents such as adrenaline and noradrenaline infusion pumps as needed. Regular arterial blood gas analyses were performed to monitor respiratory and metabolic status.
Pain management was a crucial aspect of postoperative care. Pethidine 75 mg was administered intramuscularly on an as-needed basis to control pain. Pain levels were regularly assessed using a standardized pain scale. To maintain adequate hydration and electrolyte balance, intravenous fluids were administered, and serum electrolytes were closely monitored. Potassium chloride infusions were given as needed to correct any imbalances.
Wound care involved daily inspection and dressing changes to keep the surgical wound clean and dry. The initial observation of a hematoma in the upper chest necessitated careful monitoring for signs of infection or further complications. Antibiotic prophylaxis included intravenous vancomycin (500 mg three times a day) and meropenem (1 g twice a day) to prevent postoperative infections. Additionally, omeprazole 40 mg was administered intravenously once daily to prevent stress-related mucosal damage and peptic ulcers.
Nutritional support began early with enteral feeding to maintain the patient’s nutritional status. Blood glucose levels were monitored regularly, and insulin was administered to manage hyperglycemia as required.
Anticoagulation therapy was managed meticulously. Warfarin (Coumadin) therapy was temporarily withheld due to elevated International normalized ratio (INR) levels. Low molecular weight heparin (Clexane) was administered to maintain anticoagulation until INR levels stabilized. Regular INR monitoring guided the resumption and dosage adjustments of warfarin. After a Few Days, the Warfarin was restarted at 5 mg once daily, with careful monitoring to maintain therapeutic INR levels.
Unfortunately, after a few days, the patient developed significant pericardial effusion, necessitating the drainage of 1500 cc of fluid. after that, the Daily clinical assessments were monitored for signs of pericardial effusion, and repeat echocardiography was performed to evaluate pericardial fluid levels. Respiratory support included supplemental oxygen and mechanical ventilation as needed, along with incentive spirometry and chest physiotherapy to prevent atelectasis and promote lung expansion.
Shortly thereafter, the patient presented with a sudden onset of chest pain, dyspnea, and apprehension. Physical examination revealed tachypnea, tachycardia, and hypoxemia. Given the clinical suspicion of PE, an urgent computed tomography (CT) was performed, confirming the presence of right segmental and left subsegmental pulmonary embolism (PE). Additionally, laboratory tests revealed acute liver injury, characterized by markedly elevated liver enzyme levels with aspartate aminotransferase (AST) at 560 U/L and alanine aminotransferase (ALT) at 1902 U/L. Consequently, anticoagulation therapy was adjusted based on the patient’s status, with closely monitored follow-up.
Throughout his hospital stay, the patient’s liver function and coagulation profiles were closely monitored, with serial laboratory tests and imaging to assess the resolution of complications. The patient was encouraged to mobilize early to reduce the risk of further thrombotic events. As his condition stabilized, with decreasing liver enzyme levels and improvement in clinical symptoms (see Table 1), the chest drains were removed. The patient was eventually discharged with instructions for ongoing anticoagulation therapy and regular follow-up appointments to monitor his liver function and prevent recurrence of thrombotic events.
Table 1: Laboratory test results and normal ranges.
