Abstract
A theory of petroleum formation from algal blooms is revised in the light of recently obtained data on biochemical composition of dinoflagellate lipids, microalgal blooms, “marine snow” and its composition, taxonomy of major phytoplankton groups and their relative abundances. The article is illustrated with electron micrographs of dinoflagellates, a scheme of petroleum formation and a map. It is concluded that seasonal dinoflagellate blooms or those of a longer period may be the principal agent in the petroleum formation.
Keywords
Algal blooms, Dinoflagellata, Marine phytoplankton, Microalgae, Oil, Petroleum formation, Petroleum resources
Introduction
Petroleum (crude oil) has played an important role for humans worldwide. During the 20th century until the present it has been an important factor in geopolitics and conflicts between countries, determining the welfare of individual humans through that of entire nations. During wars that depended on energy sources for military vehicles, it has been considered the blood of war and the blood of the economy. Today oil is the primary source of energy and the most important commodity traded among many countries. More than half of the energy that powers our civilization comes from this non- renewable energy source. Therefore, it is a strategic resource whose scarcity would cause the decline of the global economy [1,2]. Oil is not distributed evenly around the world, so many countries are heavily dependent on nations with oil resources. At present, the world’s largest proven petroleum reserves are in Venezuela, Saudi Arabia, Iran, Canada, Iraq, United Arab Emirates, Kuwait, Russia, United States of America, and Libya.
Among the hypotheses that could explain the origin of petroleum during the geological history of the Earth, there is one based on the suggestion that marine phytoplankton (in particular, algal blooms caused by dinoflagellates) are the principal source of petroleum resources [3]. Herein are some suggestions that we consider relevant:
- “Judging from… rough correspondences between distribution of areas of oil fields and supposedly great expanse of the so- called Old Tethys Sea, or became more and more preferable to conclude that petroleum was derived primarily from phytoplankton of this sea.”
- “Frequent occurrences of intense red water blooms in coastal shallow water may be responsible for petroleum formation throughout long continued geological periods.”
- “Planktonic diatoms are non-motile, but usually possess spiny surface extensions which give them resistance to passage through water. Largely due to this, over-crowding of diatoms is theoretically limited. In contrast, dinoflagellates are motile but frequently do not possess floatation structures.”
- “Poisonous effects caused by dinoflagellate also So that it is not phytoplankton only, but also all of higher marine organisms killed by intense red water occurrence that are most highly responsible for the formation of petroleum.”
The present study reviews some aspects of this hypothesis emphasizing the advances in our knowledge of algal blooms and major microalgal taxonomic groups, in particular, of recent marine dinoflagellates.
Material and Methods
To discuss Abe’s suggestions [3] previously presented, some old and modern literature was selected and reviewed. Some dinoflagellate species were selected to illustrate dinoflagellate morphological diversity. The cells were collected in the coastal water of the Peninsula of Yucatán, the southern Gulf of Mexico during the period of 2009-2012. Phytoplankton samples were fixed with a stock formaldehyde solution to a final concentration of 4% and stored in 100-ml plastic bottles. They were examined in a JEOL JSM-7600F scanning electron microscope (SEM) at a working distance of 8 to 15 mm and a voltage of 5.0 kV after a preliminary wash in distilled water, followed by dehydration in a series of ethanol solutions of increasing concentration (30, 50, 70 and 90% and twice in 100%), air drying on 0.5” aluminum mounts and sputter coating with gold-palladium using a Polaron SC7640 High Resolution Sputter Coater (Quorum Technologies, Newhaven, East Sussex, U.K.).
