Abstract
Aims: Neuroinflammation complicates traumatic brain injury predisposing to long-lasting neurologic impairment. The aim of the present study was to test whether parenteral administration a small peptide mimic (SN..8) of the receptor- activating- region of the human serotonin 2A receptor (1-, 3- and 5-days) after traumatic brain injury suppresses hippocampal inflammation in the rat compared to a scrambled peptide sequence of the same eight amino acids.
Methods: Adult male Sprague-Dawley rats were exposed to lateral fluid percussion (LFP)-induced, traumatic brain injury (TBI) vs. sham injury. An identical 2 mg/kg concentration of SN..8 vs LD..8 (scrambled peptide) was administered via intraperitoneal route 1-, 3- and 5-days after injury. Two weeks post injury, the bilateral hippocampal, dorsal and ventral brain regions were examined by RT-PCR for altered gene expression. Comparisons were made between TBI vs sham injury; and active vs scrambled peptide treatment.
Results: Two weeks after injury, the novel SN..8 peptide (vs scrambled peptide) significantly reduced (more than 3-fold) CD68 mRNA relative expression in ventral hippocampus in adult male Sprague-Dawley rats subjected to TBI (N=22) (1.65 ± 0.83 vs 5.27 ± 4.1; P=0.012).
Conclusion: These results suggest that the neuroprotective effects of SN..8 peptide may be due in part to its ability to substantially suppress subacute inflammation in the ventral hippocampus.
Introduction
Neuroinflammation complicates traumatic brain injury predisposing to long-lasting neurologic impairment. The serotonergic receptor system is highly expressed in certain brain regions, dentate gyrus, ventral and dorsal hippocampus involved in the normal regulation of mood and spatial learning and memory [1]. A novel peptide identical to the receptor- activating- region of the human serotonin 2A receptor (SN..8) suppressed long-lasting 5HT2AR activation in vitro [2], and had neuroprotective effects on spatial learning and memory paradigms in adult male rats exposed to traumatic brain injury (lateral fluid percussion) [3]. Since TBI is associated with inflammation, here we tested whether peripheral injections of the 5HT2A receptor peptide immediately following TBI can suppress subacute hippocampal inflammation.
Methods
Animals
All procedures were conducted in accordance with the NIH Guide for the Care and Use of Laboratory Animals and were approved by the Institutional Animal Care and Use Committee of the Veterans Affairs Medical Center (East Orange, New Jersey). Male SD rats (11-14-weeks- old) n=33 were obtained from Charles River Laboratories (Kingston, NY) and were individually housed with modest enrichment (wooden block). Rats were provided ad libitum access to food and water and maintained in a 12 h light/dark cycle with lights on at 0700. At approximately 17 weeks of age, rats underwent surgery (craniectomy) and injury.
Peptides
The linear synthetic peptide, corresponding to a fragment of the serotonin 2a receptor, SCLLADDN (SN..8) and a scrambled version LASNDCLD (LD.8) were both synthesized at Lifetein, Inc. (Hillsborough, NJ). Each peptide was provided as the hydrochloride salt and had purity > 95%. The lyophilized peptides were stored (in the presence of dessicant) at −40 degrees C prior to use. Before each experiment, peptide was reconstituted fresh in sterile saline at the indicated concentration.
Injections
Peptide (SN..8 or LD.8) was dissolved in sterile saline (2 mg/kg) and administered via intraperitoneal (IP) route 1-, 3- and 5-days after TBI vs sham injury.
Surgery/Injuries
Craniectomy and delivery of a pressure wave (lateral fluid percussion) procedures were carried out as previously reported [3,4].
Tissue Extraction and Sectioning
Whole brains were extracted at 2 weeks post-injury and snap frozen in 2-methylbutane. Tissue micropunches (1 mm diameter) were extracted from the dorsal and ventral hippocampus, bilaterally.
RT-PCR
Total RNA was extracted from the tissue using the RNAeasy Plus Mini Kit (Qiagen, Germantown, MD). The RNA concentration and A260/280 ratio of samples will be measured using a Nanodrop spectrophotometer (ThermoFisher Scientific). Total RNA was reverse transcribed, and cDNA was stored at -20 C. Taqman primer/probe sets (ThermoFisher Scientific) were used to assess changes in gene expression of the following genes: CD68 (Rn01495634_g1), Vimentin (Rn00667825_m1), BAX (Rn01480161_g1), Bcl2 (Rn99999125_m1), PCNA (Rn01514538_g1), Htr2A (Rn00568473_m1).
