Abstract
Background: Walking recovery is a key concern for post-stroke patients, with up to 46% of first-ever stroke survivors initially unable to walk and 40% of all post-stroke patients requiring rehabilitation. Regaining a more physiological walking pattern can lead to improved gait performance and, consequently, greater independence in activities of daily living.
Materials and methods: 18 post-stroke patients with drop foot completed a treatment program comprising a neurostimulation-assisted walking device (BTL WALK). Functional gait measurements, including the 10-Meter Walk Test (10MWT), 6-Minute Walk Test (6MWT), Stair Climb Test (SCT), and Manual Muscle Grading scale (MMT) were assessed both before and after the program. Additionally, the Barthel Index was used to evaluate the impact of the treatment on independence in daily activities.
Results: Statistically significant improvements were observed in all monitored parameters, with changes of -17.7% in the 10-Meter Walk Test (10MWT), 14.6% in 6-Minute Walk Test (6MWT), -37.0% in Stair Climb Test (SCT), 8.86% in Barthel Index, and 27.8% in Manual Muscle testing scale (MMT). No significant relationship was found between any parameter or its change and the time since stroke onset.
Conclusions: This study highlights the potential of BTL WALK training in mitigating drop foot in post-stroke patients. Improvements in walking speed, distance, and stair-climbing enhanced independence in daily activities and increased muscle strength, serving as a key motivational factor for continued rehabilitation, especially for patients several years post-stroke.
Keywords
Stroke, Drop foot, Neurostimulation-assisted walking, Gait training, Independence in daily activities
Introduction
Stroke remains a leading cause of mortality and long-term disability, with up to 40% of survivors requiring active rehabilitation [1]. Lower extremity motor impairment is present in 44.1% of individuals experiencing a first-ever stroke, and 46.0% are initially unable to walk [2]. Walking recovery is often one of the first concerns raised by post-stroke patients when consulting medical staff, highlighting its critical role in rehabilitation [3]. Among those who regained some walking ability within 3 to 5 days post-stroke, 76.4% were discharged to home care, compared to only 21% of non-ambulatory patients [1]. Walking is not only a key factor in determining discharge to home care but also a fundamental component in restoring independence in daily life [3].
Up to 20% of stroke survivors experience drop foot, a condition characterized by an impaired ability to dorsiflex the foot during the swing phase of gait. This persistent distal weakness in the hemiparetic leg may be associated with weakness of the anterior muscles, spasticity of the posterior leg muscles, or both. Additionally, the condition can be further complicated by the development of plantar-flexion contracture at the ankle [4]. Drop foot leads to disability by promoting undesirable compensatory movement patterns, restricting mobility, and increasing the risk of falls [4,5]. One potential treatment for drop foot in individuals with hemiparesis is Electrical Stimulation (ES) applied to the peroneal nerve and the tibialis anterior muscle [5].
The most common therapeutic strategies for drop foot are ankle-foot orthosis (AFO) and electrical stimulation [6]. Ankle-foot orthoses (AFOs) are frequently prescribed for individuals with drop foot to maintain ankle neutrality and enhance limb clearance during the swing phase [4,7]. However, this intervention presents several limitations. Restricted ankle mobility associated with AFO use may contribute to contracture development. Furthermore, AFOs do not actively promote dynamic function or facilitate motor recovery [4,6]. Challenges in sit-to-stand transitions and patient discomfort are also commonly reported [4]. Electrical stimulation presents an alternative therapeutic modality for drop foot, targeting branches of the common peroneal nerve distal to the knee. In contrast to AFOs, ES permits physiological ankle range of motion and facilitates active dorsiflexion and eversion without mechanical constraint [4,6]. Clinical evidence indicates that ES, by unrestricted ankle motion, may offer superior functional outcomes compared to AFOs, particularly in complex environments characterized by inclines, uneven terrain, and pedestrian traffic [6,7].