Results and Discussion
Elemental Composition and Generation of Petroleum Based on Organic Matter
Hydrocarbons (petroleum) originate from the thermogenic transformation of organic matter preserved in sedimentary basins. From a geological perspective, the occurrence of oil and gas reservoirs is intrinsically linked to the presence of source rocks within stratigraphic sequences [4]. Although approximately 98% of the Earth surface crust is composed of sediments or sedimentary rocks, the confluence of critical conditions for the formation of petroleum systems, such as generation, migration and entrapment, is a restrictive phenomenon, present in less than 1% of this surface [5]. At the geochemical level, while the quality of crude oil exhibits subtle variations depending on its geographic origin and thermal maturity, its elemental composition is remarkably constant. Petroleum is a complex mixture of organic compounds where the predominant elements are carbon and hydrogen, which represent approximately 95% of the total mass. The remaining percentage (between 1% and 7%) corresponds to heteroatoms, mainly sulfur, oxygen and nitrogen, whose presence and proportion define critical properties, such as viscosity and the degree of corrosivity of the fluid [6]. The generation of oil based on the amount of organic matter in marine and terrestrial systems has been accepted since the 1990s. Deng et al. (2023) [7] described the theoretical basis of oil and gas generation worldwide and confirmed that amount of hydrocarbons is closely correlated to the organic matter load present in sedimentary rocks. Furthermore, the organic matter used for oil production depends primarily on photosynthetic organisms (phytoplankton) in marine systems.
Biochemical Composition of Dinoflagellate Lipids
Due to their composition and origin, lipids produced by phytoplankton have been the subject of study for the last 50 years [8], with triacylglycerols, galactolipids and phospholipids being the main lipid components in these microorganisms [9,10]. These lipids have been of great importance in the field of biotechnology, mainly for the synthesis of a variety of saturated fatty acids, monounsaturated fatty acids, and polyunsaturated fatty acids (e.g., oleic acid (OA; 18:1(n-9), arachidonic acid (AA; 20:4(n-6), eicosapentaenoic acid (EPA; 20:5(n- 3), docosapentaenoic acid (DPA; 22:5(n-3), and docosahexaenoic acid (DHA; 22:6(n-3) that make up 5 to 60% of the cell dry weight [11,12]. In particular, triacylglycerol contributes up to 30%, free fatty acids up to 10%, and sterols approximately 5% of the phytoplankton lipid composition [10]. These lipids have multiple functions, from energy storage to the synthesis of membranes and bioactive substances that ensure their ecological success [13,14]. In particular, dinoflagellates can synthesize a mixture of highly unsaturated long-chain fatty acids (C28) and complex sterols, such as 4-methyl sterols and 4-desmethyl sterols, which serve as characteristic chemotaxonomic indicators [15,16]. These lipids are highly similar to crude oil, mainly due to the high carbon content (>85%) [4]. This is relevant for elucidating the origins of petroleum because of the quality of its organic matter; it is rich in complex lipids and specific biomarkers called dinosteranes (Table 1). These compounds are highly resistant and contribute to the formation of type I and II kerogen, both precursors of crude oil [17].
Table 1: Contribution of some groups of microorganisms to marine organic matter; Phyto – phytoplankton; Zoo – zooplankton.
|
Taxonomic group |
Type | Main contribution to organic matter | Sedimentation rate (m day-1) |
Petroleum potential (kerogen) |
| Diatoms | Phyto | Lipids and silica |
0.5-100* |
Very high: Primary source of lipids (Type II) |
| Dinoflagellates | Phyto | Dinidporins (polymers) and sterols |
0.5-10 |
High: Excellent preservation of biopolymers |
| Coccolithophorids | Phyto | Calcium carbonate |
1-50* |
Medium: Provide ballast for massive sinking |
| Cyanobacteria | Phyto | Labile organic matter (proteins) |
< 0.5 |
Low: Mostly recycled at the surface |
| Copepods | Zoo | Compact fecal pellets |
30-160 |
Indirect: Package and protect lipids |
| Krill (Euphausiaceae) | Zoo | Fecal pellets and active transport |
150-350 |
Medium: Inject carbon at great depths |
| Salps | Zoo | Large and dense fecal pellets |
400-1,200 |
High: Massive and ultra-fast carbon export |
| Appendicularians | Zoo | Marine snow (mucus houses) |
50-200 |
Medium: Trap fine particles in aggregates |
| Foraminiferans | Zoo | Calcareous skeletons |
100-500 |
Low-medium: Key to source rock density |
Algal Blooms
An algal bloom caused by dinoflagellates is a highly productive event that drastically alters carbon fluxes in the ocean. The biomass of these microorganisms can reach massive concentrations, commonly known as “red tides” [20]. Unlike diatoms, dinoflagellates possess a particular chemical composition that influences the production of organic matter, which is ultimately preserved [21]. During bloom events, the concentration of organic carbon can exceed 500 to 1,000 µg of carbon per liter, which is 10 to 50 times higher than under normal ocean conditions [22]. Furthermore, net primary production rates of 2 to 5 g C m-2 day-1 have been recorded during the peak of the bloom; additionally, many dinoflagellates produce resistant cysts, whose walls are made of dinosporins (polymers similar to sporopollenin), which increases the rate of sedimentation and transport of carbon in the deep zones [17].
