Abstract
Background: Plant-derived nanomedicine has emerged as a promising strategy for enhancing anticancer efficacy while reducing toxicity. Corallocarpus epigaeus is a medicinal plant rich in bioactive phytochemicals with reported pharmacological properties.
Objective: The present study aimed to compare the cytotoxic effects of acetone extract of Corallocarpus epigaeus tuber and gold nanoparticles (AuNPs) synthesized using the same extract against the human epidermoid carcinoma A-431 cell line.
Methods: Cytotoxicity was evaluated using the MTT assay across a range of concentrations(1-500 µg/ml). IC50 values were determined by linear regression analysis using a Log(inhibitor) verses normalized response model with variable slope.
Results: The acetone extract exhibited moderate cytotoxicity with an IC50 value of 84.67 µg/ml. In contrast Corallocarpus mediated AuNPs showed significant enhanced cytotoxic activity, with a reduced IC50 value of 57.97 µg/ml. Gold nanoparticles demonstrated greatest growth inhibition at higher concentrations, reducing cell viability to 12.94% at 500 µg/ml compared to 27.45% for the crude extract.
Conclusion: Incorporation of Corallocarpus epigaeus bioactive components into gold nanoparticles significantly enhanced anticancer efficacy, highlighting the potential of plant-mediated AuNPs as a promising nanomedicine approach for epidermoid carcinoma therapy.
Keywords
Corallocarpus epigaeus, Gold nanoparticles, Nanomedicine, Cytotoxicity, A-431 cell line, IC50
Introduction
Cancer remains one of the leading causes of mortality worldwide, necessitating the development of novel therapeutic strategies with improved efficacy and reduced side effects. Nanomedicine has emerged as a transformative approach in cancer treatment by enabling targeted delivery, enhanced cellular uptake and controlled release of therapeutic agents [1,2]. Gold nanoparticles (AuNPs) are particularly attractive in nanomedicine due to their biocompatibility, ease of synthesis, tunable size, and surface functionalization capability [3]. Recently, green synthesis of nanoparticles using plant extracts has gained considerable attention as an eco-friendly and cost- effective alternative to conventional chemical methods [4]. Plant- derived phytochemicals act as both reducing and stabilizing agents, often conferring additional biological activity to the nanoparticles. Corallocarpus epigaeus (family: Curcubitaceae) is a traditional medicinal plant known for its antioxidant, anti-inflammatory and antimicrobial properties [5]. However, its anticancer potential, particularly in nanoparticle form, remains unexplored. Epidermoid carcinoma A-431 cells serve as a well-established in-vitro model for studying skin cancer therapeutics [6]. The present study was designed to compare the cytotoxic efficacy of acetone extract of Corallocarpus epigaeus tuber and gold nanoparticles synthesized using the same extract, with the focus on evaluating the nanomedicine advantage through IC50 analysis.
Materials and Methods
Plant Collection and Identification
C. epigaeus plant species was obtained from Solavanthan village, Madurai in December 2023. The plant specimens (ARIBASKBI-1) were identified by Plant Taxonomist Dr. Karuppusamy at Department of Botany, The Madura College, Madurai, Tamil Nadu, India. The specimen is stored at Botany department under a herbarium voucher LSID-2PNI-20013498. Only the healthy and disease-free leaves of the plant were taken for testing. Fresh tubers of Corallocarpus epigaeus were collected, shade dried and powdered. The powdered material was extracted using acetone by standard solvent extraction methods. The extract was filtered and concentrated under reduced pressure.
Green Synthesis of Gold Nanoparticles
Gold nanoparticles were synthesized using the acetone extracts of Corallocarpus epigaeus tuber as a reducing and stabilizing agent. Formation of AuNPs was confirmed visually by colour change
Cell Culture
The human epidermoid carcinoma A-431 cell line was obtained from a certified repository and maintained in DEME supplemented with 10% feta bovine serum and antibiotics at 37°C in a humified 5% CO2 incubator.
