Abstract
The aim of the study was to evaluate the use of narrow implants (3.25 mm) to replace upper lateral and lower incisors according to an established immediate function protocol. A total of 49 narrow implants (Neoss Proactive 3.25 mm implants, Neoss Ltd, Harrogate, UK) in 35 patients were evaluated. Thirty-one implants were placed in the mandible and 18 in the maxilla. The mean insertion torque was measured in Ncm. Thirty-six implants were placed in fresh extraction sockets. Implant stability measurements were performed at baseline, after 2, 4, 6 weeks and 3, 6 months using resonance frequency analysis (RFA) measurements (Osstell ISQ™, Osstell AB, Gothenburg, Sweden) expressed in ISQ units (Implant Stability Quotient). The patients were followed with clinical and radiographic examinations for 18 months. One implant failed after 4 weeks giving a cumulative survival rate of 98.0 % and the marginal bone loss amounted to 0.7 + 1.0 mm after 18 months. The mean insertion torque was 36 + 9.1 Ncm. The mean ISQ values indicated firm stability at baseline in both mesial-distal and buccal-lingual directions (i.e. above 65 ISQ). The ISQ curve presented a significant drop after 2–4 weeks where after the stability recovered progressively up to 6 months.
It is concluded that upper lateral and lower incisors can be replaced with 3.25 mm implants according to an immediate loading protocol with high survival rate and minimal marginal bone loss. Moreover, the immediately loaded implants showed an initial dip of stability during the first 4 weeks followed by an an increase with time.
Keyword
Immediate Loading, Implant Stability, Narrow Implants, Resonance Frequency Analysis
Introduction
Immediate function was originally used to treat edentulous mandibles by placing conventional diameter (>3.75 mm) dental implants in the mandibular symphysis region, which is an area of dense bone offering high implant stability [1, 2]. Later, the concept of immediate function has successfully been applied to areas with lower bone density, in part depending on the development of new implant designs and surfaces aiming at high primary stability and rapid integration [3–5]. However, there are areas of the jaws in which it is difficult to place implants with a conventional diameter because of the small size of the teeth to be replaced. These areas are located the lower incisors and upper lateral incisors. In these cases, an Implant with standard diameter may result in an excessive proximity with neighbouring teeth, with possible damage to the teeth themselves or with lack of space for the osseointegration process [6]. Even the emergence profile from the soft tissues and the morphology of the papilla may be adversely affected by a diameter of the implant too large compared to the size of the origin tooth. Hence, the use of Implants with reduced diameter and reduced platform is a viable solution for the treatment of the lower incisors and upper lateral incisors, where the available space does not allow for the use of conventional diameter implants. An implant is considered of small diameter when this it is less than 3.5mm. The installations of small diameter (SDI) should not be confused with the mini-implants, characterized by a diameter of less than 3mm and a structure in one piece, and are generally used in orthodontics as anchorage [7]. The reliability of small diameter implants has been demonstrated in numerous clinical studies [8]. However, fractures of the implant body due to long-term fatigue following the load have been described for some implant types [9, 10]. Narrow 3.3 mm implants have been reported to be successful when loaded 6–10 weeks after surgery [11] as well as when loaded within 48 hours [12].
The purpose of the present work was to evaluate the implant survival rate of 3.25mm diameter implants with reduced platform, positioned in areas of the lower incisors and upper lateral incisors, and subject to a previously evaluated immediate function protocol [13–16]. Further aims were to analyse the marginal bone resorption and the behaviour of the implant stability during loading and healing as assessed by Resonance Frequency Analysis (RFA) measurements.
Material and Methods
Patient selection
A total of 35 patients (12 females and 23 males; mean age 57 years, range 16 – 87) treated with a previously established immediate implant function protocol were included in the study [13–16]. The inclusion criteria were: (i) need of implant-supported crown or bridge in the mandible incisor area or single restoration at the lateral incisor in maxilla, (ii) need, for aesthetic reasons, of the immediate restoration of the lacking teeth, (iii) available bone for at least 11 mm long and 3.25 mm wide implants. The exclusion criteria were: (i) non-compensated general diseases, (ii) poor oral hygiene, (iii) presence of acute inflammation at the teeth expected to extract. Smoking, bruxism and periodontal disease were considered as risk factors and recorded. Patients with active periodontitis were treated before implant surgery according to conventional periodontal therapy. Immediate placement of implants in extraction sockets was allowed. The study was conducted in full accordance with ethical principles, including the World Medical Association Declaration of Helsinki. All patients were carefully informed about the procedure and gave their written consent to participate and to follow a maintenance and observation program for 18 months. They could at any time point refuse further participation.
