Abstract
In this contribution, we demonstrate that samples from the Zinnwald greisen deposits also contain diamonds that crystallized under crustal conditions. The occurrence of diamond whiskers in quard and lonsdaleite in fluorite opens a wide field of sophisticated research.
Keywords
Raman spectroscopy, Supercritical fluids, Formation of diamonds in crustal regions, Diamond whiskers
Introduction
The study of some samples (quartz, cassiterite, zinnwaldite, fluorite) from the famous tin deposit of Zinnwald (Sn-W-Li deposit at the German/Czech border in the eastern Erzgebirge) has shown by the occurrence of diamond, diamond whiskers, and lonsdaleite, that these normally mantle-type minerals occur in relatively surface-near mineralization and are not uncommon [1-6]. Further studies on the Zinnwald mineralization, including the host granite, showed that the whole palette of carbon minerals is typical and present. In the past, the study of graphite and related minerals was ignored. As a result, many fundamental minerogenetic aspects could, therefore, not be correctly interpreted. The Formation of diamond and SiC whiskers (in quartz and beryl) clearly demonstrates the formation on the spot [1]. That means the formation occurs directly on the Earth’s crust, which is traditionally not possible. Furthermore, the here-described graphite or carbon differs from classic graphite (e.g., from Madagascar) by the extreme Raman band at low frequencies, which, according to Shea and Wall (2012) [7], are radial breathing modes (RBM). Such graphite nanotubes can include and transport a variety of components, including NaSn2F5, boron, and many others. The Formation of diamonds and Co. in the crustal range needs an acceptable explanation. Is the input of supercritical fluids and melts coming from the mantle range into the crust responsible for the exceptional mineralization? Is the transition from the supercritical stage to the critical and under-critical stages connected with pressure shock wave-like cavitation processes? Is the formation of spherical lonsdaleite crystals in fluorite with a diameter of about 40 µm or diamond whiskers in quartz with lengths up to 200 µm so feasible?. The following new observations on the Zinnwald samples, presented here, should help address these questions.
Sample Material and References
Granite Sample
The sample is a halogen-enriched topaz-albite granite from the Zinnwald tin-tungsten mining district (sample ZW212) of the Variscan age [8-10]. A detailed description of this and related samples and the results of fluid and melt inclusion studies can be found in Webster et al. (2004) [11] and Thomas et al. (2005) [12]. At that time, it was already clear that there was a two-stage formation of the granite, at least. The second phase exhibits a high fluorine content in the melt (~6% F). Topaz shows three different morphologic types: fine needles of topaz crystals, intergrowth of topaz, fluorite, and cryolite, as well as isometric topaz crystals [12]. Unconsidered up to now is the frequent appearance of graphite (Figure 1) and also nano- to micro diamonds in these granites. Striking is also the occurrence of spherical REE-rich fluorite crystals in quartz phenocrysts [13], which, together with the obtained pseudo-binary solvus curve (granite melt-water system) and the Lorentzian distribution of certain elements (F, Rb, Cs), demonstrate an input of supercritical fluids or melts from the Earths mantle into the crust [5,6].
Figure 1: Graphite (Gr) in granite from Zinnwald (sample ZW212) [12].
Pegmatite Quartz
Incorporated with the Variscan tin-tungsten mineralization related to the granites are pegmatite α-quartz crystals (up to 20 cm long), which are partially replaced by later β-quartz [1]. Other essential minerals are topaz, cassiterite, zinnwaldite, and fluorite. The last three contain, in part, diamond, diamond whiskers, lonsdaleite, graphite, graphite whiskers, and α-rhombohedral spheres and whiskers of boron partly with boron carbide [14].
Zinnwaldite and Fluorite
For the Zinnwald tin-tungsten deposit, Zinnwald-Cinovec is, according to Baumann et al. (2000), the 3-meter thick pegmatite of the type “Stockscheider” between quartz porphyry and albite granite characteristical. The pegmatite consists of quartz, potassium feldspar, and zinnwaldite. Some smoky quartz crystals up to up to 20 cm long can be found. In the zinnwaldite bundle, there are mostly violet to colorless fluorite crystals enclosed. Fluorite crystals contain lonsdaleite and diamond inclusions [4].