RT-PCR was performed using a LightCycler 380 (Roche Diagnostics); each reaction contained 3 ul of cDNA, 10 ul Taqman Universal PCR master mix, 1 ul of appropriate Taqman primer/probe set and 6 ul of dH20. The crossing point (Cp) value was determined for each primer/probe using the 2nd derivative maximum method. Fold change values were calculated using the delta-delta CT method using the highest Cp value as the reference value. All gene expression values were normalized to Actin β (Rn00667869_m1). Data for each target gene were assayed in duplicate and averaged; Cp values were normalized to the mean of the housekeeping gene Actin β.
Fold changes between samples for each gene product were calculated as follows:
2(Sample with highest Cp value for target gene-individual sample’s Cp value for target gene)/2(Sample with highest Cp value for Actin β-individual sample’s Cp value for Actin β)
Statistics
Two-way ANOVA was used to test for a main effect of drug (SN..8 vs scrambled peptide), injury (TBI vs sham) or drug x injury interaction on relative gene expression. Post-hoc testing was performed using Fischer’s least significant difference (LSD) test.
Results
Mean apnea time and mean righting reflex time were significantly longer in rats subjected to TBI vs sham-injury (Table 1). There was no statistically significant difference in mean apnea time or mean righting reflex time following lateral fluid percussion in rat subgroups randomized to treatment with SN..8 vs scrambled peptide injections on days 1, 3 and 5 after injury. The mean peak pressure (PSI, pounds per square inch) applied during the fluid percussion wave did not differ significantly between rats treated with SN..8 vs. scrambled peptide following TBI (Table 1).
Table 1: Acute measures following mild TBI (lateral fluid percussion) vs sham injury.
|
Cohort 1&2 |
Apnea (s) | Righting Reflex (s) | Startle |
Peak PSI |
| Males, TBI, SN.8 (n=11) |
16.90 ± 2.58 |
416.81 ± 27.226 | Yes |
15.56 ± 0.79 |
| Males, Sham, SN.8 (n=6) |
N/A |
50.16 ± 14.736 | N/A |
N/A |
| Males, TBI, Scrambled (n=11) |
12.2 ± 1.21^ |
447.27 ± 29.078^* | Yes |
18.40 ± 1.36^ |
| Males, Sham, Scrambled (n=5) |
N/A |
79.6 ± 19.314^ | N/A |
N/A |
^P > 0.05 for mean apnea or righting reflex time: TBI, SN.8 vs TBI scrambled peptide.
^Peak PSI was not significantly different for TBI, SN.8 vs TBI, scrambled peptide groups.
*P< 0.001 mean righting reflex time was significantly longer for TBI vs sham groups.
Gene Expression Changes in the Dorsal and Ventral Hippocampus Two Weeks Post-Injury
Gene expression of six genes: Bax, BcL2, Vim, 5HT2AR, PCNA, and CD68 was evaluated in pooled bilateral dorsal and ventral hippocampal brain regions from sham vs. TBI rats treated with either SN..8 vs scrambled peptide. CD68 is a marker of activated microglia in the brain. There was a significant interaction (injury x drug) effect (F(1,29)=4.0: P=0.05) in CD68 mRNA relative expression in ventral hippocampus. In post-hoc analysis, among scrambled peptide-treated rats (N=11), mean CD68 relative expression was significantly higher (P=0.011) in TBI vs. sham-injury consistent with injury-induced chronic neuro-inflammation [5]. In TBI rats (N=22) mean CD68 relative expression was significantly higher (P=0.0032) in scrambled vs. SN..8 peptide-treated rats (Figure 1) consistent with a significant anti-inflammatory effect of the SN..8 peptide (Figure 2).
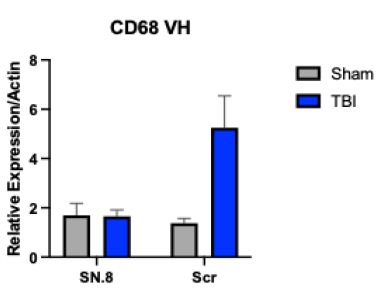
Figure 1: In mTBI (vs sham-injured) rats, SN.8 vs scrambled peptide was associated with significantly reduced gene expression of CD68 two weeks after injury.
^TBI (SN.8 vs Scr) P =0.0032;
^^Scr: TBI vs Sham P =0.011; N=33 rats; 22 TBI, 11 sham injury
Proliferating cell nuclear antigen is a marker of cell proliferation in non-post- mitotic cells in the central nervous system. The cell types potentially include: DG neural progenitor cells, astrocytes and microglia. There was a significant interaction (injury x drug) effect (F(1,29)=11.5: P=0.0023) on PCNA mRNA relative expression in ventral hippocampus. In post-hoc analysis, among scrambled peptide-treated rats (N=11), mean PCNA relative expression was significantly higher (P=0.005) (Figure 2) in TBI vs. sham-injury consistent with injury- induced cell proliferation. In TBI rats (N=22) mean PCNA relative expression was significantly higher (P=0.016) in scrambled vs. SN..8 peptide-treated rats (Figure 2) consistent with a significant anti- proliferative effect of the SN..8 peptide on unknown cell population(s).
