Abstract
Background: Primary hypothyroidism, encompassing overt and subclinical forms, induces profound disturbances in systemic metabolism and increases the susceptibility to atherosclerotic cardiovascular disease (ASCVD). South-Asian populations are particularly vulnerable owing to a unique metabolic phenotype characterized by higher visceral adiposity and insulin resistance; however, data on this population remain scarce.
Objective: This study aimed to determine the prevalence and pattern of metabolic syndrome (MetS) and its components among adults with primary hypothyroidism in a tertiary care center in Bangladesh and to explore evidence of metabolic rewiring through cardiometabolic profiling.
Methods: In this cross-sectional study, 59 adults (≥18 years) with overt or subclinical hypothyroidism were enrolled from endocrine and allied medicine units at BIRDEM General Hospital, Dhaka. Anthropometric, biochemical, and thyroid parameters were assessed using standardized protocols. MetS was defined according to the International Diabetes Federation (IDF, 2005) criteria for South Asians. Data were analyzed descriptively and stratified according to hypothyroid subtype and treatment status.
Results: The cohort (mean age 52.2 ± 13.6 years; 76.3% female) exhibited a MetS prevalence of 76.3%. Central obesity (94.9%), low HDL-C level (86%), and elevated fasting glucose level (95%) were the most frequent abnormalities. Treated overt hypothyroid patients had significantly lower total cholesterol (161.9 ± 38.7 mg/dL vs. 200.8 ± 75.3 mg/dL), triglycerides (187.5 ± 89.2 mg/dL vs. 233.0 ± 126.9 mg/dL), and TSH (3.44 ± 2.19 µIU/mL vs. 47.05 ± 34.98 µIU/ mL) compared to untreated cases.
Conclusion: The remarkably high prevalence of MetS among adults with hypothyroidism in Bangladesh suggests a state of metabolic rewiring driven by thyroid hormone deficiency. Integrating metabolic screening with thyroid management may mitigate the long-term cardiometabolic risk in this high- burden population.
Keywords
Primary hypothyroidism, metabolic syndrome, South Asia, metabolic rewiring, dyslipidemia, insulin resistance, cardiometabolic risk
Introduction
Primary hypothyroidism, a common endocrine disorder, disrupts core metabolic processes, affecting lipid metabolism, glucose homeostasis, and cellular energy regulation [1,2]. Globally, overt hypothyroidism affects approximately 4-10% of adults, whereas subclinical hypothyroidism (SCH) affects up to 15%, with even higher rates reported in South Asia, regions marked by socioeconomic and nutritional transitions with resultant complex metabolic profiles [3,4]. Community-based studies highlight overt hypothyroidism prevalence between 5 and 7% and SCH prevalence up to 20% in South Asian adults [5,6].
Thyroid hormones T4 (thyroxine) and T3 (triiodothyronine) regulate the basal metabolic rate by modulating mitochondrial oxidative phosphorylation and thermogenesis, lipid catabolism, and carbohydrate metabolism [1,2]. Deficiency impairs mitochondrial function, diminishes hepatic LDL receptor gene expression, and attenuates peripheral lipoprotein lipase activity, resulting in increased circulating LDL cholesterol, triglycerides, and total cholesterol [7,8]. Additionally, hypothyroidism disrupts glucose metabolism by hindering GLUT4 translocation in insulin-sensitive tissues, reducing endothelial nitric oxide production, and promoting insulin resistance, hypertension, and arterial stiffness [9,10].
Metabolic syndrome (MetS), a constellation of central obesity, dyslipidemia, hyperglycemia, and hypertension, increases the risk of coronary heart disease and shares pathophysiological features with hypothyroidism [11]. Studies have reported that 35–65% of hypothyroid patients meet the MetS criteria, but estimates vary regionally due to genetic background, ethnicity, diet, and lifestyle [12-14]. South Asians demonstrate unique metabolic traits, including heightened visceral fat accumulation and lower HDL-C, even at normal BMI, intensifying insulin resistance and cardiovascular risks.