Microscopy and Raman Spectroscopy
We performed all microscopic and Raman spectroscopic studies with a petrographic polarization microscope (BX 43) with a rotating stage coupled with the EnSpectr Raman spectrometer R532 (Enhanced Spectrometry, Inc., Mountain View, CA, USA) in reflection and transmission. The Raman spectra were recorded in the spectral range of 0–4000 cm-1 using an up-to-50 mW single-mode 532 nm laser, an entrance aperture of 20 µm, a holographic grating of 1800 g/mm, and spectral resolution ranging of 4 cm-1. Generally, we used an objective lens with a magnification of 100x: the Olympus long-distance LMPLFLN100x objective (Olympus, Tokyo, Japan). The laser power on the sample is adjustable down to 0.02 mW. The Raman band positions were calibrated before and after each series of measurements using the Si band of a semiconductor-grade silicon single-crystal. The run-to-run repeatability of the line position (based on 20 measurements each) is ± 0.3 cm−1 for Si (520.4 ± 0.3 cm-1) and 0.4 cm-1 for diamond (1332.7 cm-1 ± 0.4 cm-1 over the range of 80–2000 cm-1). The FWHM = 4.26 ± 0.42 cm-1. FWHM is the Full-Width at Half Maximum. We also used a water-clear natural diamond crystal (Mining Academy Freiberg: 2453/37 from Brazil) as a diamond reference (for more information, see Thomas et al., 2023) [15]. The zero-point position for the Raman spectroscopic measurements is shown in Figure 2.
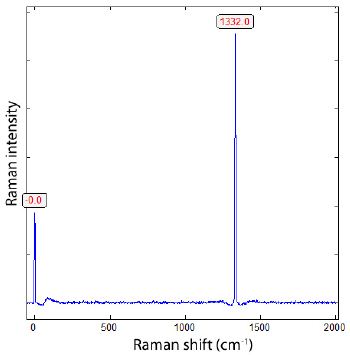
Figure 2: Position of the diamond line during the Raman measurements in the range from -40 to 2015 cm-1 (sample 2453/37 from Brazil). The mean of 20 new measurements is 1332.2 ± 0.4 cm-1.
Reference Material
Diamond and Graphite
As reference material for diamond and graphite, we used well- studied material. The diamond from the Mining Academy Freiberg, which we have used since the start of the Raman spectroscopic studies, is used daily to calibrate the device (see Figure 2). Additionally, for the same purpose, graphite from Madagascar is also used.
Since diamond, lonsdaleite, and graphite are in the central focus of this contribution, we provide the Raman reference spectra of diamond and graphite here (Figures 2 and 3).
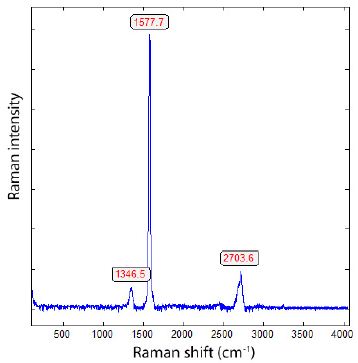
Figure 3: Typical Raman spectrum of small plate-shaped graphite crystals from Madagascar.
Single-Walled Carbon Nanotubes (SWCNTs)
This pure material is from ProGraphite GmbH, Untergriesbach, Germany. We used this reference material because the Raman spectra show strong bands in the low Raman range, unlike graphite crystals from Madagascar. The following features [7] characterize the Raman spectrum of the used single-walled carbon nanotubes as reference: a strong G-band at 1570 cm-1, a weaker D-band at 1332 cm-1, a resonant 2D band at 2650 cm-1, and between about 80 and 500 cm-1 the radial breathing modes (RBMs). Figure 4 shows a typical Raman spectrum of single-walled carbon nanotubes (SWCNT).
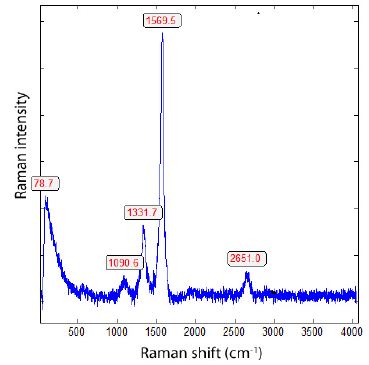
Figure 4: Typical Raman spectrum of single-walled carbon nanotubes (SWCNTs).
Results
Topaz-Albite Granite from the Zinnwald
The granite contains rare cassiterite-brown microcrystals surrounded by a halo of colorless, primary probably β-quartz (see Figure 5). The determination of β-quartz is not simple because the surrounding quartz is α-quartz, and β-quartz transforms fast into α-form. The quartz of the hallo shows extremely high Raman lines at 126.0 and 204.7 cm-1.