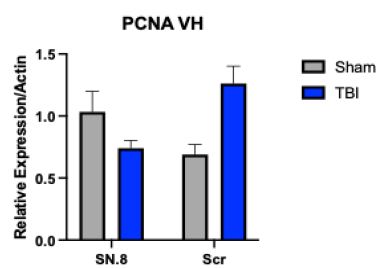
Figure 2: TBI (vs sham injury) was associated with a significant increase in PCNA gene expression in the ventral hippocampus in scrambled peptide-treated rats (Figure 2, P=0.005). In SN.8-treated rats, however, TBI (vs sham injury) was associated with significantly reduced PCNA gene expression (Figure 2, P =0.0016) in the ventral hippocampus.
Scr: TBI vs sham P=0.005
TBI: SN.8 vs Scr; P=0.0016
In two-way ANOVA there was no significant main or interaction effect of injury, drug treatment on relative gene expression of PCNA or CD68 in dorsal hippocampus. And there was no significant main or interaction effect of injury, drug treatment in ventral or dorsal hippocampal regions on relative expression of any of the other four genes (Blc2, Bax, 5ht2ar, Vim) tested (data not shown). Yet there was a borderline significant main effect of drug treatment F(1,29)=3.9; P=0.056) on the Vim relative gene expression in ventral hippocampus favoring reduced Vim expression in TBI rats treated with SN..8 vs. scrambled peptide P=0.023 (Figure 3).
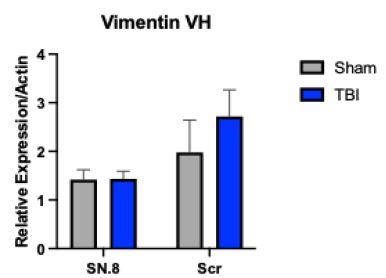
Figure 3: Serotonin 2A receptor peptide (SN.8) (vs scrambled peptide) treatment significantly reduced mean relative level of Vimentin gene expression in the ventral hippocampus at 14 days post injury in TBI rats (P=0.023).
Discussion
Dentate gyrus neural progenitor cells (NPC), astrocytes and microglia may all increase in the hippocampus following TBI, however the acute increase in DG NPCs in rodents following TBI was reported to be transient (lasting less than 7 days) [6,7]. For this reason, it is unlikely that increased PCNA mRNA expression in the hippocampus (14 days post injury) primarily reflects NPC proliferation. The PCNA mRNA expression change in the ventral hippocampus mirrored changes in CD68 mRNA expression. Since CD68 is a marker of activated microglia, this suggests that SN..8 (vs scrambled peptide) significantly suppressed proliferation in activated microglia in the ventral hippocampus in TBI rats. This may be consistent with a recent report that a 5HT2AR antagonist mitigated pathology in a mouse model of Alzheimer’s disease via reduced microglial dysfunction [8]. The 5HT2A receptor is expressed on both microglia and astrocytes [9,10]. Activation in both kinds of cell types is associated with increased expression of pro-inflammatory cytokines, i.e. neuroinflammation. Vimentin gene encodes intermediate filaments which increase in activated astrocytes. We cannot exclude the possibility that SN..8 may have also suppressed astrocyte activation in TBI rats even though the effect of drug (SN.8 vs scrambled) peptide treatment only reached borderline statistical significance.
The present data are the first to suggest that SN..8 had a (subacute) significant anti-inflammatory effect in ventral hippocampus in rats subjected to mild TBI (LFP). The striking absence of a similar significant effect(s) in the dorsal hippocampus is of interest. The mammalian ventral hippocampus receives a denser serotonergic input compared to the dorsal hippocampus [11]. The 5HT2AR expression (in situ hybridization) was also reported to be ventrally-enriched in CA3c subregion of the hippocampus [12]. Together with DG, CA3c subregion is involved in spatial memory processing [13] in which the CA3c supports the DG in modulating pattern separation [14].
In our prior report, systemic administration of SN..8 (2 mg/kg) (vs. scrambled) significantly prevented early loss of behavioral pattern separation (BPS)(2 weeks post injury) in adult male Sprague-Dawley rats exposed to lateral fluid percussion (LFP)-induced, traumatic brain injury (TBI). Taken together, the SN..8-mediated suppression of neuroinflammation (in the ventral hippocampus which includes CA3c neurons enriched in 5HT2AR) may play a permissive role in preventing loss of behavioral pattern separation two weeks post-injury [3]. Neuroinflammation was reported to cause hyperexcitability in DG [15] which is predicted to interfere with pattern separation. These data provide the proof-of-principle that a novel 5HT2AR mimic (SN.8), which is thought to act by preventing harmful long-lasting receptor activation (leading to neuron death) has additional significant neuroprotective, anti-inflammatory effect(s).