Much literature has been published about harmful algal blooms (HAB), caused by both toxic and non-toxic species, and various aspects of the so-called HAB science. Most bloom-forming marine species are dinoflagellates, and many of them have been reported toxic in any part of the world ocean, in marine environments, brackish and freshwater bodies [23]. At present, pelagic and benthic blooms are distinguished. Recently, (epi)benthic species have been paid much attention, and 242 dinoflagellate species from 63 genera have been described: most of them are sand-dwelling followed by epiphytic [24]. As for the duration of HAB events, they may last from several hours to more than a year. For example, the Karenia brevis (C.C. Davis) Gert Hansen & Moestrup (= Gymnodinium breve C.C. Davis) blooms on the West Florida shelf may last more than a year, and 8-12-month blooms are not uncommon there [25]. While in freshwater most toxic species are cyanobacteria (97 species from 10 orders), in the marine environment, especially in the coastal zone, dinoflagellates have the highest species richness (118 species from 8 orders) (Figure 1).

Figure 1: Electron micrographs of recent marine dinoflagellates from the coastal zone of the southern Gulf of Mexico (A, B, E, and H are from the Yucatán Peninsula; C, D, F and G are from the state of Veracruz): A – Pyrodinium bahamense L. Plate is highly toxic producer of paralytic shellfish poisoning (PSP) in humans, distributed in tropical waters; B – Gonyaulax polygramma F. Stein is a non-toxic species causing harmful algal blooms in subtropical waters; C – Ceratocorys horrida F. Stein is distributed mainly in the open sea in the tropical zone, possesses membraned spines characteristic of warm waters; D – Blixaea quinquecornis (T.H. Abé) Gottschling in Gottschling et al. is one of the few benthic-planktonic dinoflagellates with a tropical-boreal distribution, inhabiting both marine and estuarine brackish waters, non-toxic, causing intense blooms in the southern Gulf of Mexico; E – Scrippsiella spinifera G. Honsell & M. Cabrini is a non-toxic species described from the Mediterranean Sea in 1991, recently found in the southern Gulf of Mexico together with the blooming species Scrippsiella acuminata (Ehrenberg) Kretschmann, Elbrächter, Zinssmeister, S. Soehner, Kirsch, Kusber & Gottschling, capable of producing calcareous resting cysts unlike most dinoflagellates; F – Phalacroma rapa F. Stein is a non-toxic species; G – Dinophysis caudata Kent is a toxic neritic tropical-boreal species, producing okadaic acid and causing diarrhetic shellfish poisoning (DSP) in humans; H – Prorocentrum cordatum (Ostenfeld) J.D. Dodge is a toxic bloom-forming species, causing hepatotoxicity in mice and adverse effects on invertebrates, inhabiting both marine and brackish waters.