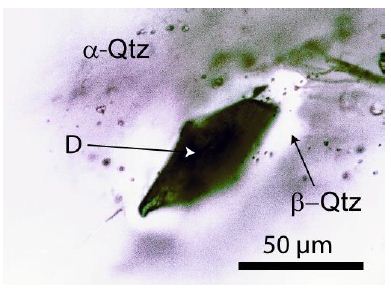
Figure 5: Diamond in granite quartz (sample ZW212) from Zinnwald. The diamond grain is 75 µm deep from the sample surface. The sample is 500 µm thick. D – diamond, α- Qtz – low-quartz, at the trapping probably β-Qtz – high-quartz. Under intense light, the diamond is in the center, cassiterite-brown, and at the rim, blue.
In Figure 5, a diamond crystal in granite quartz is depicted. Finding such crystals is not simple, as the granite sample contains many small, dark crystals, such as graphite. Figure 6 is a typical Raman spectrum of diamond shown – composed of the relatively broad diamond band at about 1326 cm-1 and the typical G-band at 1585 cm-1 of the carbon material. Depending on the isotope composition (13C vs. 12C), the band position of the diamond and the corresponding G-band can vary in a wide range [2]: pure 13C-diamond at 1283.1 cm-1 and pure 12C-diamond at 1332.7 cm-1. The same is the case for graphite: 1519 to 1581 cm-1 for both end members (13C vs 12C) respectively.
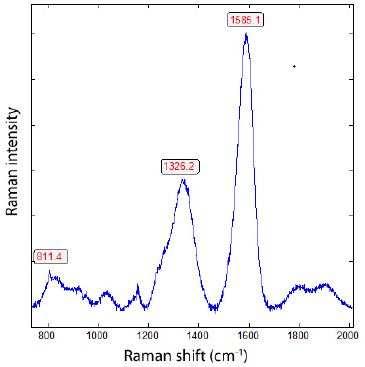
Figure 6: Raman spectrum of diamond in granite from Zinnwald (sample ZW212, Figure 5).
The diamond is partially transformed into carbon (see Figure 7) because the diamond is at high temperatures and low pressure over a long time, not stable [16].
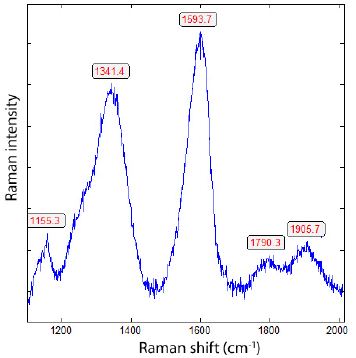
Figure 7: Carbon, transformed from primary diamond (Figure 6) by its ascent with supercritical fluids or melts into the present granite from Zinnwald (sample ZW212).
The direct diamond-related carbon is very rare in the granite sample ZW212. Most carbon is related to spheric or quasi-spheric black globules, often present and then mostly in quartz. Typical Raman spectra are in Figures 6 and 8. Characteristically, for this type of carbon, there are extreme Raman bands in the low-frequency range from about 80 to 400 cm-1. The intensity is so strong that the range from 1200 to 2000 cm-1 is nearby suppressed. Taking only the Raman spectrum in the range between 1200 and 2000 cm-1 reveals the typical carbon band.The extremely high Raman bands are identified as radial breathing modes (RBM) by Shea and Wall (2012).

Figure 8: Raman spectra of graphite in granite from Zinnwald (ZW212), about 30 µm deep in quartz. Because the Raman band of graphite between 80 and 400 cm-1 is exceptionally high (lower Figure), therefore the range of the upper Figure is strongly enlarged by using only the Raman window from about 1200 to 2000 cm-1.
In contrast to crystalline graphite (see Figure 3) from Madagascar, the carbon in the Zinnwald granite shows extremely intense Raman bands in the low-frequency range. It looks like carbon nanotubes (SWCNTs) or MWCNTs (multi-walled carbon nanotubes – see Figure 4 and Shea and Wall (2012). The relatively strong band at 763.4 cm-1 can traced back to fluoroboric acid H[BF4] [12] (Figure 8).
Most carbon spheres in the granite quartz are composed obviously of multi-walled carbon nanotubes filled with different ions (rare alkalis, SnF2, SnO2, H2O, and many see Figure 9).