Implants
A total of 49 narrow diameter (3.25 mm) implants (Proactive Straight™, Neoss Ltd, Harrogate, UK) had been inserted in the 35 patients; 31 in mandible and 18 in maxilla (Table 1). Apart from small diameter (3.25mm) this implant has a small prosthetic platform. The implant is characterized by a positive tolerance, signified by a slightly tapered geometry. The surface (Proactive™) is prepared by blasting with titanium particles followed by acid etching and chemically modified to reduce surface tensions and to exhibit electro-wetting in contact with fluids. The Sa value at the implant body is some 0.8–1 μm for the Proactive surface. According to the manufacturer, the roughness is higher on the body and less at the neck of the implant.
Table 1. Position and length of implants.
|
Position and length of implants |
||||
|
Maxilla (n = 18) |
||||
|
Position |
12 |
22 |
||
|
11mm |
1 |
– |
||
|
13mm |
5 |
7 |
||
|
15mm |
2 |
3 |
||
|
Mandible (n = 31) |
||||
|
Position |
42 |
41 |
31 |
32 |
|
13mm |
5 |
1 |
1 |
6 |
|
15mm |
9 |
1 |
1 |
7 |
Surgical and prosthetic procedures
The patients were given one gram of amoxicillin prior to implant surgery. After local anaesthesia, a mid-crestal incision was performed in edentulous sites or, in case of presence of residual teeth, a para-marginal incision was carried out in order to eliminate the internal portion of the gingival sulcus. A full thickness flap, without any releasing incisions, was elevated, and the positions of the implants were marked with a round bur. Then, the receiving sites were prepared with cylindrical burs of increasing diameter, according to the recommendations of the manufacturer (2.2 mm and 2.85 mm as the last burr). In the presence of soft bone, an under-preparation technique was used with 2.2mm as final diameter. In order to preserve as much cortical bone as possible, the use of countersink was avoided. In selected cases a flapless procedure was adopted. The implants were placed with the implant collar “below the crest” (BC), “flush to the crest” (FC) or “above the crest” (AC), depending on width and high of the gingival tissues. In immediate post extractive sites, careful curettage of the socket was performed just after the extraction of the tooth in order to remove any residual inflammatory tissue or periodontal ligament. For this purpose round burr or piezosurgery (Piezosurgery, Mectron, Genova, Italy) device were used. The residual gaps adjacent to the implants were classified as “closed defect”, if all socket bone walls were conserved, or “open defect”, if one or more bone walls were lacking. Closed defects, as they are containing defects, were treated only with auologous bone graft, whereas open defects, when they were “non spacemaking”, with a combination of grafts and resorbable membranes.
After the complete seating of the implants, healing caps were screwed on and the flaps sutured using interrupted sutures. Then, the healing cap were unscrewed and the titanium components for the temporary prosthesis screwed. A temporary resin prosthesis, arranged previously from the laboratory, was then adapted to the components and relined by self-curing resin. After final finishing the prosthesis was screwed on the implants. No occlusal contacts, centrally, laterally and in protrusion, were allowed on the temporary prostheses. The patients received postsurgical antibiotic therapy (amoxicillin, Zimox®, Pfizer Italia Srl, Latina, Italy ), 1g, twice a day for 6 days, starting just before surgery, an anti inflammatory therapy, (nimesulide, Aulin®,Helsinn Birex Pharmaceuticals Ltd, Dublin, Ireland)), twice a day, for 4 days and they were instructed to rinse with a solution of chlorexidine at 2%, twice a day for 10 days.
Radiographic examination
Intraoral radiographs were taken after insertion of the implant (baseline), and then after 1 month, 6 months and 18 months from the installation of the implant using a paralleling technique (Dentsply RINN, Elgin, Il. USA). The technique to make correct radiographies was the following: a pin (the transfer’s one) was screwed to the implant, the Rinn collimator was positioned and supported by cotton rolls to avoid any incorrect inclination, then the x-ray was taken. The radiographs were examined by an independent radiologist. The upper corner of the coronal shoulder of the implant was used as reference point. Measurements from the reference point to the first bone contact at the mesial and distal aspects of the implant were performed. A mean value was calculated for each implant and time point.