|
Lab Test |
Admission Day | 1 week after the Operation Day | Discharge Day | Normal Range |
Unit |
| Complete Blood Count | |||||
| Hemoglobin (Hb) |
17.9 |
10.0 | 13.3 | 13.5-17 |
g/dL |
| Hematocrit (Hct) |
56.6 |
35.7 | 38.8 | 43.5-53.7 |
% |
| Mean Corpuscular Volume (MCV) |
84.7 |
84.2 | 82.4 | 80-100 |
fL |
| Platelet Count |
285 |
299 | 260 | 150-450 |
10^3/μL |
| White Blood Cell (WBC) |
12.6 |
19.5 | 10.4 | 4 .0-11.0 |
K/uL |
| Coagulation Profile | |||||
| Prothrombin Time (PT) |
13 |
43 | 12 | 11-15 |
s |
| Activated Partial Thromboplastin Time (aPTT) |
32 |
39.8 | 29.7 | 25-35 |
s |
| International Normalized Ratio (INR) |
0.95 |
3.44 | 1.03 | 0.8-1.1 |
– |
| General Chemistry | |||||
| Sodium (Na) |
139 |
128 | 137 | 135-145 |
mEq/L |
| Potassium (K) |
4.3 |
4.5 | 4 | 3.5-5.3 |
mEq/L |
| Chloride (Cl) |
105 |
105 | 104 | 98-100 |
mEq/L |
| Blood Urea Nitrogen (BUN) |
13 |
13.4 | 11 | 6-20 |
mg/dL |
| Creatinine |
0.88 |
1.15 | 0.63 | 0.7-1.2 |
mg/mL |
| Liver Function Test (LFT) | |||||
| Total Bilirubin |
0.85 |
1.88 | 1.03 | 0.1-1.1 |
mg/dL |
| Direct Bilirubin |
0.311 |
0.87 | 0.432 | 0.0-0.3 |
mg/dL |
| Aspartate Aminotransferase (AST) |
23 |
560 | 41 | 0-40 |
(U/L) |
| Alanine Aminotransferase (ALT) |
24 |
1902 | 46 | 0-41 |
(U/L) |
| Others | |||||
| Troponin I |
0.3417 |
0.3318 | 0.1664 | 0-0.029 |
ng/mL |
| C-reactive protein (CRP) |
39.9 |
151 | 99.5 | 0-0.5 |
mg/dL |
| Erythrocyte Sedimentation Rate (ESR) |
12 |
22 | 15 | 0-10 |
mm/hr |
Discussion
This condition can be caused by several underlying problems, such as atrial fibrillation, deep vein thrombosis, and the presence of foreign objects, like pacemaker leads, among others. RAT carries the potential risk of severe morbidity, including pulmonary embolism and stroke, since the thrombus may pass through PFO and reach the other parts of the circulatory system. The characteristics related to size, location, and mobility are vital features in defining the clinical presentation that frequently includes signs and symptoms such as dyspnea, neck vein distension, and syncope [1,3].
RAT can develop from local intracardiac sources or as emboli originating from peripheral venous thrombi. In this case, the thrombus likely originated as a peripheral embolus, considering the patient’s history of DVT. Peripheral thrombi typically form in the deep veins of the legs due to stasis, hypercoagulability, or endothelial injury, known collectively as Virchow’s triad. When a thrombus dislodges from its peripheral origin, it travels through the venous system, ultimately reaching the right atrium. so If there is PFO, a remnant of fetal circulation present in approximately 25% of the adult population, the thrombus can pass from the right atrium into the left atrium, thereby entering the systemic circulation. This passage significantly heightens the risk of systemic embolism, including strokes or peripheral arterial emboli [1,5].
The presence of a PFO is particularly concerning because it allows for paradoxical embolism. In this scenario, a thrombus can bypass the pulmonary circulation and directly enter the systemic arterial system, leading to severe complications such as cerebral ischemia or myocardial infarction. The clinical significance of a PFO in the context of RAT lies in its potential to convert an otherwise isolated right-sided thrombus into a systemic threat. This underscores the need for immediate and precise diagnostic interventions to identify and manage such conditions effectively [1,6].
The differential diagnosis in a right atrial thrombus is exhaustive. Considerations have to be made for myxoma, other benign or malignant tumors, infective endocarditis vegetation, and foreign bodies such as pacemaker leads. To differentiate between these, requires a multi-dimensional assessment done by sophisticated imaging techniques, such as the use of echocardiography, and CT scans [7].