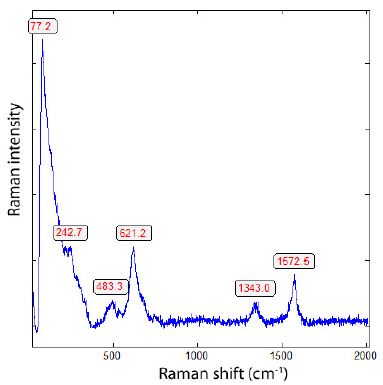
Figure 9: Raman spectrum of SnF2-loaded single-walled carbon nanotubes (SWCNTs). The RBM Raman band at 242.7 cm-1 corresponds to a SWCNT diameter of about 1 nm filled with NaSn2F5, and the Raman bands at 483.3 and 621.2 results from SnO2 [17].
Granite-related Tin-Tungsten Mineralization
Smoky Quartz
The results on smoky quartz are summarized in Thomas (2025a) [1]. Some carbon needles often have small diamond crystals at their tip (deep under the sample surface!). Figure 10a is a Raman spectrum of such carbon needles with a diamond on the tip.
Figure 10b show an about 80 µm long diamond whisker in quartz and the corresponding Raman spectrum. This whisker shows two points that held up. Chaotic processes during the crystal growth of the quartz host are so out of the question.
From the Raman band position, the diamond whisker is composed of 47.4% 13C, and the graphite is composed of 38.5% 13C, respectively.
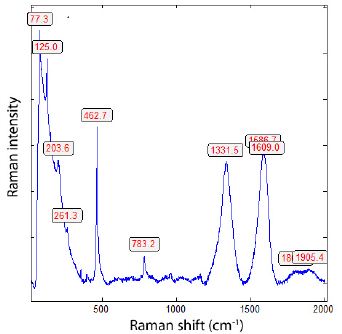
Figure 10 a: Raman spectrum of a diamond-bearing carbon needle in quartz deep under the sample surface.
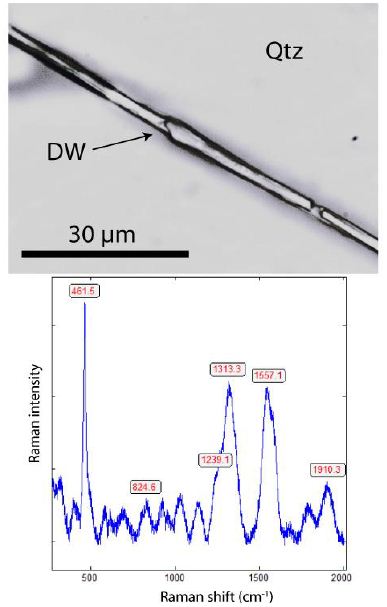
Figure 10 b: Diamond whisker in smoky quartz (upper photomicrograph) and the accompanying Raman spectrum of diamond whisker. The Raman band at 461.5 cm-1 is from the quartz host.
Zinnwaldite
A brief description of the deposits in Zinnwald (Germany) and Cinovec (Czech Republic) is provided by Baumann et al. (2000) [8], Breiter and Qiao et al. (2024). The Li-mica zinnwaldite (~2.2 % Li2O), named after this deposit, is the main constituent of this deposit and represents one of the largest Li-Sn-W greisen deposits in Europe. A careful study of zinnwaldite cleavage crystals reveals, besides small crystals of quartz, cristobalite, scheelite, cassiterite, and K2SnCl6, the presence of graphite grains. Figure 11 is an example of such a crystal with nanodiamonds (Table 1, Figures 11 and 12).
Table 1: Raman spectroscopic data of diamond and related carbon of the diamond in the Zinnwald granite ZW212; measurements on 19 crystals.
|
Sample |
Diamond (cm-1) | FWHM | Carbon (cm-1) |
FWHM |
| ZW212 |
1328.5 ± 4.3 |
63.1 ± 18.0 | 1578.7 ± 7.2 |
57.5 ± 13.5 |

Figure 11: Boron carbide-diamond crystal in zinnwaldite (Znw) from Zinnwald. The Raman spectrum of this crystal is in Figure 12.
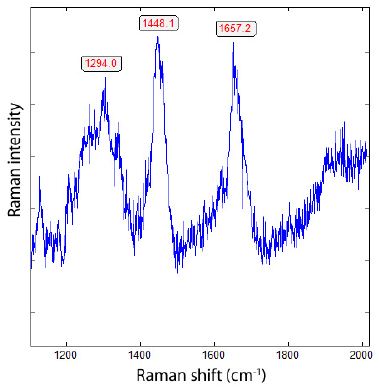
Figure 12: Raman spectrum of boron carbide and maybe a diamond grain in zinnwaldite. The Raman band 1294 cm-1 can be a hint to a 13C-rich diamond [18]. Exceptional are the strong 1448 and 1657 cm-1 bands corresponding to boron carbide.