Resonance frequency analysis (RFA)
Implant stability measurements were performed at baseline, after 2, 4, 6 weeks and 3, 6 months using resonance frequency analysis (RFA) measurements (Osstell ISQ™, Osstell AB, Gothenburg, Sweden) expressed in ISQ units (Implant Stability Quotient). For each implant, two measurements, one in mesio-distal and one in bucco-lingual direction were made.
Implant survival criteria
An implant was considered a survival if clinically stable and complying with the function of supporting the prosthesis and causing no discomfort to the patient. Failure was defined as removal of an implant due to any reason.
Results
Clinical findings
All 49 implants, installed in 35 patients, were followed for 18 months with no drop-outs (Figures 1a to e). Thirty-six implants were positioned in immediate post-extraction sites. Seven of these filled almost completely the sockets and did not require any regenerative procedure. The remaining 29 implants presented with an adjacent bone defect after placement. All “closed defects” (n=17) were filled with autologous bone particles collected in the neighbouring areas. Part of the “open defects” (n=5), since they were containing, were treated only with autologous graft, whereas the remaining (n=6) with a combination of autologous graft and resorbable membrane or with (n=1) a mix of autologous bone, bovine bone and resorbable membrane. One of the 49 implants failed after 4 weeks giving a cumulative survival rate of 98.0 % after 18 months. The failed implant was placed in an immediate extraction socket at lower lateral incisor. The baseline ISQ value was 79, but after 2 weeks had already fallen to 66 and at 4 weeks the implant was symptomatic, with swelling and pain. At that point the implant was removed and the bone reconstructed by autologous bone and resorbable membrane. During the following healing period the contralateral implant supported a four elements temporary bridge, ensuring an adequate aesthetics. Three months later another implant was placed in the same position of the failed one, it was successful and could be used for the final restoration.