MTT Cytotoxicity Assay
The sample was tested for in vitro cytotoxicity, using Cells by MTT assay. Briefly, the cultured Vero Cells were harvested by trypsinization, pooled in a 15 ml tube. Then, the cells were plated at a density of 1×105 cells/ml cells/well (200 µL) into the 96-well tissue culture plate in DMEM medium containing 10 % FBS and 1% antibiotic solution for 24-48 hour at 37°C. The wells were washed with sterile PBS and treated with various concentrations of the sample in a serum-free DMEM medium. Each sample was replicated three times and the cells were incubated at 37°C in a humidified 5% CO2 incubator for 24 h. After the incubation period, 20 µL of 5 mg/mL MTT was added to each well. The cells were then incubated for an additional 2–4 hours, until purple precipitates were clearly visible under an inverted microscope. Finally, the medium was removed from the wells and the wells were washed with 1X PBS (200 µL). Additionally, to dissolve the formazan crystals, 100 µL of DMSO was added, followed by 5 min of shaking. The absorbance for each well was measured at 570 nm using a microplate reader and the percentage cell viability and IC50 value was calculated using Graph Pad Prism 6.0 software (USA).
![]()
IC50 Determination
Dose-response curves were generated using non-linear regression analysis with a variable slope model IC50 values were calculated along with standard errors and 95% confidence intervals.
Results and Discussion
Cytotoxic Activity of Acetone Extract of Corallocarpus epigaeus on A-431 Cells
The cytotoxic activity of the acetone extract of Corallocarpus epigaeus was evaluated against human epidermoid carcinoma (A- 431). The acetone extract of Corallocarpus epigaeus tuber exhibited a concentration dependent cytotoxic effect against the human epidermoid carcinoma A-431 cell line. A significant reduction in cell viability was observed with increasing concentration of the extract (Tables 1, 2 and Figure 1).
Table 1: Cytotoxic property of acetone extract of Corallocarpus epigaeus on A-431 cell line.
|
S. No |
Tested sample concentration (μg/ml) | Cell viability (%) (in triplicates) |
Mean Value (%) |
||
| 1. | Control |
100 |
100 | 100 |
100 |
| 2. | 500 µg/ml |
25.8278 |
25.76 | 30.756 |
27.447943 |
| 3. | 400 µg/ml |
29.9669 |
31.68 | 38.3162 |
33.321013 |
| 4. | 300 µg/ml |
41.8874 |
41.92 | 41.5808 |
41.796058 |
| 5. | 200 µg/ml |
45.5298 |
44.96 | 51.2027 |
47.23085 |
| 6. | 100 µg/ml |
58.1126 |
52.96 | 56.5292 |
55.867264 |
| 7. | 50 µg/ml |
61.755 |
57.92 | 67.354 |
62.342973 |
| 8. | 25 µg/ml |
82.4503 |
75.36 | 69.244 |
75.684772 |
| 9. | 10 µg/ml |
68.3775 |
72.8 | 75.945 |
72.374167 |
| 10. | 5 µg/ml |
77.9801 |
79.68 | 79.3814 |
79.013859 |
| 11. | 1 µg/ml |
82.4503 |
66.4 | 80.9278 |
76.592722 |
Table 2: Percentage Viability acetone extract of Corallocarpus epigaeus on A-431 cell line.