Despite increasing awareness of thyroid-metabolic syndrome interplay, data from South Asian populations, where dual burdens of hypothyroidism and metabolic syndrome premortem significantly compound cardiovascular disease risk, remain scarce. This study aimed to elucidate the prevalence and patterns of MetS components in Bangladeshi patients with hypothyroidism and assess the cardiometabolic impact of levothyroxine replacement.
Materials and Methods
Study Design and Setting
This cross-sectional analytical study was conducted at the Department of Endocrinology and Allied Medicine, BIRDEM General Hospital, Dhaka, Bangladesh, between January 2018 and August 2019. This study investigated adults with biochemically confirmed primary hypothyroidism, both overt and subclinical, who attended routine outpatient and inpatient endocrine services. The protocol was reviewed and approved by the Institutional Review Board of the Bangladesh Diabetic Association (BADAS) under ethical approval number BADAS-ERC/2018/05-02. The study adhered to the Declaration of Helsinki (World Medical Association, 2013), and all participants provided written informed consent before enrolment.
Participants and Eligibility
Patients were recruited consecutively according to predefined inclusion and exclusion criteria.
Inclusion Criteria:
- Adults (≥18 years).
-
Diagnosed with primary hypothyroidism, either overt (elevated TSH with low FT4) or subclinical (elevated TSH with normal FT4) [1].
Exclusion Criteria
Secondary hypothyroidism, pregnancy, chronic renal or hepatic disease, corticosteroid or oral contraceptive use, or other systemic illnesses likely to affect metabolic parameters [7]
Data Collection and Measurements
Demographic data (age and sex), anthropometry (height, weight, and waist circumference [WC]), and blood pressure (BP) were recorded using standardized protocols. BMI was calculated as weight (kg)/height² (m²) and classified by Asian-specific cut-offs: normal < 23.0, overweight 23.0–26.9, obese ≥ 27.0 kg/m² [14].
After overnight fasting (8–12 h), venous blood samples were drawn for the following:
- Fasting plasma glucose (FPG)
- Lipid profile (TC, TG, HDL-C, LDL-C)
- Thyroid profile (TSH, FT4)
- Aspartate aminotransferase (AST)
All analyses were performed at the BIRDEM Central Biochemistry Laboratory using automated analyzers with internal quality control.
Definition of Variables
Metabolic syndrome (MetS) was defined according to the International Diabetes Federation (IDF, 2005) criteria: central obesity (WC ≥ 90 cm men; ≥ 80 cm women) plus any two of the following: (1) TG ≥ 150 mg/dL, (2) HDL-C < 40 mg/dL men/< 50 mg/dL women, (3) BP ≥ 130/85 mmHg or treated hypertension, and (4) FPG ≥ 100 mg/ dL or known diabetes.
Thyroid status was classified as:
- Overt hypothyroidism: elevated TSH with low
- Subclinical hypothyroidism: elevated TSH with normal FT4 [8].
Statistical Analysis
Data were analyzed using SPSS v26.0 (IBM Corp., Armonk, NY, USA). Continuous variables were expressed as mean ± SD and compared using independent sample t-tests or one-way ANOVA. Categorical variables are expressed as frequencies (%) and compared using χ² tests. p < 0.05. Subgroup analyses compared overt versus subclinical hypothyroidism and treated versus untreated overt cases.
Ethical Considerations
The study protocol was approved by the Institutional Review Board of the Bangladesh Diabetic Association (BADAS) (Ref: BADAS-ERC/2018/05-02). All procedures conformed to the ethical standards of the Declaration of Helsinki (World Medical Association, 2013). Written informed consent was obtained from each participant, and anonymity was ensured during data analysis and reporting.