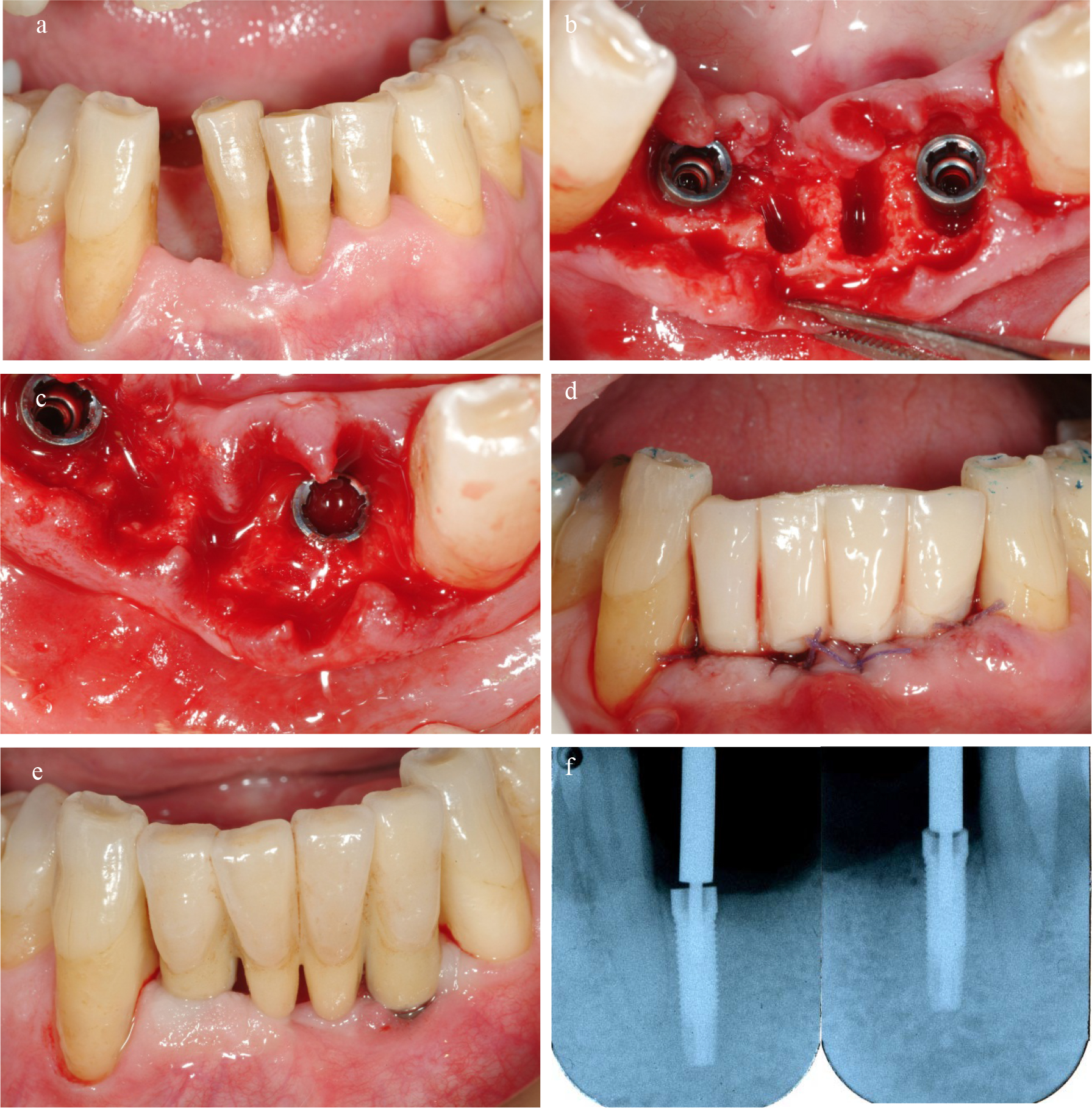
Figure 1. a/Lower incisors suffering from severe periodontal disease with extensive loss of bone support. b/Teeth were extracted and 3.25 diameter implants placed into extraction sockets. c/The remaining gap adjacent to the implants were filled with autologous bone particles. d/Temporary bridge were immediately connected to the implants. e/The final restoration. f/ Radiographs after 18 months.
Radiographic findings
The radiographic measurements showed the bone level at baseline to be 0.7 + 0.6 mm apical to the implant platform and 1.2 + 0.9 mm and 1.4 mm + 0.8 after 6 and 18 months, respectively (Table 2 and 3). The marginal bone loss after 18 months amounted to 0.7 mm + 1.0 and 88% of the total number of implants presented a marginal bone loss not exceeding 1.9mm after 18 months. Only six implants showed a bone resorption more than 2mm. (Table 4). Most of the marginal bone resorption occurred during the first 6 months (0.5mm), with only 0.2 mm for the remaining 12 months (Figure 2).
Table 2. Marginal bone level measurements.
|
Bone level (mm) |
|
|
Baseline (mm + SD) |
0.7 + 0.6 |
|
6 months (mm + SD) |
1.2 + 0.9 |
|
18 months (mm + SD) |
1.4 + 0.8 |
Table 3. Frequency distribution of marginal bone levels.
|
Baseline |
18 months |
|
|
Bone level (mm + SD) |
0.7 + 0.6 (n=49) |
1.4 + 0.8 (n=49) |
|
Frequency distribution (mm) |
No (%) |
No (%) |
|
0 |
9 (18.0) |
0 |
|
0.1–0.9 |
25 (50.0) |
20 (40.0) |
|
1.0–1.9 |
14 (28.0) |
18 (36.0) |
|
2.0–2.9 |
1 (2.0) |
10 (20.0) |
|
>3.0 |
0 |
2 (4.0) |
Table 4. Frequency distribution of marginal bone loss measurements.