The diagnosis of RAT generally requires a multimodal approach using TTE, TEE, and CT to confirm. TTE is often the initial imaging modality, but sensitivity, especially for small thrombi, remains a limitation. TEE is more effective at providing clear imaging of the heart and great vessels and thus plays an important role both at the time of the initial diagnosis and during the subsequent management of patients with right atrial thrombi. A 360-degree view is provided with TEE and it is particularly useful when results of TTE are inconclusive. For example, in one study, TEE detected right atrial thrombi in all patients, whereas TTE diagnosed only a small fraction of cases with thrombi. It is also done to exclude concomitant pulmonary embolism which often can coexist with RAT [1,3,5].
Cases of right atrial thrombi mimicking myxomas are rare but have been documented in medical literature. These cases often present diagnostic challenges due to the difficulty in distinguishing between thrombus and myxoma using imaging techniques alone. In a case reported by Ondrusek et al., a 65-year-old male had a right atrial tumor initially suspected to be a myxoma, but it was found to be an aneurysm of the atrioventricular septum filled with thrombus during surgery and confirmed histologically. Similarly, Hashmath et al. described a 24-year-old woman with a right atrial mass that was initially thought to be a myxoma but was confirmed as a thrombus post-surgery. This patient had antiphospholipid antibodies, a rare factor in such cases [8,9].
Another interesting case by Khusnurrokhman and Wulandari involved a 32-year-old male with mediastinal non-Hodgkin’s lymphoma, who had a mass in the right atrium that mimicked a myxoma. The mass turned out to be a metastatic tumor thrombus. Additionally, Al-Sarraf et al. discussed a case in which a right atrial mass in a patient with antiphospholipid syndrome was misdiagnosed as myxoma but was actually a thrombus upon surgical excision [10,11].
Our case aligns with these reports in terms of diagnostic challenges and the necessity of histopathological confirmation for a definitive diagnosis. Similar to the documented cases, our patient, a 35-year-old non-smoking male with a history of DVT, presented with acute respiratory and systemic symptoms. Echocardiographic findings initially suggested a myxoma, but surgical excision and histopathological examination confirmed the mass as a thrombus. The patient’s presentation with post-exertion palpitations, respiratory distress, and acute liver injury adds a distinctive aspect to our case, not commonly reported in the literature. Moreover, our case involved a thrombus extending through a PFO into the left atrium, a unique anatomical finding compared to the other reported cases. Postoperative complications included significant pericardial effusion and pulmonary embolism, highlighting the complexity of managing such patients. These features contribute valuable insights into the clinical spectrum and management of right atrial thrombi mimicking myxomas, underscoring the importance of thorough postoperative monitoring and multidisciplinary care.
RAT should not be left untreated, especially if it moves through a PFO into the left atrium. This would demand an extensive approach since the risk of developing paradoxical embolism and systemic complications such as stroke is very high. Surgical intervention to extract the thrombus in question, supplemented by PFO closure, might also need to be done to prevent the immediate risks of embolization. This is followed by anticoagulation therapy through heparin and warfarin to prevent further thrombus formation and reduce the risk for thromboembolism. The other percutaneous approaches include the use of AngioVac aspiration system guided by TEE and fluoroscopy, and even though they are quite effective, they could be complicated by such as mechanical dislodgement and even cardiac chamber rupture. Post-procedure, long-term anticoagulation therapy and regular monitoring through imaging techniques like TEE form an important component of treatment to ensure no residual thrombus remains and the prevention of recurrence leading to poor outcomes in the patients [6,7,12].
This case describes the paramount significance of early diagnosis, timely surgical intervention, and meticulous postoperative management in handling intra-atrial thrombi. Additionally, it highlights the diagnostic complexities and the essential role of multimodal imaging in distinguishing RAT from other cardiac masses. The substantial risks associated with RAT, including pulmonary embolism and stroke, underscore the urgency of surgical excision and PFO closure. Vigilant postoperative monitoring is crucial to address potential complications such as pericardial effusion, pulmonary embolism, or SIRS, as observed in our case
We have encountered several limitations in our study. Firstly, the lack of long-term follow-up with the patient restricts our ability to assess the enduring efficacy of the surgical intervention and the ongoing risk of thromboembolic events. Secondly, although we employed multimodal imaging techniques, the case could have benefited from additional diagnostic tests, such as cardiac MRI, to provide further detail on the thrombus’s characteristics and the surrounding cardiac structures. Additionally, the absence of genetic testing to explore potential underlying thrombophilic conditions represents a gap in the comprehensive evaluation of the patient’s thrombotic predisposition
The prognosis of RAT depends on a number of factors, such as size and mobility of the thrombi, underlying health conditions, and treatment given timely and type-wise. For instance, the larger and more mobile the thrombi are, the greater the risk of complications. Additionally, underlying conditions such as atrial fibrillation, heart failure, and cancer can adversely affect outcomes. Prompt and appropriate treatment significantly improves the prognosis, although outcomes can still be variable [4].