Other major microalgal groups are Bacillariophyceae (31 species), Haptophyta (8), Raphidophyceae (4) and Dictyochophyceae (3) [23]. Sixty-seven species and the taxa identified to the generic level are harmful non-toxic, not considering 20 species in the so-called grey list. It is worth considering that about one half of the recent dinoflagellate species are heterotrophs, and the other half are photosynthetic, most of which are mixotrophs. Strictly speaking, heterotrophic species are not phytoplankton if we define it as composed of photosynthetic organisms. Furthermore, ciliates, which also belong to the clade Alveolata as do dinoflagellates, include many photosynthetic species (also known as plastidic ciliates; facultative or obligate autotrophs); e.g., Mesodinium rubrum (Lohmann) (= Myrionecta rubra Lohmann) is an obligate autotroph that contains endosymbiotic cryptophytes [26].
“Marine Snow” and Its Composition
Microalgal cells and, in particular, dinoflagellates cells, can be part of the phenomenon known as “marine snow”, organic aggregates, particle flux, detrital material, pelagic sediment(s) or particulate organic matter [27-37]. “The scientists used the term “marine snow” for the abundant, readily visible, suspended particles in the water.” [37: 6]. “Material exported from the euphotic zone leaves as large, fast-sinking particles, and constitutes a source of food for pelagic and benthic organisms.” [35: 565]. Marine snow consists of detritus (organic debris including fecal pellets), living organisms (phytoplankton, protozoans, zooplankton) and inorganic matter, with associated microbial communities [30]. The history of marine snow research, emphasizing the major discoveries prior to the end of the 20th century, was presented by Silver (2015) [37]. In addition, marine snow-like particles, such as fecal pellets (mainly of the crustacean zooplankton) and amorphous aggregations, are distinguished [34]. It is also worth noting that microaggregates found in the water column [38] are different from large or macroscopic aggregates [31,32], which are synonymous with marine snow. The composition of marine snow has been actively studied during several decades hitherto. Both prokaryotic and eukaryotic microalgae are part of it. In earlier studies, as components of sinking particles, diatoms, dinoflagellates, coccolithophorids and unidentified (micro)flagellates [35,36] have been commonly mentioned. Apart from those, the following taxonomic groups of unicellular eukaryotic organisms have been mentioned more frequently: radiolarians and foraminiferans [34]. It is logical to suggest that, considering the advances in the field of taxonomy of major microalgal groups (discussed below), most become a part of marine snow or microaggregates and participate in vertical carbon flux. Based on the data obtained with the use of sediment trap deployments in all oceans with a sampling duration of more than one year, it was concluded that at some locations the particle flux maxima are the results of bloom periods of individual phytoplankton species [33]; the highest primary production values up to more than 100 g m-2 yr-1 were related to the blooming species of dinoflagellates, diatoms and coccolithophorids [39,40].
The Origin of Petroleum
A historical overview of the origin of petroleum was given by Walters (2006) and Höök et al. (2010) [40,41]. All theories of petroleum origin can be divided into abiotic (= abiogenic or non- biogenic) and biotic (= biogenetic) (Figure 2).
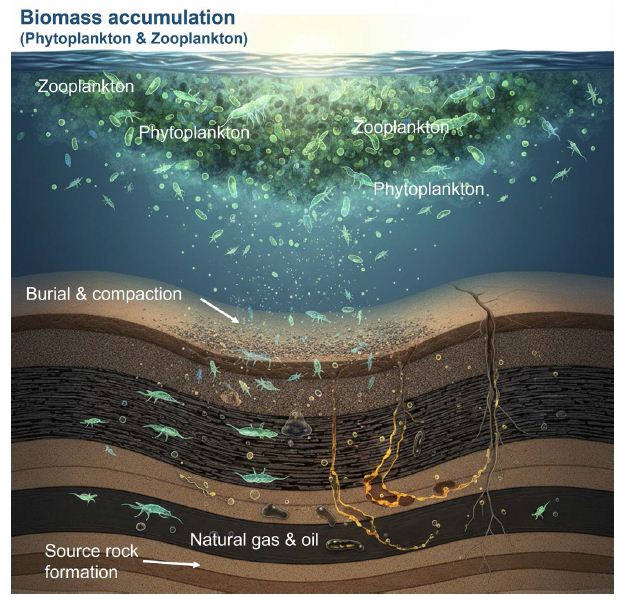
Figure 2: The importance of phytoplankton and zooplankton in the formation of petroleum in marine systems.