Result
Demographic and Clinical Characteristics
A total of 59 adults diagnosed with primary hypothyroidism were included; the mean age was 52.2 ± 13.6 years, with females representing 76.3% of the cohort. Among these, 34 patients (57.6%) had overt hypothyroidism, whereas 25 (42.4%) had subclinical hypothyroidism. Within the overt group, 28 (82.4%) patients were on levothyroxine therapy, whereas six (17.6%) were newly diagnosed and untreated at study entry. The mean BMI was 26.8 ± 4.1 kg/m², and central obesity was highly prevalent (93.2%), indicative of the typical visceral adiposity in this population.
Prevalence of Metabolic Syndrome and Its Components
Metabolic syndrome (MetS), as defined by the International Diabetes Federation criteria adapted for South Asians, was diagnosed in 45 of 59 patients, corresponding to a prevalence of 76.3%, underscoring the metabolic burden associated with thyroid hormone deficiency.
The breakdown by hypothyroid subtype showed that MetS was present in 62.2% of patients with overt hypothyroidism and 57.1% of individuals with subclinical hypothyroidism, suggesting that metabolic derangements manifest before overt hormonal insufficiency.
Key individual MetS components were markedly prevalent:
- Central obesity was observed in 9% of patients.
- Elevated fasting plasma glucose levels were measured in 95%.
- Low HDL cholesterol affected 86% of
- Hypertriglyceridemia was present in approximately 68–76%.
- Hypertension, defined as a systolic/diastolic BP ≥ 130/85 mmHg or treatment, had an impact of approximately 85%.
The clustering of these components illustrates profound metabolic dysfunction associated with thyroid hormone deficiency (Figure 1).
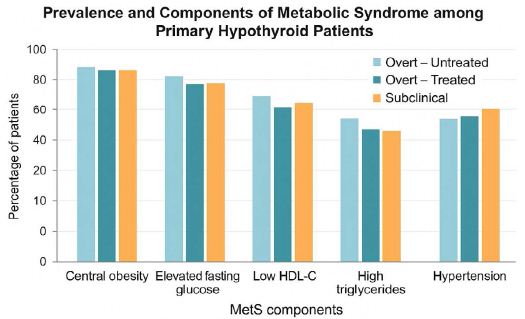
Figure 1: Prevalence and Components of Metabolic Syndrome among Primary Hypothyroid Patients. A clustered bar chart displaying percent prevalence of MetS components across overt treated, overt untreated, and subclinical subgroups.
Impact of Levothyroxine Therapy on Biochemical Parameters
Subgroup analysis comparing treated versus untreated overt hypothyroid patients revealed significant metabolic improvements attributable to hormone replacement therapy.
- Lipid Parameters: Treated patients showed significantly lower total cholesterol levels (161.9 ± 7 mg/dL) compared to untreated (200.8 ± 75.3 mg/dL) (p < 0.05). Triglycerides were also reduced in treated patients (187.5 ± 89.2 mg/dL versus 233.0 ± 126.9 mg/dL), although the difference was statistically significant (p ≈ 0.06).
- Thyroid Function Tests: TSH levels were dramatically lower among treated patients (3.44 ± 19 µIU/mL) than among untreated cases (47.05 ± 34.98 µIU/mL, p < 0.001). Correspondingly, FT4 levels were considerably higher (14.57 ± 3.28 pmol/L versus 7.71 ± 1.41 pmol/L, p < 0.001), indicating effective thyroid hormone restoration.
Subclinical hypothyroid patients exhibited intermediate lipid and thyroid profiles, suggesting an early onset of metabolic disturbances preceding overt hypothyroidism (Figure 2).
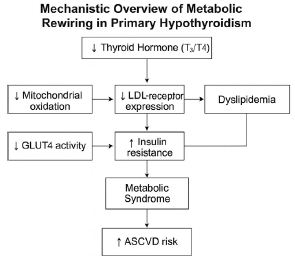
Figure 2: The interconnected metabolic pathways affected by primary hypothyroidism. The deficiency of T₃/T₄ triggers a cascade of metabolic disturbances, from mitochondrial energy inefficiency to lipid retention and insulin signaling impairment. This metabolic rewiring underpins the high prevalence of MetS observed in this study and mechanistically links endocrine dysfunction to cardiometabolic disease progression. T₃/ T₄ – Triiodothyronine and Thyroxine; LDL receptor – Low-Density Lipoprotein Receptor; GLUT4 – Glucose Transporter Type 4; ASCVD – Atherosclerotic Cardiovascular Disease.