|
S. No |
Tested sample concentration (μg/ml) | Cell viability (%) (in triplicates) |
Mean Value (%) |
||
| 1. | Control |
100 |
100 | 100 |
100 |
| 2. | 500 µg/ml |
13.9738 |
8.11639 | 16.7192 |
12.936476 |
| 3. | 400 µg/ml |
29.5488 |
27.1057 | 26.6562 |
27.770193 |
| 4. | 300 µg/ml |
34.3523 |
39.3568 | 39.2744 |
37.661173 |
| 5. | 200 µg/ml |
48.4716 |
45.3292 | 45.5836 |
46.461487 |
| 6. | 100 µg/ml |
43.2314 |
50.3828 | 48.5804 |
47.398244 |
| 7. | 50 µg/ml |
50.655 |
52.8331 | 55.6782 |
53.055444 |
| 8. | 25 µg/ml |
57.3508 |
65.3905 | 64.0379 |
62.25972 |
| 9. | 10 µg/ml |
65.3566 |
71.0567 | 75.3943 |
70.602535 |
| 10. | 5 µg/ml |
75.2547 |
80.5513 | 84.858 |
80.221359 |
| 11. | 1 µg/ml |
91.4119 |
91.1179 | 94.3218 |
92.283873 |
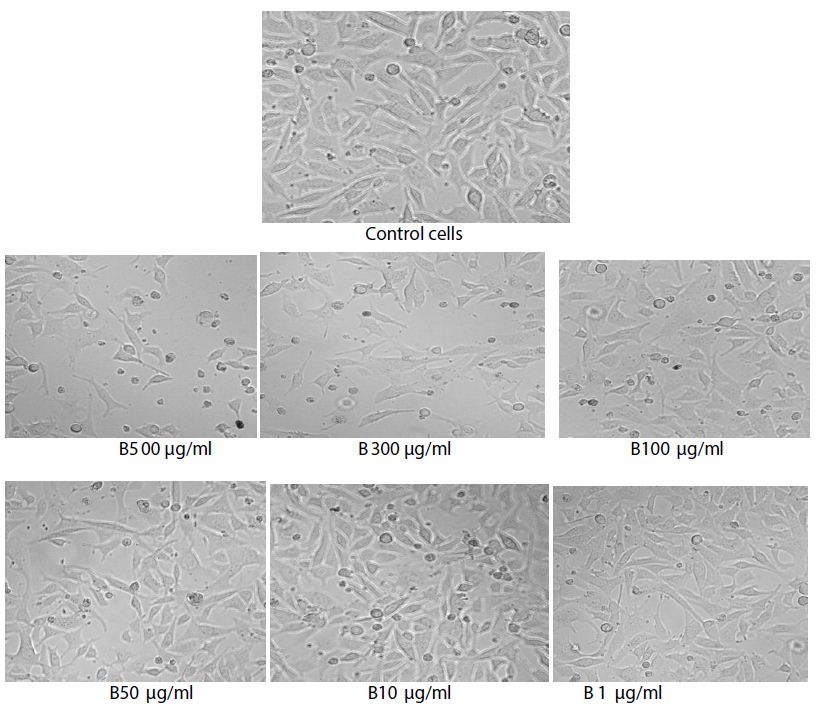
Figure 1: Images of control cells and Acetone extract of C. epigaeus treated cells.
The highest concentration (500 µl/ml) showed 83.1% growth inhibition indicating the extract exhibited pronounced cytotoxicity, with a mean cell viability of 27.45% while 500 µl/ml showed33.32% viability. A gradual increase in cell viability was observed with decreasing concentrations, reaching 76.59% at 1 µl/ml, indicating reduced cytotoxicity at lower doses, Control cells exhibited 100% viability, confirming assay reliability and normal cellular metabolism.
The decrease in optical density at 570 nm reflects a reduction in mitochondrial dehydrogenase activity, suggesting impaired metabolic function and reduced proliferation of cancer cells. Similar dose-dependent cytotoxic effects of plant acetone extracts against epidermoid carcinoma cell lines have been previously reported and attributed to the presence of bioactive secondary metabolites [7-9]
Cytotoxic Activity of Corallocarpus epigaeus Tuber Mediated AuNPs on A-431 Cells
The cytotoxic activity of gold nanoparticles synthesized using the acetone extract of Corallocarpus epigaeus tuber extract was evaluated against the human epidermoid carcinoma cell line (A-431) using MTT assay. The results demonstrated a dose-dependent reduction in cell viability, confirming the antiproliferative efficacy of the biosynthesized AuNPs. (Tables 3, 4 and Figure 2).