Previously, it was concluded that the origin of petroleum is duplex, organic and inorganic, stating that petroleum was laid down in Cambrian and Ordovician strata during the last 500 million years [42] for the distribution of the land masses and water basins on the Earth 500 Mya, see Figure 3).
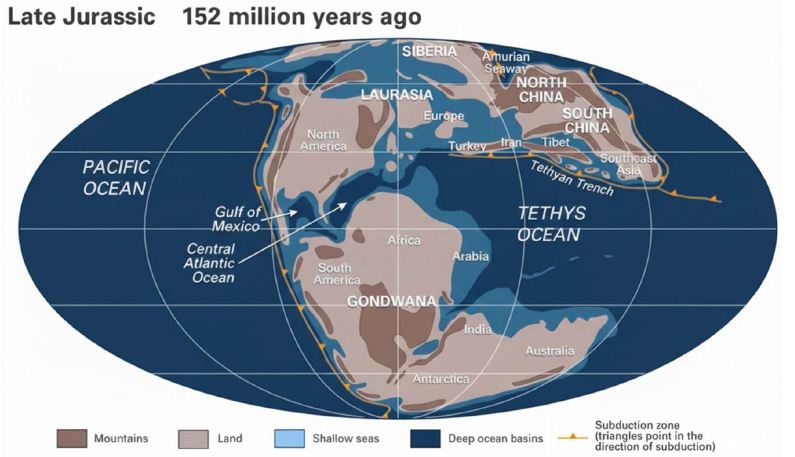
Figure 3: Distribution of the land masses and oceans on the Earth in the Late Jurassic; modified from Scotese (2001), and Encyclopædia Britannica.
Paleontologists indicate a possible Precambrian origin of dinoflagellates more than 570 years ago [44]. Ehrenberg (1836) [45] was the first to recognize fossil dinoflagellate cysts. At least 2,294 dinoflagellate species have been described [46], and ca. 13-16% of them produce resting cysts also called dinocysts [47]. Resting cyst are the result of sexual reproduction, and they are diploid hypnozygotes (zygotic resting cysts), unlike haploid vegetative cells (except for Noctilucophyceae that have diplontic life-cycle, i.e. with the predominant diplontic stage). However, sexual reproduction has been known only for 1% of recent dinoflagellate species, but it is suggested that it could be a universal phenomenon for this taxonomic group; the zygotic nature of most fossil cysts remains unproven [44].
Recent Advances in the Taxonomy of Major Phytoplankton Groups and Their Relative Abundances
During the last decades a number of new classes of microalgae were described, including previously described taxa of the lower taxonomic level: Dictyochophyceae Silva 1980; Coleochaetophyceae Jeffrey 1982; Pavlovophyceae Cavalier-Smith 1986, emend. Green & Medlin 2000; Pedinophyceae Moestrup 1991, emend. Fawley, Zechman & Buchheim in Adl et al. 2012; Pelagophyceae Andersen & Saunders 1993; Noctilucophyceae Fensome et al. 1993; Klebsormidiophyceae van den Hoek et al. 1995; Trebouxiophyceae Friedl 1995; Zygnematophyceae van den Hoek et al. 1995, emend. Hall et al. 1999; Bolidophyceae Guillou et al. 1999; Pinguiophyceae Kawachi et al. 2003; Chlorokybophyceae Lewis & McCourt 2004; Mesostigmatophyceae Marin & Melkonian 1999, emend. Lewis & McCourt 2004; Mediophyceae Jouse & Proshkina-Lavrenko in Medlin & Kaczmarska 2004; Mamiellophyceae Marin & Melkonian 2010; Palmophyllophyceae Lelaert et al. 2016; Chloropicophyceae Lopes dos Santos & Eikrem 2017, Picocystophyceae Lopes dos Santos & Eikrem 2017; Biddulphiophyceae D.G. Mann in Adl et al. 2019; Leprocylindrophyceae D.G. Mann in Adl et al. 2019; Corethrophyceae D.G. Mann in Adl et al. 2019 [48]. Most major microalgal taxa have been described since 1990s based on transmission electron and light microscopy observations, and some were also based on biochemical data (e.g., Pelagophyceae and Bolidophyceae). Recently, metagenomic data of the relative abundances of different taxonomic groups in marine phytoplankton of all the oceans, based on the Tara Oceans Project in 2009-2013, have been published. Although dinoflagellates and diatoms remain the dominant groups in terms of the cell abundance, there are other groups (the picocyanobacterial genera Prochlorococcus Chisholm, Frankel, Goericke, Olson, Palenik, Waterbury, West- Johnsrud & Zettler ex Komárek et al. and Synechococcus Nägeli, Haptophyta (= Prymnesiophyta), Chlorophyta and Pelagophyta [49,50].