Studies evaluating the immediate effects of ES during therapy have reported positive impacts on walking quality. However, research investigating its long-term effectiveness has yielded divergent results [8,9]. Guzel et al. observed statistically significant improvements in gait quality following a combined conventional physiotherapy, ES, and balance-weighting rehabilitation program in post-stroke patients. Similarly, Street et al. reported positive outcomes in individuals with multiple sclerosis [10,11]. In both patient populations, significant improvements were also observed following ES therapy when compared to the use of AFO [12,13].
This study aims to assess the impact of BTL WALK therapy on post-stroke patients. The research will evaluate the effects on gait parameters, lower limb disability, and the influence of time since stroke on individual outcomes [14,15].
Materials and Methods
The study was conducted at the Hamzova Léčebna Luže-Košumberk rehabilitation center between April and December 2025. Its design adhered to the ethical principles of the 1975 Declaration of Helsinki, as adopted by the Convention on Human Rights and Biomedicine of the Council of Europe and endorsed by the General Assembly of the World Medical Association [17].
Inclusion criteria required participants to be first-time stroke survivors, medically stable, with no cognitive impairment, and presenting with lower extremity involvement in the form of drop foot. Exclusion criteria included febrile conditions, cachexia of any etiology, tuberculosis or other bacterial infections, suspected malignancy, bleeding disorders, menses, the presence of electronic or metal implants in the treatment area, skin inflammation, trophic skin changes, irritated or damaged skin, cardiovascular diseases, sensation disorders, lower limb fractures, dislocations, or conditions adversely affected by motion, electroanalgesia without a confirmed pain etiology, psychopathological syndromes, and inflammatory conditions in the treatment area.
Participants underwent a 4-week treatment program consisting of neurostimulation-assisted walking (BTL WALK) therapy, administered five times per week. Functional assessments were performed before and after treatment, including the 10-Meter Walk Test (10MWT), the 6-Minute Walk Test (6MWT), and the Stair Climb Test (SCT) to evaluate mobility. Muscle strength was assessed by the Manual Muscle Test (MMT). Additionally, participants completed the Barthel Index questionnaire, which assessed their subjective level of disability related to gait impairment.
Before therapy, the patient was seated with the hip, knee, and ankle positioned at a 90° angle. The BTL WALK Pro device (BTL Industries, Ltd.) was placed on the inner calf, with electrode positioning adjusted to stimulate the common peroneal nerve that innervates the peroneal muscles and the tibialis anterior muscle (Figure 1). Following the initiation of therapy, stimulation intensity was gradually increased until ankle dorsiflexion was achieved. The electrode was then further adjusted to maintain the foot in a neutral position, preventing excessive eversion or inversion. The patient was encouraged to walk, with continuous adjustments made to electrode placement and stimulation intensity based on their response. Once optimized, a walking session was conducted, during which drop foot pathology was mitigated through muscle stimulation.

Figure 1: Image depicting the placement of BTL WALK on the inner calf to treat drop foot. Permission was granted by BTL Industries, Ltd.
To assess objective gait improvements, several functional tests were performed. The 10MWT evaluated gait speed, where the patient was instructed to walk at a comfortable pace over a 10-meter distance. The total time taken to complete the walk was recorded in seconds. No physical assistance was provided unless required for safety reasons. Timing began when the first foot crossed the start line and stopped when the first foot crossed the end line at 10 meters [18].
The 6MWT assessed functional exercise capacity by measuring the maximum distance the patient could walk in six minutes. The test was conducted in a long, straight hallway with cones marking each end of the walking course. The patient was instructed to wear comfortable clothing and non-slip footwear and walk at their own pace. They were allowed to rest if needed, but the timer continued running, and the total distance walked in meters was recorded [19].
Lower limb strength and balance were assessed using the SCT, which measured the time taken to ascend a standard flight of 12–14 steps, each 17–19 cm high. The patient was instructed to climb the stairs at their normal pace, using handrails only if necessary for balance. Timing began at the first step and stopped upon reaching the top, with results recorded in seconds [20].
Strength of the tibialis anterior, peronei, soleus, and gastrocnemius muscles was measured using the 0, meaning no contraction, to 5, meaning normal strength against full resistance, scale of the MMT. Muscle tests were performed by trained examiners following standardized limb positions and stabilization to minimize substitution and optimize repeatability.