Table 3: Cytotoxic property of gold nanoparticle synthesized from C. epigaeus tuber acetone extract.
|
S. No |
Tested sample concentration (μg/ml) |
OD Value at 570 nm (in triplicates) |
||
| 1. | Control |
0.687 |
0.653 |
0.634 |
| 2. | 500 µg/ml |
0.096 |
0.053 |
0.106 |
| 3. | 400 µg/ml |
0.203 |
0.177 |
0.169 |
| 4. | 300 µg/ml |
0.236 |
0.257 |
0.249 |
| 5. | 200 µg/ml |
0.333 |
0.296 |
0.289 |
| 6. | 100 µg/ml |
0.297 |
0.329 |
0.308 |
| 7. | 50 µg/ml |
0.348 |
0.345 |
0.353 |
| 8. | 25 µg/ml |
0.394 |
0.427 |
0.406 |
| 9. | 10 µg/ml |
0.449 |
0.464 |
0.478 |
| 10. | 5 µg/ml |
0.517 |
0.526 |
0.538 |
| 11. | 1 µg/ml |
0.628 |
0.595 |
0.598 |
Table 4: Percentage Viability acetone extract mediated AuNPs on A-431 cell line.
|
S. No |
Tested sample concentration (μg/ml) | Cell viability (%) (in triplicates) |
Mean Value (%) |
||
| 1. | Control |
100 |
100 | 100 |
100 |
| 2. | 500 µg/ml |
13.9738 |
8.11639 | 16.7192 |
12.936476 |
| 3. | 400 µg/ml |
29.5488 |
27.1057 | 26.6562 |
27.770193 |
| 4. | 300 µg/ml |
34.3523 |
39.3568 | 39.2744 |
37.661173 |
| 5. | 200 µg/ml |
48.4716 |
45.3292 | 45.5836 |
46.461487 |
| 6. | 100 µg/ml |
43.2314 |
50.3828 | 48.5804 |
47.398244 |
| 7. | 50 µg/ml |
50.655 |
52.8331 | 55.6782 |
53.055444 |
| 8. | 25 µg/ml |
57.3508 |
65.3905 | 64.0379 |
62.25972 |
| 9. | 10 µg/ml |
65.3566 |
71.0567 | 75.3943 |
70.602535 |
| 10. | 5 µg/ml |
75.2547 |
80.5513 | 84.858 |
80.221359 |
| 11. | 1 µg/ml |
91.4119 |
91.1179 | 94.3218 |
92.283873 |
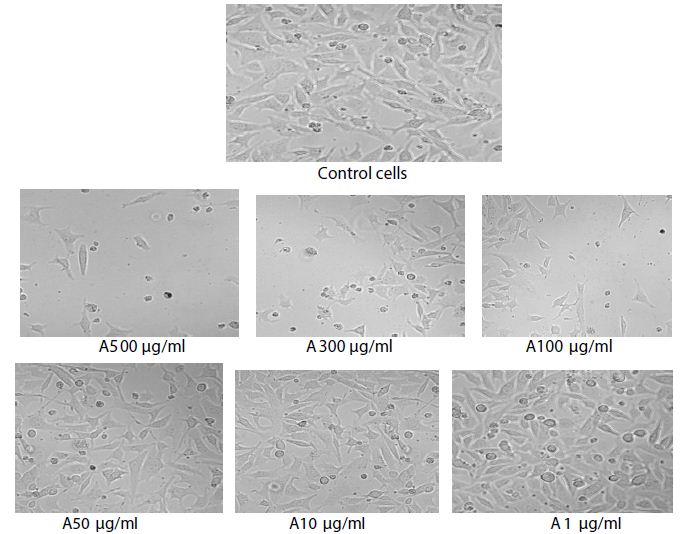
Figure 2: Images of control cells and gold nanoparticles synthesized from C. epigaeus against treated cells.
The highest concentration (500 µl/ml) the AuNPs induced pronounced cytotoxicity, with mean cell viability of 12.94% and 27.77% respectively. A gradual increase in cell viability was observed with decreasing concentrations reaching 92.28% viability at 1 µl/ml, indicating minimal cytotoxicity at lower doses. The untreated control cell maintained 100% viability, confirming the reliability of the assay conditions.
The observed reduction in optical density values at 570 nm directly correlates with decreased mitochondrial activity, reflecting compromised cellular metabolism and loss of viable cells. Similar concentration dependent cytotoxic effects have been reported for plant-based AuNPs against various cancer cell lines, including, skin, breast and cervical cancer, attributed to the enhanced nanoparticle- cell interactions and intracellular accumulation [9,10]
IC50 Determination of Corallocarpus epigaeus Acetone Extract and Corallocarpus epigaeus – Mediated Gold Nanoparticles on A-431 Cells
The cytotoxic activity of acetone extract and gold nanoparticles synthesized using Corallocarpus epigaeus tuber was quantified against the human epidermoid carcinoma A-431 cell line using MTT assay. Dose- response data were analyzed by non-linear regression of log(inhibitor) versus normalized response using a variable-slope model.