Besides such irregular grains, there are spherical crystals of a mixture of BxC, BN, and Si3N4 with strong Raman bands between 400 and 1460 cm-1 (see Figure 13). Similar spherical crystals are in smoky quartz [1] from Zinnwald and in cassiterite from Ehrenfriedersdorf (Sn-58) – Thomas, 1982. Boron carbides form at high temperatures and are stable at high pressure (GPa-range) and are also stable under intense Raman laser irradiation (50mW).
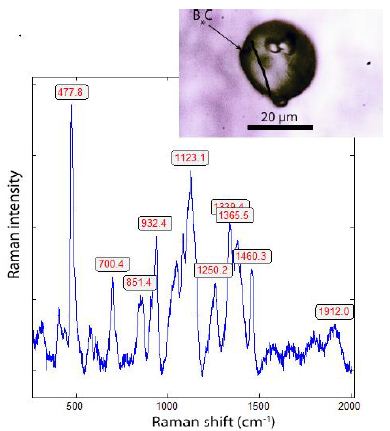
Figure 13: Raman spectrum of the spherical boron carbide BxC crystal (shown at the top right) in zinnwaldite (Znw).
In the low-frequency range, a band appears at 187 cm-1 (due to the B-C-B-C chain defect) as the pressure increases, as reported by Jay et al. (2023) [19]. After these authors, a significant kinetic barrier stabilizes the high-temperature configuration. Furthermore, in nature, it is probably a stoichiometric composition, not realistically – it is more a family of compounds of different compositions. However, the Raman spectra of such globules are more or less identical in zinnwaldite, quartz, and cassiterite. Very frequently, black films of graphite are observed. Figure 14 shows such aggregates.

Figure 14: Thick graphite (Gr) film in zinnwaldite (Zwn) from Zinnwald.
The Raman spectrum of such graphite is shown in Figure 15. Opposite to the graphite from Madagascar is FWHM (Full Width at Half Maximum), noticeably larger (see Table 2).
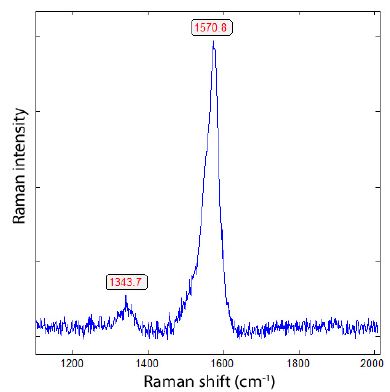
Figure 15: Raman spectrum of a graphite film in zinnwaldite (Figure 14), characterized by a strong Raman band at 1570.8 cm-1. This value corresponds to approximately 16.5% 13C.
Table 2: Results of the Raman measurements on the graphite main peak (G band).
|
Sample |
G band | FWHM |
n (measured crystals) |
| Graphite in Zwn |
1567.9 ± 4.6 |
21.8 ± 2.8 | 14 |
|
Gr from Madagascar |
1580.2 ± 1.1 | 15.3 ± 1.9 |
14 |
Gr – graphite, Zwn – zinnwaldite.
Besides graphite, there are also inclusions filled with abiotic organic material (Figure 16).
Table 2 shows the Raman data for the graphite-G peak. In most cases, the D band, at approximately 1345 cm-1, is mostly absent (Figure 16).
Figure 16 shows the inclusion of abiotic organic material in zinnwaldite. It is a complex mixture of saturated hexane (Author Collective, 1987 – [20]; Hurai et al., 2015 – [21]; Thomas, 2024d – [22]), produced by a natural Fischer-Tropsch process with zinnwaldite as the catalyst.

Figure 16: Complex abiotic organic material in zinnwaldite (Zwn) from Zinnwald.
Fluorite
In zinnwaldite, purple fluorite crystals (~5 mm in diameter) are often present. Thomas and Trinkler (2024) [4] found spherical lonsdaleite in these crystals. Besides the frequent lonsdaleite crystals, another rare diamond type is also present (Figure 17), characterized by two diamond Raman bands corresponding to high 13C contents.