In addition to these functional tests, the Barthel Index was used to evaluate the patient’s level of functional independence in performing ten essential daily activities. The assessment was conducted through direct observation and patient interviews, with activities scored based on the level of independence—categorized as independent, partially independent, or dependent. The total score ranged from 0 to 100, with higher values indicating greater independence [21].
The sample size was determined based on the capacity of the rehabilitation center and the minimum required sample size calculation. This calculation was conducted using 6MWT data from a previous study on post-stroke patients, assuming a study power of 80% and an estimated effect size of 15 meters [22,23]. The minimum required sample size of nine patients was increased to 20 to account for facility capacity and potential dropout risk.
For statistical processing and data evaluation, a customized script was developed in the Matlab environment. The data were initially analyzed using the Shapiro-Wilk test to assess normality. Except for the 10-Meter Walk Test (10MWT), the assumption of normal distribution was rejected for all parameters. Consequently, these data were analyzed using the Wilcoxon Signed-Rank test and presented as median values with interquartile range (IQR). In contrast, the 10MWT data, which met the criteria for normality, were compared using a T-test and reported as mean values with Standard Deviation (SD). The Pearson correlation coefficient was used to determine if there was a correlation between the time since stroke onset and the therapy outcome. P-values less than 0.05 were considered indicative of statistical significance.
Results
A total of 18 post-stroke patients, 16 men and 2 women, with a mean age of 70.22 ± 12.70 years and a mean time of symptom duration of 8.7 years. No adverse events occurred during the program, and none of the patients experienced any discomfort that would have hindered their participation. The therapy was generally well tolerated and resulted in improved walking function in the majority of patients.
Monitored parameters showed statistically significant improvements, mean score change, and mean within-patient percentage change in Table 1. After the treatments, 83.33% of patients improved in 10 MWT, 88.89% of patients improved in 6MinWT, 83.33% of patients improved in SCT, and 50% of patients improved in BI. These patients showed mean improvements in the aforementioned parameters of -18.41%, +23.06%, -28.46%, and 21.61%. The changes in MMT measurements showed the number of patients that improved after the therapies was 22.22% in the tibialis anterior, 33.33% in the peronei muscle, 27.78% in the soleus muscle, and 27.78% in the gastrocnemius muscle. The average change for the MMT score was 27.78%.
Table 1: Results of functional tests and functional independence questionnaire obtained before and after the treatment program. P values lower than 0.05 were considered statistically significant.
|
Before |
After | Diff | %Diff | %Mean within-patient percentage change | P (0.05) | |
| Mean (SD) | Mean (SD) | Mean score change (SD) |
T-test Other: Wilcoxon Signed Rank test |
|||
| 10MWT (seconds) |
24.86 (13.50) |
20.44 (10.43) | -4.43 (5.41) | -17.77% | -14.20% | 0.006 |
|
6MWT (meters) |
166.17 (75.37) | 190.50 (80.67) | 24.33 (39.10) | 14.64% | 19.53% |
0.002 |
| SCT (seconds) |
68.00 (63.54) |
42.83 (20.55) | -25.17 (51.53) | -37.01% | -22.69% | 0.006 |
|
Barthel Index |
75.28 (11.13) | 81.94 (5.18) | 6.67 (8.91) | 8.86% | 10.80% |
0.035 |
IQR: Interquartile range, Diff: difference, 10MWT: 10-Meter Walk Test, 6MWT: 6-Minute Walk Test, SCT: Stair Climb Test.
The bar graph in Figure 2 visually represents the improvements achieved. The 6MWT and Barthel Index showed increases in the distance covered and the degree of independence in activities of daily living, respectively. Conversely, both the 10MWT and SCT showed decreases in the time needed to walk a set distance or climb a flight of stairs. These results all suggest an overall improvement in lower limb function over the course of the BTL WALK treatment program.