The analysis revealed that Corallocarpus epigaeus mediated AuNPs exhibited IC50 value of 57.97 µl/ml, whereas the acetone extract showed a higher IC50 value of 84.67 µg/ml, indicating greater potency of the nanoparticle formulation (Table 5). The AuNPs showed a moderate negative Hill slope (-0.6962) suggesting a gradual inhibitory effect, while the acetone extract showed a steeper slope (-0.9403), reflecting a sharper but less potent response. Both exhibited high goodness-of- fit values (R2>0.9177), confirming the reliability of the concentration dependent inhibitory response. The 95% confidence interval for the IC50 ranged from 45.15 to 74.43 µl/ml (AuNPs) and IC50 ranged from 70.00-102.4 µl/ml, confirming the accuracy of tailored model. Gold nanoparticle reduced the IC50 by~32% indicating enhanced cytotoxic efficacy compared to the crude acetone extract. Compared to the reports on crude plant extracts, the IC50 value obtained in the present study within the biologically relevant range and comparable to acetone extract of medicinal plants reported to exert anticancer activity against skin cancer cell lines [11,12].
Table 5: Comparative IC50 analysis.
|
log(inhibitor) vs. normalized response — Variable slope |
||||
|
Acetone Extract of Corallocarpus epigaeus tuber |
Acetone Extract mediated AuNPs |
|||
| Best-fit values | ||||
| LogIC50 |
1.928 |
1.763 |
||
| HillSlope |
-0.9403 |
-0.6962 |
||
| IC50 |
84.67 |
57.97 |
||
| Std. Error | ||||
| LogIC50 |
0.04036 |
0.05301 |
||
| HillSlope |
0.07893 |
0.06188 |
||
| 95% Confidence Intervals | ||||
| LogIC50 |
1.845 to 2.010 |
1.655 to 1.872 |
||
| HillSlope |
-1.102 to -0.7787 |
-0.8229 to -0.5695 |
||
| IC50 |
70.00 to 102.4 |
45.15 to 74.43 |
||
| Goodness of Fit | ||||
| Degrees of Freedom |
28 |
28 |
||
| R square |
0.9420 |
0.9177 |
||
| Absolute Sum of Squares |
1770 |
2064 |
||
| Sy.x |
7.950 |
8.585 |
||
| Number of points | ||||
| Analyzed |
3 |
30 | 3 |
30 |
The comparative results of the study clearly demonstrate the nanomedicine advantage of Corallocarpus epigaeus mediated gold nanoparticles over the acetone extract alone. While the crude extract exhibited notable cytotoxic activity against A-431 cells, incorporation into gold nanoparticles significantly enhanced efficacy, as evidenced by a lower IC50 values and greater maximal growth inhibition. The enhancement can be attributed to several factors intrinsic to nanoparticles-based delivery systems. In contrast crude acetone extract rely on passive diffusion and may undergo metabolic degradation, limiting their intracellular bioavailability. Gold nanoparticles offer increased surface area for phytochemical adsorption, improved cellular uptake through endocytosis and prolonged intracellular retention, all of which contribute to enhanced anticancer activity [12-14]. Moreover, AuNPs are known to induce oxidative stress and mitochondrial dysfunction in cancer cells, thereby amplifying apoptosis-related signaling pathways [15]. Importantly the observed dose-dependent response and acceptable goodness-of-fit values indicate reliable and reproducible cytotoxic effects. The higher cell viability observed at lower concentrations further suggests a favorable therapeutic opening, an essential consideration for translational nanomedicine. Rather replacing plant-based therapeutics, this study demonstrates that nano structuring of traditional medicinal plant extracts can significantly improve their biological performance, thereby bridging ethnopharmacology and modern medicine. The comparative evaluation clearly demonstrates that the acetone extract of Corallocarpus epigaeus exhibits intrinsic anticancer activity its nano formulation as gold nanoparticles significantly enhances cytotoxic potency, reduces the effective dose (IC50) and improves biological activity against A-431 cells. These findings support the use of Corallocarpus epigaeus as a valuable bioresource for green nano- medicine based cancer therapeutics.