Acknowledgement
Supported in part by a grant from the New Jersey Commission on Brain Injury Research NJCBIR PIL022 to MBZ; and a grant from the Department of Veterans Affairs, Office of Research and Development, Technology Transfer Program (Wash, DC) to MBZ.
Dr. Zimering is Inventor on a patent (Assigned to the U.S Department of Veterans Affairs) with claims about the neuroprotective effects of the serotonin 2A receptor peptide reported here.
References
- Xu T and Pandey SC (2000) Cellular localization of serotonin (2A) (5HT (2A)) receptors in the rat Brain Res Bull 51: 499-505. [crossref]
- Zimering MB (2019) Autoantibodies in Type-2 Diabetes having Neurovascular Complications Bind to the Second Extracellular Loop of the 5-Hydroxytryptamine 2A Endocrinol Diabetes Metab J [crossref]
- Agbolou XM, Yoe CW, Cominski TP, Zimering MB (2024) Effects of a Serotonin Receptor Peptide on Behavioral Pattern Separation in Sham- vs. Mild Traumatic Brain Injured Endocrinol Diabetes Metab J. [crossref]
- Stamper CE, Cominski TP, Hoisington AJ, Yoe CW, Agbolou XM, Stiritz VA, Interian A, Goodman M, Hazlett EA, Myers CE, Beck KD, Brenner LA (2025) J Neurotrauma.
- Navabi SP, Badreh F, Khombi Shooshtari M, Hajipour S, Moradi Vastegani S, Khoshnam SE (2024) Microglia-induced neuroinflammation in hippocampal neurogenesis following traumatic brain injury. Heliyon.[crossref]
- Komoltsev IG, Tret’yakova LV, Frankevich SO, Shirobokova NI, Volkova AA, Butuzov AV, Novikova MR, Kvichansky AA, Moiseeva YV, Onufriev MV, Bolshakov AP, Gulyaeva NV (2022) Neuroinflammatory cytokine response, neuronal death, and microglial proliferation in the hippocampus of rats during the early period after lateral fluid percussion-induced traumatic injury of the neocortex. Mol Neurobiol. [crossref]
- Clark LR, Yun S, Acquah NK, Kumar PL, Metheny HE, Paixao RCC, Cohen AS, Eisch AJ (2021) Mild Traumatic Brain Injury Induces Transient, Sequential Increases in Proliferation, Neuroblasts/Immature Neurons, and Cell Survival: A Time Course Study in the Male Mouse Dentate Gyrus. Front Neurosci. [crossref]
- Lu J, Zhang C, Lv J, Zhu X, Jiang X, Lu W, Lu Y, Tang Z, Wang J, Shen X (2021) Antiallergic drug desloratadine as a selective antagonist of 5HT2Areceptor ameliorates pathology of Alzheimer’s disease model mice by improving microglial dysfunction. Aging Cell. [crossref]
- Xu, T., and Pandey, S. C. (2000). Cellular localization of serotonin(2A) (5HT(2A)) receptors in the rat Brain Res Bull [crossref]
- Glebov, , Lochner, M., Jabs, R., Lau, T., Merkel, O., Schloss, P., et al. (2015). Serotonin stimulates secretion of exosomes from microglia cells. Glia [crossref]
- Bjarkam CR, Sørensen JC, Geneser FA (2003) Distribution and morphology of serotonin-immunoreactive axons in the hippocampal region of the New Zealand white rabbit. I. Area dentata and hippocampus. Hippocampus. [crossref]
- Tanaka KF, Samuels BA, Hen R (2012) Serotonin receptor expression along the dorsal-ventral axis of mouse Philos Trans R Soc Lond B Biol Sci. [crossref]
- Hunsaker MR, Rosenberg JS, Kesner RP (2008) The role of the dentate gyrus, CA3a,b, and CA3c for detecting spatial and environmental novelty. Hippocampus. [crossref]
- Kesner RP (2013) A process analysis of the CA3 subregion of the Front Cell Neurosci. [crossref]
- Kurki SN, Srinivasan R, Laine J, Virtanen MA, Ala-Kurikka T, Voipio J, Kaila K (2023) Acute neuroinflammation leads to disruption of neuronal chloride regulation and consequent hyperexcitability in the dentate gyrus. Cell Rep. [crossref]