|
Bone loss baseline to 18 m (mm + SD) |
0.7 + 1.0 (n=49) |
|
Frequency distribution (mm) |
No (%) |
|
<0 |
10 (20.0) |
|
0–0.9 |
22 (44.0) |
|
1.0–1.9 |
12 (24.0) |
|
>2.0 |
6 (12.0) |
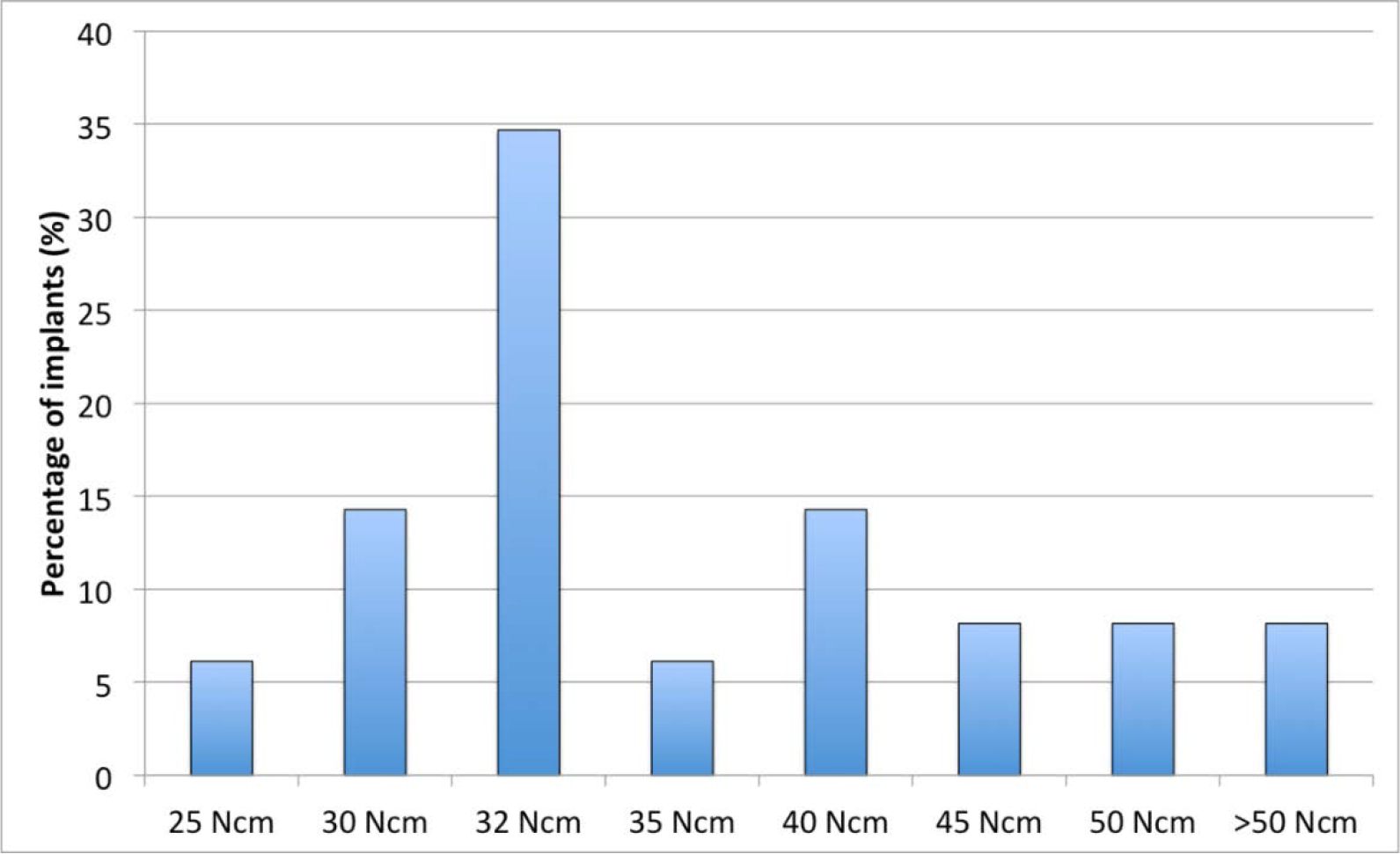
Figure 2. Distribution of insertion torque at implants placement.
Implant stability
The mean insertion torque was 36 (SD 9.1) Ncm (range 25–60 Ncm) (Figure 3). The mean ISQ values indicated firm stability at baseline in both mesial-distal and buccal-lingual directions (i.e. above 65 ISQ. The ISQ curve presented a significant drop after 2–4 weeks, then the stability recovered progressively up to 6 months (Figure 4).