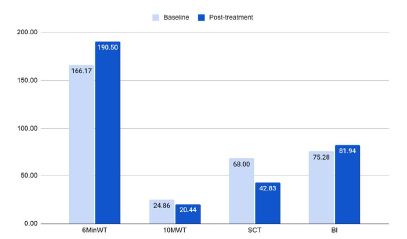
Figure 2: The baseline and post-treatment values of the 10-Meter Walk Test (10MWT), the 6-Minute Walk Test (6MWT), and the Stair Climb Test (SCT) to evaluate mobility.
Table 2 displays the Pearson correlation coefficients, which show the relationship between the time since stroke onset and individual outcome measures. The results indicate that time since stroke onset did not have a statistically significant effect on any of the indicators.
Table 2: P-value and Pearson correlation coefficient between time since stroke onset and individual outcome measures obtained before starting and after completing the treatment program, and the difference between them.
|
Time vs 10MWT |
Time vs 6MWT | Time vs SCT | Time vs Barthel Index | |||||
| P (0.05) | r | P (0.05) | r | P (0.05) | r | P (0.05) |
r |
|
| Before |
0.928 |
0.027 | 0.662 | -0.128 | 0.355 | -0.268 | 0.578 | 0.163 |
|
After |
0.873 | 0.047 | 0.57 | -0.166 | 0.591 | -0.157 | 0.757 |
-0.091 |
| Diff |
0.934 |
0.024 | 0.711 | -0.109 | 0.358 | 0.266 | 0.336 |
-0.278 |
Time: Time since stroke onset, r: Pearson correlation coefficient, Diff: difference, 10MWT: 10-Meter Walk Test, 6MWT: 6-Minute Walk Test, SCT: Stair Climb Test.
Discussion
The present study documents statistically significant changes in post-stroke patients’ gait as a result of a rehabilitation program based on BTL WALK gait training. These conclusions are supported by existing evidence, both in terms of statistical significance and the extent of the changes achieved [16,22,24]. Comparing the recalculated 10MWT values for speed (before: 0.51 m/s, after: 0.59 m/s) with similar studies, it is evident that the absolute values of gait speed differ significantly across publications. This may be due to variations in measurement methodology (patient instruction, use of aids, treadmill, etc.) and the different composition of patients in terms of indication, age, time from onset of stroke, severity, or brain damage, etc. Patients in the acute and subacute post-stroke phase tend to be more limited in movement and their gait speed reaches an average of around 0.25 m/s [25]. The current research includes patients who are a few weeks post-stroke, but also those who have been suffering from the consequences of stroke for many years. Correlation analysis did not reveal a statistically significant relationship between time since stroke and any outcome measure or its change. The main influence on walking limitation is likely the extent of brain damage [26].
The observed improvements in walking speed, functional exercise capacity, balance, and lower limb strength, as demonstrated through various functional assessments, can be further explained by the underlying mechanism of BTL WALK in post-stroke drop foot rehabilitation. BTL WALK facilitates neuromuscular activation by delivering electrical impulses to the dorsiflexor muscles, inducing muscle contraction, and enabling ankle dorsiflexion during the swing phase of gait. In post-stroke patients experiencing drop foot or impaired foot clearance, this timely and coordinated muscle activation enhances gait performance, contributing to a more physiological walking pattern [27]. Moreover, intensive and prolonged gait training incorporating ES promotes motor relearning, which, over time, can lead to sustained improvements in gait mechanics, potentially reducing the need for assistive devices [28]. This device can significantly affect the quality of life of patients in everyday life if they use the device as a compensatory aid.
Improvements observed in functional gait assessments were also positively reflected in subjective evaluations of activities of daily living. Although these changes were less pronounced compared to speed and distance-based assessments, they nonetheless reached statistical significance. Future research should consider utilizing more sensitive assessment tools capable of detecting the impact of even minor gait pattern deviations on an individual’s independence in daily activities.
The conclusions of this study must be interpreted in consideration of its limitations. The primary limitation is the absence of a control group, which would have enabled a direct comparison with patients receiving conventional therapy or no treatment, thereby accounting for the potential effects of spontaneous motor recovery. Additionally, the small sample size may have influenced the findings, particularly in the analysis of the impact of time since stroke onset. Notably, patient inclusion was not stratified based on time post-stroke, which adds a distinctive aspect to the study but also presents a limitation. Future research should aim to investigate specific subgroups of stroke survivors, particularly those several years post-stroke, as clinical evidence in this population remains limited.