Protecting Marine Ecosystems Where Phytoplankton Live Could Ensure Petroleum Resources in the Future
Phytoplankton are a group of unicellular and colonial photosynthetic organisms capable of inhabiting diverse marine and freshwater bodies [51]. In aquatic ecosystems, phytoplankton comprises a network of biological interactions that have enabled successful conversion of light energy to chemical energy through biogeochemical cycles. Therefore, along with higher plants, phytoplankton provides a solid foundation of ecosystem services in both terrestrial and marine environments [52]. These services include supporting, regulating, provisioning and cultural services. It is estimated that 45% of global net primary production or more (≈108 pg C yr-¹) comes from phytoplankton [53,54]. In particular, groups such as diatoms and dinoflagellates are the primary source of the organic matter that makes up kerogen types I and II. These microorganisms synthesize lipid compounds that, when attached to sediments, possess the necessary energy density to transform into liquid petroleum [17]. Therefore, the protection of marine systems should be treated globally as a matter of energy security. This will ensure phytoplankton biodiversity, which, in turn, guarantees that the chemical quality of the organic matter reaching the seabed is optimal for hydrocarbon generation [55].
Acknowledgments
We thank Marcia M. Gowing (Seattle, WA, USA) for improving the English style and for valuable advice, and Dora A. Huerta- Quintanilla (CINVESTAV-IPN, Mérida, Yucatán) for her help with the scanning electron microscope. Financial support to the FOMIX CONACYT-Yucatán project “Análisis de las causas, dispersión y consecuencias ambientales de la marea roja en Yucatán” (No. 108897; 2009–2012) given to Jorge A. Herrera-Silveira (CINVESTAV-IPN), and the FOMIX CONACYT-Yucatán (No. 108160) and CONACYT LAB-2009-01 (No. 123913) projects of the Laboratorio Nacional de Nano y Biomateriales (CINVESTAV-IPN) given to Patricia Quintana- Owen is much appreciated.
References
- Palazuelos E (2009) El petróleo y el gas en la geoestrategia Editions Akal.
- Krishna S, Gambelli AM, Sreenivasan H, Vadim F, et al. (2026) The petroleum industry and climate change. In Al-Enazi A, Al-Musleh E (Eds) Decarbonizing the petroleum Elsevier.
- Abé TH (1965) Red water and dinoflagellates as primary sources of In Second International Conference on Protozoology, London, August 1965. Reprinted from Excerpta Medica International Congress Series No. 91.
- Liu H, Jiang Y (2025) Petroleum Springer. Singapore.
- Biju-Duval B (2002) Sedimentary geology: sedimentary basins, depositional environments, petroleum formation. Editions Technip.
- Kundu N (2023) Geoscience for petroleum engineers. Springer, Singapore.
- Deng Y, Yang Y, Yang T (2023) Three systems of the oil and gas formation in the Acta Petrolei Sinica 44.
- Barkia I, Saari N, Manning SR (2019) Microalgae for high-value products towards human health and Marine Drugs 17.
- Falk-Petersen S, Sargeant JR, Henderson J, Hegseth EN, et (1998) Lipids and fatty acids in ice algae and phytoplankton from the Marginal Ice Zone in the Barents Sea. Polar Biology 20: 41-47.