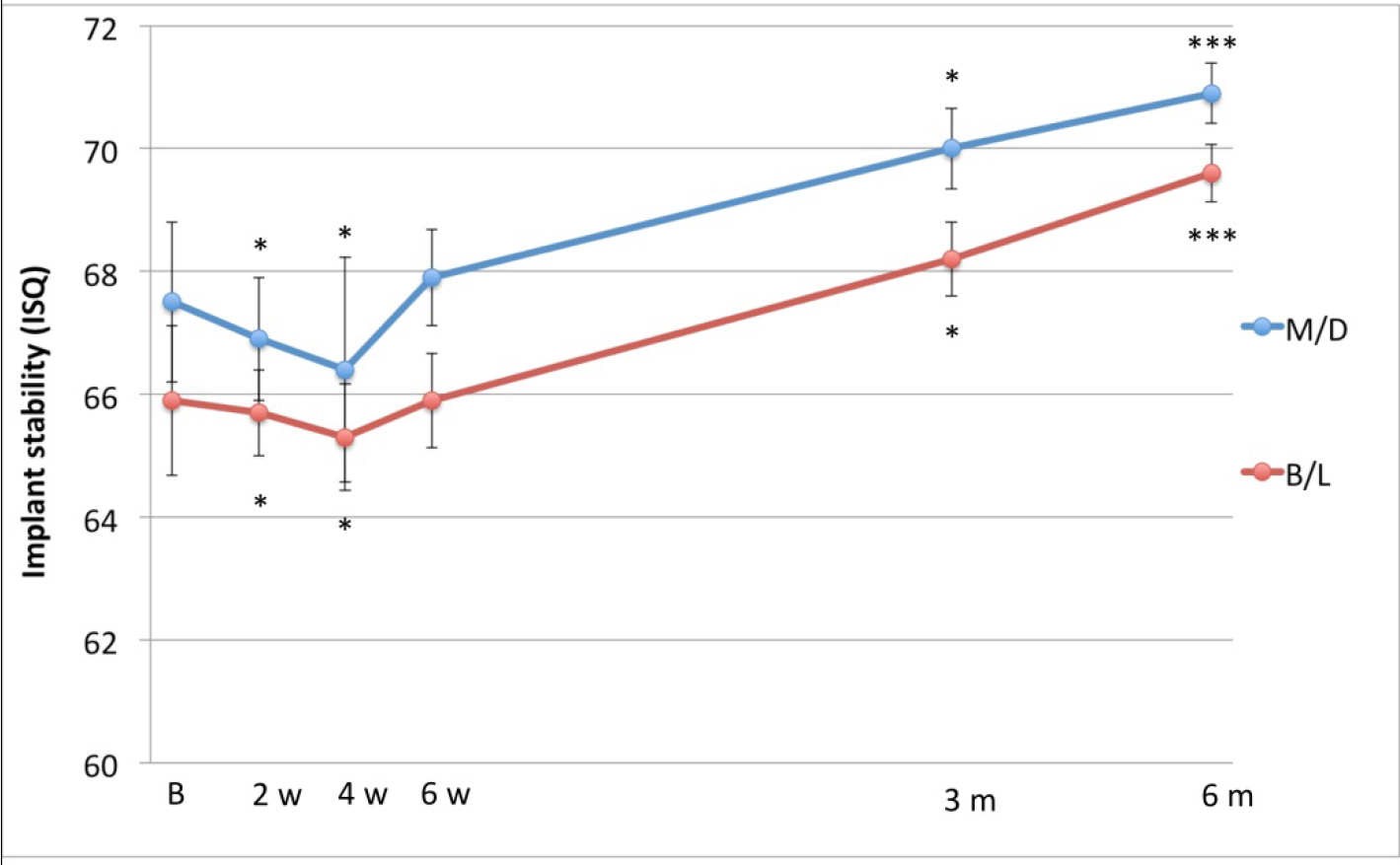
Figure 3. Time-stability curve based on ISQ measurements taken in mesio-distal and bucco-lingual directions.
Discussion
The present study demonstrated the possibility of immediate function in aesthetic areas with limited interproximal space and narrow alveolar crests (lower incisors and upper lateral incisors) using 3.25 mm implants. Only one of 49 implants were lost (2%) and minimal bone marginal bone resorption was seen during the 18 months of follow up. This is in line with Lambert and co-workers, who reported a 97.4% one-year survival rate for 39 narrow implants (3.3 mm) in 20 patients in both anterior and posterior areas with reduced thickness (< 6 mm) of the alveolar crest [12]. In a multicentre study, 97 narrow implants (3 mm) were placed in 69 patients and loaded after 6–10 weeks with a permanent fixed prosthesis [11]. The survival rate was 95.5 % after 3 years with stable marginal bone levels, which is in line with the findings from the present study. A systematic review of the literature showed an overall survival rate of 97.2 % for 672 narrow implants with a diameter of 3.0 to 3.25 mm, which further supports the idea that the use of narrow implants is an effective treatment option [8].