Age-Stratified Metabolic Trends
The prevalence of MetS increased significantly with advancing age across hypothyroid subtypes, peaking in those older than 60 years for overt hypothyroidism and between 51 and 60 years for subclinical hypothyroidism. Age-correlated metabolic factors included increased triglyceride and decreased HDL-C levels (p < 0.05). Importantly, levothyroxine treatment was associated with favorable metabolic trends in all age groups, highlighting the restorative potential of early hormone replacement therapy.
Discussion
Elevated Metabolic Syndrome Burden
This study found an exceptionally high (76%) prevalence of MetS among Bangladeshi adults with hypothyroidism, exceeding many reports and implying a major cardiometabolic risk accumulation. The high MetS rates in subclinical hypothyroidism suggest that metabolic injury begins early.
Thyroid-Driven Metabolic Rewiring
Thyroid hormone deficiency disrupts mitochondrial oxidative function and β-oxidation, resulting in hepatocellular triglyceride accumulation and potentially metabolically associated fatty liver disease (MAFLD) [2,7,15] Suppression of hepatic LDL receptor expression and lipoprotein lipase activity elevates cholesterol and triglyceride levels. Levothyroxine therapy reverses this metabolic bottleneck.
Insulin Resistance and Vascular Effects
Impaired GLUT4 translocation and suppressed hepatic insulin signaling contribute to insulin resistance, as confirmed by widespread fasting hyperglycemia (95%). Vascular stiffness and hypertension (~85%) are associated with decreased endothelial nitric oxide production [10]. Metabolomic biomarkers such as branched-chain amino acids and acyl-carnitines indicate mitochondrial dysfunction within this milieu [16].
South Asian Predisposition and Aging
The South Asian adiposity phenotype—excess visceral adipose and low HDL-C at lower BMI—exacerbates the metabolic risk multiplicatively with hypothyroidism. Aging furthers insulin resistance through thyroid decline and adipocyte senescence (Kumar et al., 2020).
Levothyroxine as Therapeutic Modifier
Hormone replacement improves thyroid function and partially corrects lipid and vascular function abnormalities, suggesting that early intervention may prevent progression to atherosclerotic cardiovascular disease and MAFLD [17].
Outlook: Precision Endocrine-Metabolic Care
The integration of multi-omics approaches, such as lipidomics, metabolomics, and transcriptomics, may pinpoint thyroid hormone– sensitive metabolic networks, allowing tailored hormone replacement therapy targeting metabolic phenotypes rather than TSH alone [18,19].
Conclusion
Primary hypothyroidism in South-Asian adults is tightly linked to widespread metabolic reprogramming, manifested by a high prevalence of metabolic syndrome and atherogenic dyslipidemia. Both the overt and subclinical forms exhibit early insulin resistance and lipid abnormalities, highlighting that metabolic injury begins before overt hormonal failure. Levothyroxine therapy ameliorated these disturbances, indicating that thyroid hormone replacement exerts dual endocrine-metabolic benefits.
From a translational standpoint, incorporating cardiometabolic screening into routine thyroid evaluation and exploring molecular biomarkers of metabolic rewiring may reduce the ASCVD burden in hypothyroid populations.
Limitations and Future Directions
The present study had several limitations. First, its cross- sectional design restricts its ability to infer causality between thyroid dysfunction and metabolic outcomes. Second, the sample size was modest and recruited from a single tertiary-care center, which may limit the generalizability to broader South-Asian populations. Third, due to resource constraints, insulin resistance markers (e.g., HOMA- IR) and inflammatory biomarkers were not measured, which could have provided a deeper mechanistic insight.