- Jónasdóttir SH (2019) Fatty acid profiles and production in marine Marine Drugs 17.
- D’Alessandro EB, Antoniosi Filho NR (2016) Concepts and studies on lipid and pigments of microalgae: A Renewable and Sustainable Energy Reviews 58: 832-841.
- Morales M, Aflalo C, Bernard O (2021) Microalgal lipids: A review of lipids potential and quantification for 95 phytoplankton species. Biomass and Bioenergy 150.
- Ianora A, Bentley MG, Caldwell GS, Casotti R, Allan DC, et al. (2011) The relevance of marine chemical ecology to plankton and ecosystem function: an emerging field. Marine Drugs 9: 1625-1648. [crossref]
- Goncalves EC, Johnson JV, Rathinasabapathi B (2013) Conversion of membrane lipid acyl groups to triacylglycerol and formation of lipid bodies upon nitrogen starvation in biofuel green algae Chlorella Planta 238: 895-906. [crossref]
- Cárdenas P, Acuña V, Contreras S, Salgado P, et (2025) The sterol composition of harmful dinoflagellates distinguishes environmental characteristics of Chilean Patagonia. Marine Environmental Research 214. [crossref]
- Rathinavel L, Ravikumar Y, Jothinathan D, Pandey, A, et (2025). Extraction and enrichment of fatty acids from marine microalgae. In Marine molecules from algae and cyanobacteria. Elsevier. [crossref]
- Tissot BP, Welte DH (1984) Petroleum formation and Springer, Berlin, Germany.
- Bach LT, Bauke C, Meier KJS, Riebesell U, et (2012) Influence of changing carbonate chemistry on morphology and weight of coccoliths formed by Emiliania huxleyi. Biogeosciences 9: 3449-3463.
- Turner JT (2015) Zooplankton fecal pellets, marine snow, phytodetritus and the ocean’s biological Progress in Oceanography 130: 205-248.
- Hallegraeff GM (2003) Harmful algal blooms: A global In Hallegraeff GM, Anderson DM, Cembella AD (Eds) Manual on harmful marine microalgae (2nd ed). UNESCO, Paris, France.
- Inomura K, Deutsch C, Jahn O, Dutkiewicz S, et al. (2022) Global patterns in marine organic matter stoichiometry driven by phytoplankton ecophysiology. Nature Geoscience 15: 1034-1040. [crossref]
- Smayda TJ (1997) Harmful algal blooms: their ecophysiology and general relevance to phytoplankton blooms in the Limnology and Oceanography 42: 1137-1153.
- Lundholm N, Bernard C, Churro C, Escalera L, et al. (Eds) (2026) IOC-UNESCO taxonomic reference list of harmful microalgae.
- Hoppenrath M, Chomérat N, Horiguchi T, Murray SA, Rhodes L (2023) Marine benthic dinoflagellates – their relevance for science and society (2nd ed). Senckenberg Gesellschaft für Naturforschung, Germany.
- Kusek KM, Vargo GA, Steidinger KA (1999) Gymnodinium breve – A scientific and journalistic analysis. Contributions in Marine Science 34: 1-229.
- Barber RT, White AW, Siegelman HW (1969) Evidence for a cryptomonad symbiont in the ciliate, Cyclotrichium meunieri. Journal of Phycology 5: 86-88. [crossref]
- Riley GA (1963) Organic aggregates in seawater and the dynamics of their formation and Limnology and Oceanography 8: 372-381.
- Riley GA (1970) Particulate matter in seawater. Advances in Marine Biology 8: 1-118.
- Silver MW, Shanks A, Trent JD (1978) Marine snow: microplankton habitat and source of small-scale patchiness in pelagic populations. Science 201: 371-373. [Crossref]
- Trent JD, Shanks AL, Silver MW (1978) In situ and laboratory measurements on macroscopic aggregates in Monterey Bay, Limnology and Oceanography 23: 626-635.
- Shanks AL, Trent JD (1979) Marine snow: Microscale nutrient patches. Limnology and Oceanography 24: 850-854.