Firm primary implant stability is considered to be the most important factor for successful osseointegration [17]. Insertion torque (IT) is commonly used as a parameter of stability but gives only one measurement at placement surgery. The RFA technique on the other hand is a non-invasive method to assess implant stability at any time of implant treatment and follow-up as supported by numerous publications [18, 19]. In the present study, a series of RFA measurements were obtained at different time points following surgery (baseline, 2,4,6 weeks, 3,6 months). Hence, a stability curve could be plotted, which represents the stability conditions for each implant over the whole healing period. The mean baseline value in mesial-distal direction was 67.5 ISQ in the present study, which is similar to the 68 ISQ achieved in a previous study where standard diameter Neoss implants (4mm) were used [16]. During the weeks following implant installation the ISQ curve showed a drop followed by a recovery after 6-7 weeks up to the initial values, which is in line with previous studies [15, 16, 20, 21]. In the further period the stability continued to raise over the observation period (18 months). Also the shape of the stability curve was similar to that recorded in the mentioned above study with standard Neoss implants. The fall of stability after implant installation can be attributable to the surgery-induced inflammatory process and initial bone resorption, which is part of the repair process. When the inflammation decreases and the new bone formation takes place and stabilizes the interface, the ISQ values augment progressively. A previous study demonstrated the implant surface to be important for the development of stability during immediate loading [20]. The failed implant in the present study showed a significant reduction of stability after 2 weeks but the values were still in the security range. Unfortunately, the measurements after 4 weeks, along with an evident symptomatology, showed a rapid a dramatic loss of stability that led to implant failure. Thus, in this case the stability curve could not be used to save the implant by unloading it due to the rapid progress of stability loss, which was the case in a previous study [15], probably due infection.
The possibility to apply a temporary prosthesis to an implant placed in a fresh extraction socket to immediately or early after surgery has been previously demonstrated in many studies. For instance, Vanden Bogaerde (2005) placed 50 oxidized surface implants directly into fresh extraction sockets and applied a function the same day (immediate function) or within 7 days (early function) [15]. None of the 50 installed implants had failed at the end of the 18-month observation period, giving an implant survival rate of 100%. At the end of the observation period the mean marginal bone resorption in the total group was 0.9 mm. Of the 38 implants regularly examined by RFA, 19 showed no significant variations in stability from baseline to the 6-month follow-up, whereas 15 showed an increasing stability over time. Early loaded implants inserted into fresh extraction sockets were retrospectively analysed in a study by Nordin et al. [22]. The authors placed 116 implants, 77 of which into fresh, extraction sockets and 39 in healed bone. One hundred and ten implants were loaded by permanent fixed complete dentures within 10 days after placements and six after 14 days. Two implants were lost, giving a 98% of implant survival rate. The radiographic measurements after 2–3 years did not reveal any difference in marginal bone height at the implants placed in extraction sockets vs. in healed bone.
It is concluded that upper lateral and lower incisors can be replaced with 3.25 mm implants according to an immediate loading protocol with high survival rate and minimal marginal bone loss. Moreover, the immediately loaded implants showed an initial dip of stability during the first 4 weeks followed by an an increase with time.