Future research should include longitudinal and interventional studies to clarify the temporal relationship between thyroid dysfunction and metabolic syndrome development. Integrating omics-based analyses such as metabolomics and lipidomics could further elucidate the molecular underpinnings of metabolic rewiring in hypothyroidism.
Acknowledgments
The authors gratefully acknowledge the support of the Department of Endocrinology, BIRDEM General Hospital, Dhaka, Bangladesh for providing laboratory and logistic assistance. Appreciation was also extended to all the participants for their voluntary contributions.
Author Contributions
Shirazum Munira (1): Conceptualization, data collection, and primary manuscript drafting.
Nafisa Abedin (1,2): Data interpretation, figure and table preparation, critical revision of the manuscript, and corresponding author responsibilities.
Farhad Ahmed (3), Md. Shayedat Ullah (3), Indira Roy (4), Rhyma Karim (4), Umme Sumyia (4), Khaled Hassan (5), Afroza Begum (5), and Md. Arifuzzaman (5): Provided technical support, contributed to data analysis, and assisted in manuscript revision.
Conflict of Interest Statement
The authors declare no conflicts of interest and confirm that the study was conducted independently, without financial or institutional bias.
References
- Brent GA and Davies TF (2011) Hypothyroidism and thyroiditis. In: De Groot LJ, et (Eds.), Williams Textbook of Endocrinology. Elsevier.
- Mullur R, Liu YY and Brent GA (2014) Thyroid hormone regulation of Physiol. Rev. [crossref]
- Canaris GJ, Manowitz NR, Mayor G and Ridgway EC (2000) The Colorado thyroid disease prevalence study. Intern. Med. [crossref]
- Vanderpump MPJ (2011) The epidemiology of thyroid Br. Med. Bull [crossref]
- Paul R, et (2006) Prevalence of hypothyroidism in a Bangladeshi community. Bangladesh J. Med. Biochem.
- Shantha GPS, et al. (2009) Prevalence of hypothyroidism in metabolic syndrome patients and its association with insulin resistance. Indian J. Endocrinol. Metab
- Duntas LH and Brenta G (2012) The effect of thyroid disorders on lipid Med. Clin. North Am. [crossref]
- Peppa M, Betsi G and Dimitriadis G (2011) Lipid abnormalities and cardiometabolic risk in Eur. J. Endocrinol.
- Singla R, Gupta Y, Khemani M and Aggarwal S (2010) Thyroid disorders and insulin resistance: An emerging Indian J. Endocrinol. Metab.
- Taddei S, et al. (2003) Impaired endothelium-dependent vasodilation in subclinical J. Clin. Endocrinol. Metab. [crossref]
- Alberti KGMM, Zimmet P and Shaw J (2006) The metabolic syndrome—a new worldwide definition. Lancet 366(9491). [crossref]
- Kota SK, Kota SK, Jammula S, Meher LK and Modi KD (2012) Prevalence of metabolic syndrome in hypothyroidism and effect of replacement J. Clin. Endocrinol. Metab.
- Koirala S, Sharma R and Singh S (2016) Metabolic syndrome in hypothyroid patients: A cross-sectional study from J. Clin. Diagn. Res.
- Misra A and Khurana L (2011) Obesity-related non-communicable diseases: South Asians Caucasians. Nutr. [crossref]
- Eslam M, Sanyal AJ and George J (2020) MAFLD: A consensus-driven proposed nomenclature for metabolic associated fatty liver Gastroenterology. [crossref]
- Kim HJ, Lee SY and Park TJ (2023) Serum metabolomic signatures in hypothyroidism reveal mitochondrial Front. Endocrinol.
- Pérez-Méndez O, Hernández A and Mora-Martínez S (2022) Integrating metabolic screening in thyroid Endocr. Metab. Res.
- Sun J, Li X and Zhou H (2023) Multi-omics profiling of thyroid hormone-regulated Front. Mol. Biosci.
- Zhao L, et (2021) Integrative lipidomics reveals thyroid hormone-driven metabolic remodeling. Nat. Metab.