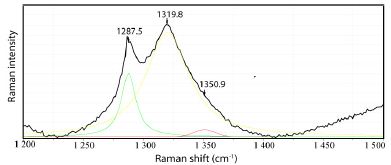
Figure 17: Diamond in a fluorite crystal in zinnwaldite from Zinnwald/Saxony. The main band lies at 1313.7 ± 3.3 cm-1 (10 measurements), which corresponds to 46.5% 13C.
Lonsdaleite is more frequently in the fluorite sample out of the zinnwaldite. Figure 18 shows the lonsdaleite crystal (a) and the Raman spectrum (b) from this crystal. In Table 3, the Raman-active vibrational modes are shown [23].
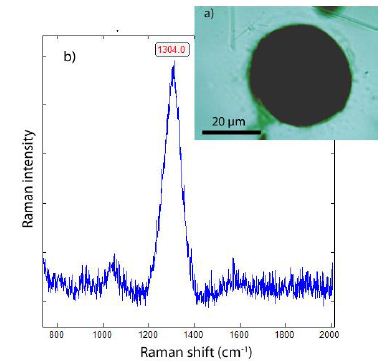
Figure 18: Lonsdaleite in fluorite from Zinnwald. a) Photomicrograph of lonsdaleite in fluorite. b) Raman spectrum of lonsdaleite. The Gaussian fit gives three components typical for lonsdalite.
Table 3: Raman-active vibrational modes.
|
Raman mode |
Band position (cm-1) |
FWHM (cm-1) |
| E2g |
1245.6 |
36.1 |
|
A1g |
1308.4 |
79.3 |
| E1g |
1356.5 |
24.4 |
According to Goryainov et al. (2018) [23], the three Raman-active vibration modes E2g (here 1245.6 cm-1), A1g (1308.6 cm-1), and E1g (1356.5 cm-1) are present.
The high 13C values of diamond in fluorite from Zinnwald spoke for a late supercritical input. The first input must be 12C richer.
Altenberg Tin Deposit
Not far from the Zinnwald deposit (~5 km) is the famous, however, closed Variscan tin deposit Altenberg [9,24]. Because there are also clear hints of supercritical fluids/melts present, the first data from that source should be given here in brief. Table 4 shows the results for diamonds found in quartz from pycnite rock. Pycnite is a variety of topaz, and the rock with the same name is composed of pycnite-topaz (44.2 ± 9.7), quartz (28.7 ± 8.8), and Li-mica (27.1 ± 8.0 %(vol/vol). The pycnite-topaz contains 19.8 % F [25].
Table 4: Raman spectroscopic data of diamond and related carbon of the diamond in the Altenberg pycnite rock; measurements on 18 crystals.
|
Sample |
Diamond (cm-1) | FWHM | Carbon (cm-1) | FWHM |
n |
| Pycnite-Qtz |
1324.2 ± 10.2 |
74.8 ± 18.0 | 1585.8 ± 7.6 | 57.0 ± 7.8 | 18 |
|
Pycnite |
– | – | 1572.2 ± 7.1 | 20.8 ± 5.2 |
12 |
n – number of studied crystals.
The Altenberg example demonstrates that the pycnite cupola in the mine is the result of the interaction of a primary pegmatitic (quartz- feldspar) stock with an F-rich supercritical fluid into the pycnite rock. Remnants of potassium feldspar are present. Such interaction is also valid for the entire Greisen body.
Interpretation
In this contribution, further data are presented, which underline the interpretation of the interaction of supercritical fluid and/ or melts coming from the Earth’s mantle with the more crustal granites forming the granite-related Variscan tin mineralizations. The more or less subparallel with the granite cupola from Zinnwald [9] proceeding mineralizations contain diamond, graphite, boron carbide, and orthorhombic cassiterite, which are clear high-pressure and high-temperature indicators. Additionally, the formation of diamond whiskers in smoky quartz [1] clearly demonstrates that not all high-pressure minerals were transported with supercritical fluids. They crystalized on the spot. The finding of such minerals also in typical greisen formations (zinnwaldite) demonstrates the significant influence of supercritical fluids, which bring a lot of water, solid indicator minerals, and dissolved ore elements for the formation of greisen and pegmatite bodies. These scientific proofs necessitate a new interpretation of the formation of this type of Variscan tin mineralization. From our studies, it is clear that supercritical fluids or melts have a significant impact on ore and pegmatite formation. At the transition of the supercritical fluid into the critical and undercritical fluids at the formation site of the Variscan mineralizations, processes occur that are not yet well understood. The example of Zinnwald shows that such processes occur over a large spatial area, demonstrating that also the input of supercritical fluids is not only spatial restricted. Figure 19 shows the pressure-temperature diagram for the equilibria of diamond and graphite, and coesite and β-quartz.