Despite its limitations, this study provides valuable insights, particularly by demonstrating the positive effects of BTL WALK therapy on gait in patients undergoing rehabilitation not only in the early post-stroke phase but also several years after stroke onset. The finding that mobility and independence in daily activities can still be improved in chronic stroke survivors may serve as an important motivational factor for continued rehabilitation efforts.
Conclusions
The present study highlights the potential of BTL WALK training in mitigating drop foot in post-stroke patients. The observed improvements in walking speed, distance covered, and stair-climbing ability contributed to enhanced independence in daily activities, which may serve as a crucial motivational factor for continued rehabilitation, particularly for individuals undergoing therapy several years post-stroke. The therapy is an effective intervention for rehabilitating patients with limited mobility and gait impairments resulting from brain damage, promoting functional recovery and improved movement patterns.
References
- van de Port IG, Kwakkel G, Schepers VP, Lindeman E (2006) Predicting mobility outcome one year after stroke: a prospective cohort study. Journal of Rehabilitation Medicine 38: 218-223. [crossref]
- Louie DR, Simpson LA, Mortenson WB, Field TS, Yao J, et al. (2022) Prevalence of Walking Limitation After Acute Stroke and Its Impact on Discharge to Home. Physical Therapy 102. [crossref]
- Preston E, Ada L, Dean CM, Stanton R, Waddington G (2011) What is the probability of patients who are nonambulatory after stroke regaining independent walking? A systematic review. International Journal of Stroke: Official Journal of the International Stroke Society 6: 531-540. [crossref]
- O’Dell MW, Dunning K, Kluding P, Wu SS, Feld J, et al. (2014) Response and prediction of improvement in gait speed from functional electrical stimulation in persons with poststroke drop foot. PM & R: The Journal of Injury, Function, and Rehabilitation 6: 587-601. [crossref]
- Lairamore CI, Garrison MK, Bourgeon L, Mennemeier M (2014) Effects of functional electrical stimulation on gait recovery post-neurological injury during inpatient rehabilitation. Perceptual and Motor Skills 119: 591-608. [crossref]
- Li X, Li H, Liu Y, Liang W, Zhang L, et al. (2024) The effect of electromyographic feedback functional electrical stimulation on the plantar pressure in stroke patients with foot drop. Frontiers in Neuroscience 18: 1377702. [crossref]
- Nevisipour M, Honeycutt CF (2020) The impact of ankle-foot-orthosis (AFO) use on the compensatory stepping response required to avoid a fall during trip-like perturbations in young adults: Implications for AFO prescription and design. Journal of Biomechanics 103: 109703. [crossref]
- Böhm H, Döderlein L, Dussa U, Ch (2020) Functional electrical stimulation for foot drop in the upper motor neuron syndrome: does it affect 3D foot kinematics during the stance phase of walking? Fuß & Sprunggelenk 18: 115-124.
- Robison J, Gibbons R, Achelis D, Bent B, Wajda D, et al. (2022) Augmenting gait in a population exhibiting foot drop with adaptive functional electrical stimulation. medRxiv27.22273623.
- Güzel Şükran, Umay Ebru, Ozturk Erhan, ÇAKCI Aytül (2022) The Efficiency of Functional Electrical Stimulation and Balance-Weighted Rehabilitation Therapy in Stroke Patients with Foot-Drop: A Pilot Study. Fiziksel Tıp ve Rehabilitasyon Bilimleri Dergisi 25: 1-10.