- Alldredge AL, Silver MW (1988) Characteristics, dynamics and significance of marine Progress in Oceanography 20: 41-82.
- Wefer, G (1989) Particulate flux in the ocean: Effects of episodic In Berger WH, Smetacek VS, Wefer G (Eds) Productivity of the ocean. John Wiley & Sons Limited, S. Bernhardt, Dahlem Konferenzen, Pg: 139-153.
- Silver MW, Gowing MM (1991) The “particle” flux: origins and biological Progress in Oceanography 26: 75-113.
- Lee C, Wakeham S, Arnosti C (2004) Particulate organic matter in the sea: The composition AMBIO 33: 565-575.
- Ploug H, Iversen MH, Fischer G (2008) Ballast, sinking velocity, and apparent diffusivity within marine snow and zooplankton fecal pellets: Implications for substrate turnover by attached bacteria. Limnology and Oceanography 53: 1878-1886.
- Silver M (2015) Marine snow: A brief historical ASLO.
- Goldman JC (1984) Conceptual role for microaggregates in pelagic waters. Bulletin of Marine Science 35: 462-476.
- Honjo S (1982). Seasonality and interaction of biogenic and lithogenic particulate flux at the Panama Science. 218: 883-884. [crossref]
- Walters CC (2006) The origin of petroleum. In: Hsu CS, Robinson PR (Eds) Practical advances in petroleum Springer, New York, NY, USA. Pg: 79-101.
- Höök M, Bardi U, Feng L, Pang X (2010) Development of oil formation theories and their importance for peak Marine and Petroleum Geology 27: 1995-2004.
- Robinson R (1960) The origins of Nature 5068: 1291-1295.
- Scotese CR (2001) Atlas of Earth PAKEOMAP Project.
- Fensome RA, Taylor FJR, Norris G, Sarjeant WAS, et al. (1993) A classification of living and fossil Sheridan Press, Hanover, PA, USA.
- Ehrenberg CG (1836) Zusätze zur Erkenntnis grosser organischer Ausbildung in den kleinsten thierschen Organismen. Preussische Akademie der Wissenschaften, Abhandlungen 1835: 151-180.
- Gómez F (2012) A checklist and classification of living dinoflagellates (Dinoflagellata, Alveolata). CICIMAR Oceánides 27: 65-140.
- Head MJ (1996) Modern dinoflagellate cysts and their biological In Jansonius J, McGregor DC (Eds). Palynology: principles and applications. The American Association of Stratigraphic Palynolologists Foundation. Pg: 1197-1248.
- Adl SM, Bass D, Lane CE, Lukeš J, et (2019) Revisions to the classification, nomenclature, and diversity of eukaryortes. Journal of Eukaryotic Microbiology 66: 4-119. [crossref]
- Pierella Karlusich JJ, Ibarbalz FM, Bowler C (2020) Phytoplankton of the Tara Annual Review of Marine Science 12: 233-265. [crossref]
- Pierella Karlusich JJ, Pelletier E, Zinger L, Lombard F, et (2023). A robust approach to estimate relative phytoplankton cell abundances from metagenomes. Molecular Ecology Resources 23: 16-40. [crossref]
- Reynolds CS (2006) The ecology of Cambridge University Press.
- Naselli-Flores L, Padisák J (2023) Ecosystem services provided by marine and freshwater Hydrobiologia 850: 2691-2706. [crossref]
- Field CB, Behrenfeld MJ, Randerson JT, Falkowski P (1998) Primary production of the biosphere: Integrating terrestrial and oceanic components. Science 281: 237-240. [crossref]
- Wanek E, Esteban-Cantillo OJ, Bourgeois-Gironde S (2025) Valuing marine plankton: a review of ecosystem services and disservices and an expert assessment of the potential of area-based Frontiers in Marine Science 12.
- Peters KE, Walters CC, Moldowan JM (2005) The biomarker guide: Biomarkers and isotopes in petroleum systems and Earth Cambridge University Press.