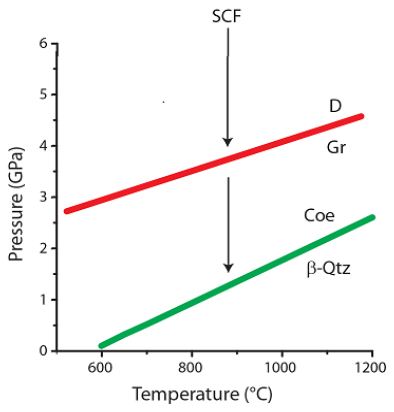
Figure 19: Schematic phase diagram of diamond and graphite as well as between coesite and β-quartz. SCF – supercritical fluid, D – diamond, Gr – graphite, β-Qtz – high-quartz, Coe – coesite.
The transport of diamonds, as an example, with the supercritical fluid is not the problem. However, the formation of diamonds, for instance, as whiskers at a crustal level with about 0.1 to 0.3 GPa is, at the moment, a significant problem. One explanation is that during the sudden change from the supercritical state to the critical state, an enormous pressure rise occurs. Conceivable is, however, a more slow transition. Another possibility is that processes like cavitation have a significantly detrimental effect. However, cavitation processes are more chaotic. Here is already a vast playground of sophisticated research.
Discussion
In the last years, starting from 2023, the author and coauthors have found in minerals of Variscan tin deposits of the Erzgebirge (Germany), Krušné Hory (Czech Republic), and Slakoský les (Kaiserwald) in the Czech Republic high-pressure and high-temperature minerals in more crustal regions (pressure about 0.1 to 0.3 GPa. These minerals belong to diamond, lonsdaleite, moissanite, complex boron carbides, orthorhombic cassiterite, coesite, cristobalite-X-I, as well as beryl-II. Studied were the deposits Zinnwald, Altenberg, Cinovec, Krupka, Sadisdorf, Ehrenfriedersdorf, the granite from Greifenstein, granites from Annaberg, Schlaggenwald (Slavkovský les). Because samples can be contaminated during preparation [26-31] with diamond and SiC (moissanite), the samples were carefully cleaned, and only diamonds were used, which are far enough below the surface. The discovery of diamond- and moissanite whiskers in these samples enhanced the reliability of our findings. Furthermore, the proof of extremely 13C-rich diamonds and graphite in the studied samples clearly demonstrates the reality of our findings.
Acknowledgment
This contribution is dedicated to Adolf Rericha for his encouragement and his intense discussions of the problems related to the meaning of supercritical fluids.
References
- Thomas R (2025a) Diamond, diamond whisker, graphite, carbon, and coesite in a quartz crystal from Zinnwald, E-Erzgebirge. Geol Earth Mar Sci 7: 1-6.
- Thomas R (2025b) Extremely 13C-rich diamond in orthorhombic cassiterites in the Variscan Erzgebirge, Saxony/Germany. Geol Earth Mar Sci 7: 1-5.
- Thomas R (2025c) SnF2-rich supercritical fluid in the Variscan tin deposit Zinnwald/ Saxony, Germany (2025b). Geol Earth Mar Sci 7: 1-5.
- Thomas R, Trinkler M (2024) Monocrystalline lonsdaleite in REE-rich fluorite from Sadisdorf and Zinnwald/E-Erzgebirge, Geol Earth Mar Sci 6(4): 1-5.
- Thomas R, Rericha A (2024) Extreme element enrichment, according to the Lorentzian distribution at the transition of supercritical to critical and under-critical melt or fluids. Geol Earth Mar Sci 6: 1-6.
- Thomas R, Rericha A (2025) Extreme element enrichment by the interaction of supercritical fluids from the mantle with crustal rocks. Minerals 15: 1-10.
- Shea Mj, Wall MH (2012) Representative Raman measurements of carbon nanotubes. Spectroscopy Europe 14: 14-16.
- Baumann L, Kuschka E, Seifert T (2000) Lagerstätten des Enke Stuttgart. Pg: 300.
- Breiter K (2014) 800 years of mining activity and 450 years of geological research in the Krušné Hory/Erzgebirge Mountains, Central Boletim do Museu Parense Emílio Goeldi. Ciȇncias Naturalis 9: 105-134.