References
- Henry P, Rosenberg J (1994) Single-stage surgery for rehabilitation of the edentulous mandible: preliminary results. Pract Periodontics Aesthet Dent 6: 1–8. [crossref]
- Ericsson I, Randow K, Nilner K, Petersson A (2000) Immediate functional loading of Brånemark dental implants. A 5-year clinical follow-up study. Clin Implant Dent Relat Res 2: 70–77. [crossref]
- Glauser R, Rèe A, Lundgren AK, Gottlow J, Hämmerle C, Schärer P (2001) Immediate occlusal loading of Brånemark implants applied in various jawbone regions: a prospective, 1-year clinical study. Clin Implant Dent Relat Res 4: 204–213. [crossref]
- Grunder U (2001) Immediate functional loading of immediate implants in edentulous arches: two-year results. Int J Periodontics Restorative Dent 21:545–551. [crossref]
- De Bruyn, H, Raes S, Östman PO, Cosyn J (2014) Immediate loading in partially and completely edentulous jaws: a review of the literature with clinical guidelines. Periodontol 2000 66: 153–87. [crossref]
- Froum SJ, Cho SC, Cho YS, Elian N, Tarnow D (2007) Narrow-diameter implants: a restorative option for limited interdental space. Int J Periodontics Restorative Dent 27: 449–455. [crossref]
- Motoyoshi M, Hirabayashi M, Uemura M, Shimizu N (2006) Recommended placement torque when tightening an orthodontic mini-implant. Clinical Oral Implants Research 17:109–114. [crossref]
- Klein MO, Schiegnitz E, Al-Nawas B (2014) Systematic review on success of narrow-diameter dental implants. Int J Oral Maxillofac Implants 29 Suppl: 43–54. [crossref]
- Zinsli B, Sagesser T, Mericske E, Mericske-Sterne R (2004) Clinical evaluation of small diameter ITI implants: a prospective study. Int J Oral Maxillofac Implants 19: 92–99. [crossref]
- Quek CE, Tan KB, Nicholls JI (2006) Load fatigue performance of a single-tooth implant abutment system: effect of diameter. Int J Oral Maxillofac Implants 21: 929–936. [crossref]
- Maiorana C, King P, Quaas S, Sondell K, Worsaae N, Galindo-Moreno P (2015) Clinical and radiographic evaluation of early loaded narrow-diameter implants: 3 years follow-up. Clin Oral Implants Res 26:77–82. [crossref]
- Lambert FE, Lecloux G, Grenade C, Bouhy A, Lamy M, et al. (2015) Less Invasive Surgical Procedures Using Narrow-Diameter Implants: A Prospective Study in 20 Consecutive Patients. J Oral Implantol 41: 693–699. [crossref]
- Vanden Bogaerde L, Pedretti G, Dellacasa P, Mozzati M, Rangert B (2003) Early function of splinted implants in maxillas and posterior mandibles using Brånemark System® machined-surface implants: an 18-month prospective clinical multicenter study. Clin Implant Dent Relat Res 5: 21–28. [crossref]
- Vanden Bogaerde L, Pedretti G, Dellacasa P, Mozzati M, Rangert B, et al. (2004) Early function of splinted implants in maxillas and posterior mandibles, using Brånemark System® TiUnite™ implants: an 18-month prospective clinical multicenter study. Clin Implant Dent Relat Res 6: 121- 129.
- Vanden Bogaerde L., Rangert B, Wendelhag I (2005) Immediate early function of Brånemark System® TiUnite™ implants in fresh extraction sockets in maxillae and posterior mandibles: an 18-month prospective clinical study. Clin Implant Dent Relat Res 7: 121–130. [crossref]
- Vanden Bogaerde L, Pedretti G, Sennerby L, Meredith N (2010) Immediate/Early Function of Neoss Implants Placed in Maxillas and Posterior Mandibles: An 18-Month Prospective Case Series Study Clin Implant Dent Relat Res 12: 83–94. [crossref]
- Gapski R, Wang HM, Mascarenhas P, Lang NP (2003) Critical review of immediate implant loading. Clin Oral Implants Res 14: 515–527. [crossref]
- Meredith N, Alleyne D, Cawley P (1996) Quantitative determination of the stability of the implant-tissue interface using resonance frequency analysis. Clin Oral Implants Res 7: 261–267. [crossref]
- Sennerby L, Meredith N (2008) Implant stability measurements using resonance frequency analysis: biological and biomechanical aspects and clinical implications. Periodontol 2000 47: 51–66. [crossref]
- Vanden Bogaerde L, Sennerby L (2016) A Randomized Case-Series Study Comparing the Stability of Implant with Two Different Surfaces Placed in Fresh Extraction Sockets and Immediately Loaded. Int J Dent2016: 8424931. [crossref]
- Glauser R, Lundgren AK, Gottlow J, Sennerby L, Portmann M, et al. (2003) Immediate occlusal loading of Brånemark TiUnite implants placed predominantly in soft bone: 1-year results of a prospective clinical study. Clin Implant Dent Relat Res 5 Suppl 1: 47–56. [crossref]
- Nordin T, Graf J, Frykholm A, Helldén L (2007) Early functional loading of sand-blasted and acid-etched (SLA) Straumann implants following immediate placement in maxillary extraction sockets. Clinical and radiographic result. Clin Oral Implants Res 18: 441–451. [crossref]