Conclusion
The present study provides convincing evidence that gold nanoparticles synthesized using Corallocarpus epigaeus tuber acetone extract exhibit significantly enhanced cytotoxic activity against human epidermoid carcinoma A-431 cells compared to the crude extract. The reduction in IC50 value and increased growth inhibition highlight the potential of Plant-mediated AuNPs as a promising nanomedicine platform for cancer therapy. More experimental work similar to In- vivo studies are necessary to support clinical translation.
Acknowledgement
The authors sincerely acknowledge the Management for providing seed money to carry out this research work and the Principal and Dean of Research and Development, for their constant support and encouragement. We also thank Tribiotech for providing the necessary analytical facilities and technical support required for the successful completion of the analytical work.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Ferrari M (2005) Cancer nanotechnology: opportunities and Nature Reviews Cancer 5: 161-171. [crossref]
- Peer D, Karp JM, Hong S, Farokhzad OC, Margalit R, et (2020) Nanocarriers as an emerging platform for cancer therapy. Nano-Enabled Medical Applications 23: 61-91. [crossref]
- Dykman LA, Khlebtsov NG (2011) Gold nanoparticles in biology and medicine: recent advances and prospects. Acta Naturae (англоязычная версия) 3: 34-55. [crossref]
- Iravani S (2011) Green synthesis of metal nanoparticles using Green Chemistry 13: 2638-2650.
- Singh H, Du J, Singh P, Yi TH (2018) Ecofriendly synthesis of silver and gold nanoparticles by Euphrasia officinalis leaf extract and its biomedical Artificial Cells, Nanomedicine, and Biotechnology 46: 1163-1170. [crossref]
- Nadkarni AK (2007) KM Nadkarni’s Indian materia medica: with Ayurvedic, Unani-tibbi, Siddha, allopathic, homeopathic, naturopathic & home remedies, appendices & indexes. Popular Prakashan.
- Fulda S, Debatin KM (2004) Targeting apoptosis pathways in cancer Current Cancer Drug Targets 4: 569-576. [crossref]
- Greenwell M, Rahman PK (2015) Medicinal plants: their use in anticancer International Journal of Pharmaceutical Sciences and Research 6.
- Rajeshkumar S, Bharath LV (2017) Mechanism of plant-mediated synthesis of silver nanoparticles–a review on biomolecules involved, characterisation and antibacterial Chemico-Biological Interactions 273: 219-27. [crossref]
- Ahmed S, Ahmad M, Swami BL, Ikram S (2016) A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: a green expertise. Journal of Advanced Research 7: 17-28. [crossref]
- Cragg GM, Newman DJ (2013) Natural products: a continuing source of novel drug Biochimica et Biophysica Acta (BBA)-General Subjects 1830: 3670-3695. [crossref]
- Kuppusamy P, Ichwan SJ, Al-Zikri PN, Suriyah WH, Soundharrajan I (2016) et In vitro anticancer activity of Au, Ag nanoparticles synthesized using Commelina nudiflora L. aqueous extract against HCT-116 colon cancer cells. Biological Trace Element Research 173: 297-305. [crossref]
- Yeh YC, Creran B, Rotello VM (2012) Gold nanoparticles: preparation, properties, and applications in Nanoscale 4: 1871-1880.
- Arvizo RR, Rana S, Miranda OR, Bhattacharya R, Rotello VM, et al. (2025) Mechanism of anti-angiogenic property of gold nanoparticles: role of nanoparticle size and surface Nanomedicine: Nanotechnology, Biology and Medicine 7: 580-587. [crossref]
- Khlebtsov N, Dykman L (2011) Biodistribution and toxicity of engineered gold nanoparticles: a review of in vitro and in vivo Chemical Society Reviews 40: 1647-1671. [crossref]