- Street T, Taylor P, Swain I (2015) Effectiveness of functional electrical stimulation on walking speed, functional walking category, and clinically meaningful changes for people with multiple sclerosis. Archives of Physical Medicine and Rehabilitation 96: 667-672. [crossref]
- Renfrew LM, Paul L, McFadyen A, Rafferty D, Moseley O, et al. (2019) The clinical- and cost-effectiveness of functional electrical stimulation and ankle-foot orthoses for foot drop in Multiple Sclerosis: a multicentre randomized trial. Clinical Rehabilitation 33: 1150-1162. [crossref]
- Wang He, Li Yaning, Yu Shaohong (2025) Effect of electrical stimulation in the treatment on patients with foot drop after stroke: a systematic review and network meta-analysis. Journal of Stroke and Cerebrovascular Diseases 34: 108279. [crossref]
- Friðriksdóttir R (2020) Functional Electrical Stimulation as a treatment option for foot drop, with a focus on gait velocity: A retrospective quality improvement study.
- Bethoux F, Rogers HL, Nolan KJ, Abrams GM, Annaswamy TM, et al. (2014) The effects of peroneal nerve functional electrical stimulation versus ankle-foot orthosis in patients with chronic stroke: a randomized controlled trial. Neurorehabilitation and Neural Repair 28: 688-697. [crossref]
- Lairamore CI, Garrison MK, Bourgeon L, Mennemeier M (2014) Effects of functional electrical stimulation on gait recovery post-neurological injury during inpatient rehabilitation. Perceptual and Motor Skills 119: 591-608. [crossref]
- Hendriks A (1997) Council of Europe-Convention for Protection of Human Rights and Dignity of the Human Being with Regard to the Application of Biology and Biomedicine: Convention on Human Rights and Biomedicine. Kennedy Institute of Ethics Journal 7: 277-290.
- Peters DM, Fritz SL, Krotish DE (2013) Assessing the reliability and validity of a shorter walk test compared with the 10-Meter Walk Test for measurements of gait speed in healthy, older adults. Journal of Geriatric Physical Therapy 36: 24-30. [crossref]
- Agarwala P, Salzman SH (2020) Six-Minute Walk Test: Clinical Role, Technique, Coding, and Reimbursement. Chest 157: 603-611. [crossref]
- Gagliano-Jucá T, Li Z, Pencina KM, Traustadóttir T, Travison TG, et al. (2020) The Stair Climb Power Test as an Efficacy Outcome in Randomized Trials of Function Promoting Therapies in Older Men. The Journals of Gerontology. Series A, Biological Sciences and Medical Sciences 75: 1167-1175. [crossref]
- Collin C, Wade DT, Davies S, Horne V (1988) The Barthel ADL Index: a reliability study. International Disability Studies 10: 61-63. [crossref]
- Jongbae Ch, Sungryoung M, Jongeun Y (2019) Effect of Electric Stimulation Training on Walking Ability of Patients with Foot Drop after Stroke. J Int Acad Phys Ther Res, Journal of International Academy of Physical Threrapy Research 10: 1903-1906.
- Charan J, Biswas T (2013) How to calculate sample size for different study designs in medical research?. Indian Journal of Psychological Medicine 35: 121-126. [crossref]
- Wilkinson Hart IA (2016) Team flow: The missing piece in performance [Thesis for the degree of Doctor of Philosophy, University of Southhampton]. Southhampton University Research Repository.
- Cheng DK, Dagenais M, Alsbury-Nealy K, Legasto JM, Scodras S, et al. (2021) Distance-limited walk tests post-stroke: A systematic review of measurement properties. NeuroRehabilitation 48: 413-439. [crossref]
- Scott JN, Buchan AM, Sevick RJ (1999) Correlation of neurologic dysfunction with CT findings in early acute stroke. The Canadian journal of neurological sciences. Le Journal Canadien des Sciences Neurologiques 26: 182-189. [crossref]
- Shin HE, Kim M, Lee D, Jang JY, Soh Y, et al. (2022) Therapeutic Effects of Functional Electrical Stimulation on Physical Performance and Muscle Strength in Post-stroke Older Adults: A Review. Annals of Geriatric Medicine and Research 26: 16-24. [crossref]
- Dantas MTAP, Fernani DCGL, Silva TDD, Assis ISA, Carvalho AC, et al. (2023) Gait Training with Functional Electrical Stimulation Improves Mobility in People Post-Stroke. International Journal of Environmental Research and Public Health 20: 5728. [crossref]