- Qiao S, John T, Loges A (2024) Formation of topaz-greisen by boiling fluid: A case study from the Sn-W-Li deposit, Zinnwald/Cinovec. Economic Geology 119: 805-828.
- Webster J, Thomas R, Förster H-J, Seltmann R, Tappen C (2004) Geochemical evolution of halogen-enriched granite magmas and mineralizing fluids of the Zinnwald tin-tungsten mining district, Erzgebirge, Germany. Mineralium Deposita 39: 452-472.
- Thomas R, Förster H-J, Rickers K, Webster JD (2005) Formation of extremely F-rich hydrous melt fractions and hydrothermal fluids during differentiation of highly evolved tin-granite magmas: a melt/fluid-inclusion study. Contrib Mineral Petrol 148: 582-601.
- Thomas R (2024a) REE-rich fluorite in granite from Zinnwald/East Erzgebirge/Germany. Geol Earth Mar Sci 6: 1-5.
- Werheit H, Filipov V, Kuhlmann U, Schwarz U, Armbrüster M, et al. (2010) Raman effect in icosahedral boron-rich solids. Science and Technology of Advanced Materials 11: 1-27.
- Thomas R, Davison P, Rericha A, Recknagel U (2023) Ultrahigh-pressure mineral inclusions in a crustal granite: Evidence for a novel transcrustal transport mechanism. Geosciences 13: 1-13.
- Cebik J, McDonough JK, Peerally F, Medrano R, Neitzel I, et (2013) Raman spectroscopy study of the nanodiamond-to-carbon onion transformation. Nanotechnology 24: 1-10.
- Zakalyukin RM, Levkevich EA, Kumskov AS (2023) One-dimensional NaSn2F5 crystals inside single-walled carbon nanotubes. Crystals 13: 1-15.
- Zaitsev AM (2001) Optical Properties of Diamond. A Data Handbook. Springer, I-XI, and 1-502 pages.
- Jay A, Duparc OH, Sjakste J, Vast N (2023) Theoretical Raman spectrum of boron carbide B4.3C under pressure. Acta Materialia 255: 1-11.
- Author Collective (1987) Spectral Database for Organic Compounds, SDBS, National Institute of Advanced Industrial Science and Technology (AIST), Japan.
- Hurai V, Huraiová M, Slobodník M, Thomas R (2015) Geofluids – Developments in Microthermometry, Spectroscopy, Thermodynamics, and Stable Isotopes. Elsevier.
- Thomas R (2024d) Another fluid inclusion type in pegmatite quartz: Complex organic compounds. Geol Earth Mar Sci 6: 1-5.
- Goryainov SV, Likhacheva AY, Ovsyuk NN (2018) Raman scattering in lonsdaleite. Journal of Experimental and Theoretical Physics 127: 20-24.
- Weinhold G (2002) Die Zinnerz-Lagerstätte Altenberg/Osterzgebirge. Bergbaumonographie 9, Freiberg 274 + X pages.
- Thomas R, Davidson P (2013) The missing link between granites and granitic pegmatites. Journal of Geosciences 58: 183-200.
- Keller DS, Ague JJ (2022) Possibilities for misidentification of natural diamond and coesite in metamorphic rocks. Neues Jb. Mineral. Abh 197: 253-261.
- Blank VD, Kulnitsky BA, Rerezhogin IA, Tyukalova EV, Denisov VN, et (2016) Graphite-to-diamond (13C) direct transition in a diamond anvil high-pressure cell. Int. J. Nanotechnol 13: 604-611.
- Enkovich PV, Brazhkin VV, Lyapin SG, Novikov AP, Kanada H, et (2016) Raman spectroscopy of isotopically pure (12C, 13C) and isotopically mixed (12.5C) diamond single crystals at ultrahigh pressures. Journal of Experimental and Theoretical Physics 123: 443-451.
- Thomas R (1982) Ergebnisse der thermobarometrischen Untersuchungen an Flüssigkeitseinschlüsse in Mineralen der postmagmatischen Zinn-Wolfram- Mineralisation des Freiberger Forschungshefte C370. Pg: 85.
- Thomas R (2024b) The SnO2 -to-rutile phase transition in SnO2 from high to low pressure in nature. Geol Earth Mar Sci 6: 1-4.
- Thomas R (2024c) Rhomboedric cassiterite as inclusions in tetragonal cassiterite from Slavkovský les – North Bohemia (Czech Republic). Geol Earth Mar Sci 6: 1-6.