Conclusion
This case highlights both the complexity and high risks associated with right atrial thrombi, especially those extending into a patent foramen ovale. Management that guaranteed survival in such a case entailed timely surgical intervention, comprehensive postoperative care, and close follow-up; it clearly underlines the critical need for a multidisciplinary approach. Pericardial effusion and pulmonary embolism are examples of the complications suffered that show high-risk outcomes and call for careful surveillance and adaptive treatment strategies. The report should increase awareness for right atrial thrombi and the importance of early diagnosis, complete surgical excision, and tailored postoperative management in order to enhance patient prognosis and prevent a recurrence of thrombotic events.
Conflicts of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethic Approval
Ethics approval was not required for this case report, as our institution does not mandate ethical approval for the reporting of individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for the publication of his anonymized information in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
- Carda R, Almería C, Lennie V, Serra V, Zamorano JL (2008) What to do with an atrial thrombus? Eur J Echocardiogr. [crossref]
- THE EUROPEAN WORKING GROUP ON ECHOCARDIOGRAPHY, KRONIK G (1989) The European Cooperative Study on the clinical significance of right heart thrombi. Eur Heart J. [crossref]
- Aroke D, Nnaoma CB, Nubong TF, Okoye OC, Visveswaran G (2021) Right Atrial Thrombi, the Management Conundrum 2 Case Reports. Am J Case Rep. [crossref]
- Oldershaw PJ, Coll L (1982) Echocardiographic appearances of right atrial thrombus. Clin Cardiol. [crossref]
- Crowley JJ, Kenny A, Dardas P, Connolly DL, Shapiro LM (1995) Identification of right atrial thrombi using transoesophageal echocardiography. Eur Heart J. [crossref]
- Ramamoorthy S, Mahmoud SA, High K (2017) Extraction of Atrial and Pulmonary Thrombi Using Angiovac Aspiration System with Transesophageal Echocardiography and Fluoroscopic Guidance. Open J Anesthesiol. [crossref]
- Nakashima K, Uchino H, Shimanuki T (2020) Right Atrial Thrombus which was Difficult to Differentiate from Tumor Report of a Case. Kyobu Geka. [crossref]
- Ondrusek M, Artemiou P, Gasparovic I, Hulman M (2022) Aneurysm of the atrioventricular septum mimicking myxoma in the right atrium. Kardiochirurgia Torakochirurgia Pol Pol J Cardio-Thorac Surg. [crossref]
- Hashmath Z, Bose A, Thabet R, Mishra AK, Kranis M (2022) Right Atrial Thrombus Mimicking a Myxoma Synergism of Hormonal Contraceptives and Antiphospholipid Antibodies. Tex Heart Inst J. [crossref]
- Khusnurrokhman G, Wulandari L (2021) Mediastinal Non-Hodgkin’s Lymphoma Metastatic to Right Atrium Mimicking Right Atrial Myxoma. Folia Medica Indones
- Al-Sarraf N, Abdelmoaty A, Abu Alam S, Al-Fadhli J (2019) Right Atrial Mass Mimicking a Myxoma as a First Presentation of Antiphospholipid Syndrome. Heart Surg Forum. [crossref]
- Feuchter AC, Katz KD (2012) Right Atrial Thrombus Secondary to Pacemaker Wires. J Emerg Med. [crossref]